Vitamin D supplementation prior to or during COVID-19 associated with better 3-month survival in geriatric patients: Extension phase of the GERIA-COVID study
et al., The Journal of Steroid Biochemistry and Molecular Biology, doi:10.1016/j.jsbmb.2021.105958, Jul 2021
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
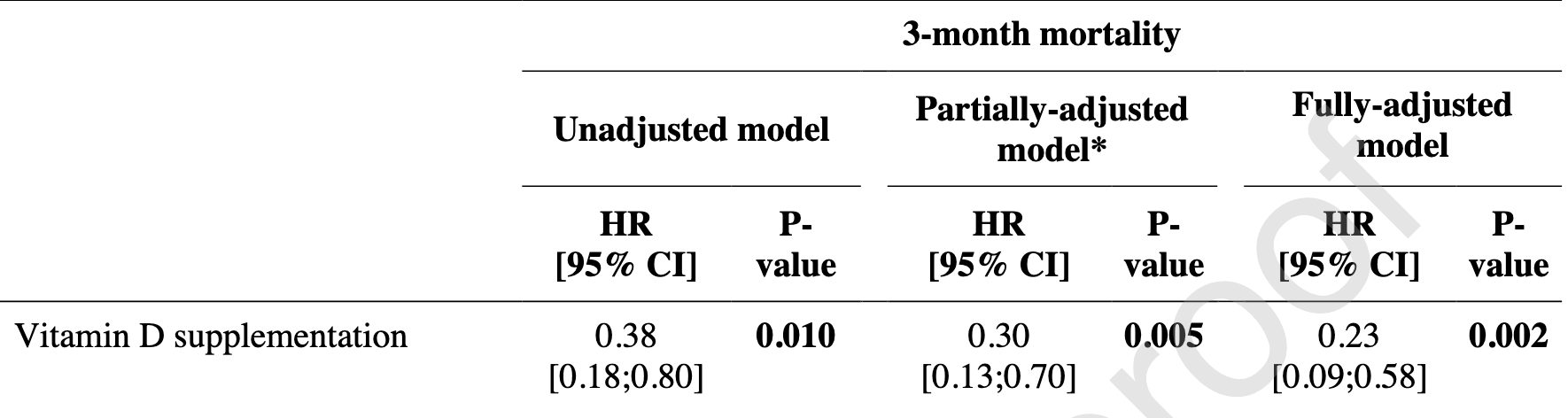
Report on extended results from the GERIA-COVID study, showing significantly lower mortality at 3 months with vitamin D treatment. Results combine prophylaxis and early treatment.
|
risk of death, 64.2% lower, RR 0.36, p = 0.002, treatment 16 of 67 (23.9%), control 13 of 28 (46.4%), NNT 4.4, adjusted per study, odds ratio converted to relative risk, multiple Cox proportional hazards.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Annweiler et al., 29 Jul 2021, prospective, France, peer-reviewed, 7 authors.
Vitamin D supplementation prior to or during COVID-19 associated with better 3-month survival in geriatric patients: Extension phase of the GERIA-COVID study
The Journal of Steroid Biochemistry and Molecular Biology, doi:10.1016/j.jsbmb.2021.105958
Background: The objective of this extension phase of the quasi-experimental GERIA− COVID study was to determine whether vitamin D3 supplementation taken prior to or during COVID-19 was associated with better 3month survival in geriatric patients hospitalized for COVID-19. Methods: Intervention group was defined as all participants supplemented with vitamin D3 prior to or during COVID-19 (n = 67). Supplements were either bolus vitamin D3 (ie, 50,000 IU per month, or 80,000 IU or 100,000 IU or 200,000 IU every 2-3 months), or daily supplementation with 800 IU. Comparator group involved those without vitamin D supplements (n = 28). Outcome was 3-month mortality. Covariables were age, sex, functional abilities, history of malignancies, cardiomyopathy, undernutrition, number of acute health issues, antibiotics use, systemic corticosteroids use, and 25(OH)D concentration. Results: 76.1 % (n = 51) of participants survived at 3 months in Intervention group, compared to only 53.6 % (n = 15) in Comparator group (P = 0.03). The fully-adjusted hazard ratio for 3-month mortality was HR = 0.23 [95 %CI: 0.09;0.58](P = 0.002) in Intervention group compared to Comparator group. Intervention group had also longer survival time (log-rank P = 0.008). Conclusions: Vitamin D3 supplementation was associated with better 3-month survival in older COVID-19 patients.
Declaration of Competing Interest CA occasionally serves as a consultant for Mylan Laboratories Inc (2020). All authors declare they do not have any other financial and personal conflicts of interest with this manuscript.
References
Annweiler, Cao, Sabatier, Point of view: Should COVID-19 patients be supplemented with vitamin D?, Maturitas
Annweiler, Corvaisier, Gautier, Vitamin d supplementation associated to better survival in hospitalized frail elderly COVID-19 patients: the GERIA-COVID quasi-experimental study, Nutrients
Annweiler, Hanotte, Grandin De L'eprevier, Sabatier, Lafaie et al., Vitamin D and survival in COVID-19 patients: a quasi-experimental study, J. Steroid Biochem. Mol. Biol
Annweiler, Legrand, Souberbielle, Vitamin D in adults: update on testing and supplementation, Geriatr. Psychol. Neuropsychiatr. Vieil
Annweiler, Mercat, Souberbielle, Learning from previous methodological pitfalls to propose well-designed trials on vitamin D in COVID-19, J. Steroid Biochem. Mol. Biol
Baktash, Hosack, Patel, Vitamin D status and outcomes for hospitalised older patients with COVID-19, Postgrad. Med. J, doi:10.1136/postgradmedj-2020-138712
Bishop, Ismailova, Dimeloe, Hewison, White, Vitamin D and immune regulation: anti-bacterial, antiviral, and anti-inflammatory, JBMR Plus, doi:10.1002/jbm4.10405
Bruyère, Deroisy, Dardenne, A phase IV, two-armed, randomized, cross-over study comparing compliance with once-a-month administration of vitamin D3 to compliance with daily administration of a fixed-dose combination of vitamin D3 and calcium during two 6-month periods, Osteoporos. Int
Castillo, Costa, Vaquero Barrios, Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study, J. Steroid Biochem. Mol. Biol
Dijkman, Jebbink, Deijs, Replication-dependent downregulation of cellular angiotensin-converting enzyme 2 protein expression by human coronavirus NL63, J. Gen. Virol
Glinsky, Tripartite combination of candidate pandemic mitigation agents: vitamin d, quercetin, and estradiol manifest properties of medicinal agents for targeted mitigation of the COVID-19 pandemic defined by genomics-guided tracing of SARS-CoV-2 targets in human cells, Biomedicines
Grant, Lahore, Mcdonnell, Evidence that vitamin d supplementation could reduce risk of influenza and COVID-19 infections and deaths, Nutrients
Hernández, Nan, Fernandez-Ayala, Vitamin d status in hospitalized patients with SARS-CoV-2 infection, J. Clin. Endocrinol. Metab, doi:10.1210/clinem/dgaa733
Hossein-Nezhad, Holick, Vitamin D for health: a global perspective, Mayo Clin Proc
Kong, Zhu, Shi, VDR attenuates acute lung injury by blocking Ang-2-Tie-2 pathway and renin-angiotensin system, Mol. Endocrinol
Martineau, Jolliffe, Hooper, Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data, BMJ
Murai, Fernandes, Sales, Pinto, Goessler et al., Effect of a single high dose of vitamin D3 on hospital length of stay in patients with moderate to severe COVID-19: a randomized clinical trial, JAMA
Raman, Cassar, Tunnicliffe, Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge, EClinicalMedicine
Rastogi, Bhansali, Khare, Short term, high-dose vitamin D supplementation for COVID-19 disease: a randomised, placebo-controlled, study (SHADE study), Postgrad. Med. J, doi:10.1136/postgradmedj-2020-139065
Rauf, Abu-Izneid, Olatunde, COVID-19 pandemic: epidemiology, etiology, conventional and non-conventional therapies, Int. J. Environ. Res. Public Health
Smet, Smet, Herroelen, Gryspeerdt, Martens, Serum 25(OH)D level on hospital admission associated with COVID-19 stage and mortality, Am. J. Clin. Pathol
Vetel, Leroux, Ducoudray, Practical use. Geriatric autonomy group resources needs, Soins Gerontol
Yang, Zhang, Xu, Effect of Vitamin D on ACE2 and Vitamin D receptor expression in rats with LPS-induced acute lung injury, Chinese J Emerg Med
DOI record:
{
"DOI": "10.1016/j.jsbmb.2021.105958",
"ISSN": [
"0960-0760"
],
"URL": "http://dx.doi.org/10.1016/j.jsbmb.2021.105958",
"alternative-id": [
"S0960076021001515"
],
"article-number": "105958",
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Vitamin D supplementation prior to or during COVID-19 associated with better 3-month survival in geriatric patients: Extension phase of the GERIA-COVID study"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "The Journal of Steroid Biochemistry and Molecular Biology"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.jsbmb.2021.105958"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 Elsevier Ltd. All rights reserved."
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-7199-8109",
"affiliation": [],
"authenticated-orcid": false,
"family": "Annweiler",
"given": "Cédric",
"sequence": "first"
},
{
"affiliation": [],
"family": "Beaudenon",
"given": "Mélinda",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5138-0822",
"affiliation": [],
"authenticated-orcid": false,
"family": "Simon",
"given": "Romain",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Guenet",
"given": "Mialy",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-7791-7376",
"affiliation": [],
"authenticated-orcid": false,
"family": "Otekpo",
"given": "Marie",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Célarier",
"given": "Thomas",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gautier",
"given": "Jennifer",
"sequence": "additional"
}
],
"container-title": "The Journal of Steroid Biochemistry and Molecular Biology",
"container-title-short": "The Journal of Steroid Biochemistry and Molecular Biology",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2021,
7,
29
]
],
"date-time": "2021-07-29T06:50:13Z",
"timestamp": 1627541413000
},
"deposited": {
"date-parts": [
[
2023,
3,
13
]
],
"date-time": "2023-03-13T01:33:53Z",
"timestamp": 1678671233000
},
"indexed": {
"date-parts": [
[
2024,
3,
6
]
],
"date-time": "2024-03-06T15:07:56Z",
"timestamp": 1709737676725
},
"is-referenced-by-count": 31,
"issued": {
"date-parts": [
[
2021,
10
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
1
]
],
"date-time": "2021-10-01T00:00:00Z",
"timestamp": 1633046400000
}
},
{
"URL": "http://www.elsevier.com/open-access/userlicense/1.0/",
"content-version": "am",
"delay-in-days": 307,
"start": {
"date-parts": [
[
2022,
8,
4
]
],
"date-time": "2022-08-04T00:00:00Z",
"timestamp": 1659571200000
}
},
{
"URL": "https://doi.org/10.15223/policy-017",
"content-version": "stm-asf",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
1
]
],
"date-time": "2021-10-01T00:00:00Z",
"timestamp": 1633046400000
}
},
{
"URL": "https://doi.org/10.15223/policy-037",
"content-version": "stm-asf",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
1
]
],
"date-time": "2021-10-01T00:00:00Z",
"timestamp": 1633046400000
}
},
{
"URL": "https://doi.org/10.15223/policy-012",
"content-version": "stm-asf",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
1
]
],
"date-time": "2021-10-01T00:00:00Z",
"timestamp": 1633046400000
}
},
{
"URL": "https://doi.org/10.15223/policy-029",
"content-version": "stm-asf",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
1
]
],
"date-time": "2021-10-01T00:00:00Z",
"timestamp": 1633046400000
}
},
{
"URL": "https://doi.org/10.15223/policy-004",
"content-version": "stm-asf",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
10,
1
]
],
"date-time": "2021-10-01T00:00:00Z",
"timestamp": 1633046400000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0960076021001515?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0960076021001515?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "105958",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
10
]
]
},
"published-print": {
"date-parts": [
[
2021,
10
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.3390/ijerph17218155",
"article-title": "COVID-19 pandemic: epidemiology, etiology, conventional and non-conventional therapies",
"author": "Rauf",
"doi-asserted-by": "crossref",
"first-page": "8155",
"journal-title": "Int. J. Environ. Res. Public Health",
"key": "10.1016/j.jsbmb.2021.105958_bib0005",
"volume": "17",
"year": "2020"
},
{
"DOI": "10.3390/biomedicines8050129",
"author": "Glinsky",
"doi-asserted-by": "crossref",
"first-page": "129",
"journal-title": "Biomedicines",
"key": "10.1016/j.jsbmb.2021.105958_bib0010",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.3390/nu12113377",
"article-title": "Vitamin d supplementation associated to better survival in hospitalized frail elderly COVID-19 patients: the GERIA-COVID quasi-experimental study",
"author": "Annweiler",
"doi-asserted-by": "crossref",
"first-page": "E3377",
"journal-title": "Nutrients",
"key": "10.1016/j.jsbmb.2021.105958_bib0015",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1016/j.eclinm.2020.100683",
"article-title": "Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge",
"author": "Raman",
"doi-asserted-by": "crossref",
"journal-title": "EClinicalMedicine",
"key": "10.1016/j.jsbmb.2021.105958_bib0020",
"volume": "31",
"year": "2021"
},
{
"DOI": "10.1016/j.mayocp.2013.05.011",
"article-title": "Vitamin D for health: a global perspective",
"author": "Hossein-nezhad",
"doi-asserted-by": "crossref",
"first-page": "720",
"journal-title": "Mayo Clin Proc",
"key": "10.1016/j.jsbmb.2021.105958_bib0025",
"volume": "88",
"year": "2013"
},
{
"DOI": "10.1016/j.jsbmb.2020.105771",
"article-title": "Vitamin D and survival in COVID-19 patients: a quasi-experimental study",
"author": "Annweiler",
"doi-asserted-by": "crossref",
"journal-title": "J. Steroid Biochem. Mol. Biol.",
"key": "10.1016/j.jsbmb.2021.105958_bib0030",
"volume": "204",
"year": "2020"
},
{
"article-title": "AGGIR. Practical use. Geriatric autonomy group resources needs",
"author": "Vetel",
"first-page": "23",
"journal-title": "Soins Gerontol.",
"key": "10.1016/j.jsbmb.2021.105958_bib0035",
"year": "1998"
},
{
"DOI": "10.1093/ajcp/aqaa252",
"article-title": "Serum 25(OH)D level on hospital admission associated with COVID-19 stage and mortality",
"author": "De Smet",
"doi-asserted-by": "crossref",
"first-page": "381",
"journal-title": "Am. J. Clin. Pathol.",
"key": "10.1016/j.jsbmb.2021.105958_bib0040",
"volume": "155",
"year": "2021"
},
{
"article-title": "Vitamin D status and outcomes for hospitalised older patients with COVID-19",
"author": "Baktash",
"journal-title": "Postgrad. Med. J.",
"key": "10.1016/j.jsbmb.2021.105958_bib0045",
"year": "2020"
},
{
"article-title": "Vitamin d status in hospitalized patients with SARS-CoV-2 infection",
"author": "Hernández",
"journal-title": "J. Clin. Endocrinol. Metab.",
"key": "10.1016/j.jsbmb.2021.105958_bib0050",
"year": "2020"
},
{
"key": "10.1016/j.jsbmb.2021.105958_bib0055",
"unstructured": "https://www.norcrin.no/aims-of-the-study/cod-liver-oil-for-covid-19-prevention-study/ Accessed 23 December 2020."
},
{
"article-title": "Short term, high-dose vitamin D supplementation for COVID-19 disease: a randomised, placebo-controlled, study (SHADE study)",
"author": "Rastogi",
"journal-title": "Postgrad. Med. J.",
"key": "10.1016/j.jsbmb.2021.105958_bib0060",
"year": "2020"
},
{
"DOI": "10.1016/j.jsbmb.2020.105751",
"article-title": "Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study",
"author": "Entrenas Castillo",
"doi-asserted-by": "crossref",
"journal-title": "J. Steroid Biochem. Mol. Biol.",
"key": "10.1016/j.jsbmb.2021.105958_bib0065",
"volume": "203",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.26848",
"article-title": "Effect of a single high dose of vitamin D3 on hospital length of stay in patients with moderate to severe COVID-19: a randomized clinical trial",
"author": "Murai",
"doi-asserted-by": "crossref",
"first-page": "1053",
"journal-title": "JAMA",
"key": "10.1016/j.jsbmb.2021.105958_bib0070",
"volume": "325",
"year": "2021"
},
{
"DOI": "10.1016/j.jsbmb.2021.105901",
"article-title": "Learning from previous methodological pitfalls to propose well-designed trials on vitamin D in COVID-19",
"author": "Annweiler",
"doi-asserted-by": "crossref",
"journal-title": "J. Steroid Biochem. Mol. Biol.",
"key": "10.1016/j.jsbmb.2021.105958_bib0075",
"volume": "211",
"year": "2021"
},
{
"DOI": "10.3390/nu12040988",
"article-title": "Evidence that vitamin d supplementation could reduce risk of influenza and COVID-19 infections and deaths",
"author": "Grant",
"doi-asserted-by": "crossref",
"issue": "4",
"journal-title": "Nutrients",
"key": "10.1016/j.jsbmb.2021.105958_bib0080",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1016/j.maturitas.2020.06.003",
"article-title": "Point of view: Should COVID-19 patients be supplemented with vitamin D?",
"author": "Annweiler",
"doi-asserted-by": "crossref",
"first-page": "24",
"journal-title": "Maturitas",
"key": "10.1016/j.jsbmb.2021.105958_bib0085",
"volume": "140",
"year": "2020"
},
{
"DOI": "10.1210/me.2013-1146",
"article-title": "VDR attenuates acute lung injury by blocking Ang-2-Tie-2 pathway and renin-angiotensin system",
"author": "Kong",
"doi-asserted-by": "crossref",
"first-page": "2116",
"journal-title": "Mol. Endocrinol.",
"key": "10.1016/j.jsbmb.2021.105958_bib0090",
"volume": "27",
"year": "2013"
},
{
"DOI": "10.1099/vir.0.043919-0",
"article-title": "Replication-dependent downregulation of cellular angiotensin-converting enzyme 2 protein expression by human coronavirus NL63",
"author": "Dijkman",
"doi-asserted-by": "crossref",
"first-page": "1924",
"journal-title": "J. Gen. Virol.",
"key": "10.1016/j.jsbmb.2021.105958_bib0095",
"volume": "93",
"year": "2012"
},
{
"article-title": "Effect of Vitamin D on ACE2 and Vitamin D receptor expression in rats with LPS-induced acute lung injury",
"author": "Yang",
"first-page": "1284",
"journal-title": "Chinese J Emerg Med",
"key": "10.1016/j.jsbmb.2021.105958_bib0100",
"volume": "25",
"year": "2016"
},
{
"article-title": "Vitamin D and immune regulation: anti-bacterial, antiviral, and anti-inflammatory",
"author": "Bishop",
"journal-title": "JBMR Plus",
"key": "10.1016/j.jsbmb.2021.105958_bib0105",
"year": "2020"
},
{
"DOI": "10.1136/bmj.i6583",
"article-title": "Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data",
"author": "Martineau",
"doi-asserted-by": "crossref",
"first-page": "i6583",
"journal-title": "BMJ",
"key": "10.1016/j.jsbmb.2021.105958_bib0110",
"volume": "356",
"year": "2017"
},
{
"DOI": "10.1007/s00198-015-3205-z",
"article-title": "A phase IV, two-armed, randomized, cross-over study comparing compliance with once-a-month administration of vitamin D3 to compliance with daily administration of a fixed-dose combination of vitamin D3 and calcium during two 6-month periods",
"author": "Bruyère",
"doi-asserted-by": "crossref",
"first-page": "2863",
"journal-title": "Osteoporos. Int.",
"key": "10.1016/j.jsbmb.2021.105958_bib0115",
"volume": "26",
"year": "2015"
},
{
"article-title": "Vitamin D in adults: update on testing and supplementation",
"author": "Annweiler",
"first-page": "7",
"journal-title": "Geriatr. Psychol. Neuropsychiatr. Vieil.",
"key": "10.1016/j.jsbmb.2021.105958_bib0120",
"volume": "16",
"year": "2018"
}
],
"reference-count": 24,
"references-count": 24,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S0960076021001515"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Vitamin D supplementation prior to or during COVID-19 associated with better 3-month survival in geriatric patients: Extension phase of the GERIA-COVID study",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "213"
}
