REGEN-COV Antibody Combination and Outcomes in Outpatients with Covid-19
et al., NEJM, doi:10.1056/NEJMoa2108163, NCT04425629, May 2021 (preprint)
18th treatment shown to reduce risk in
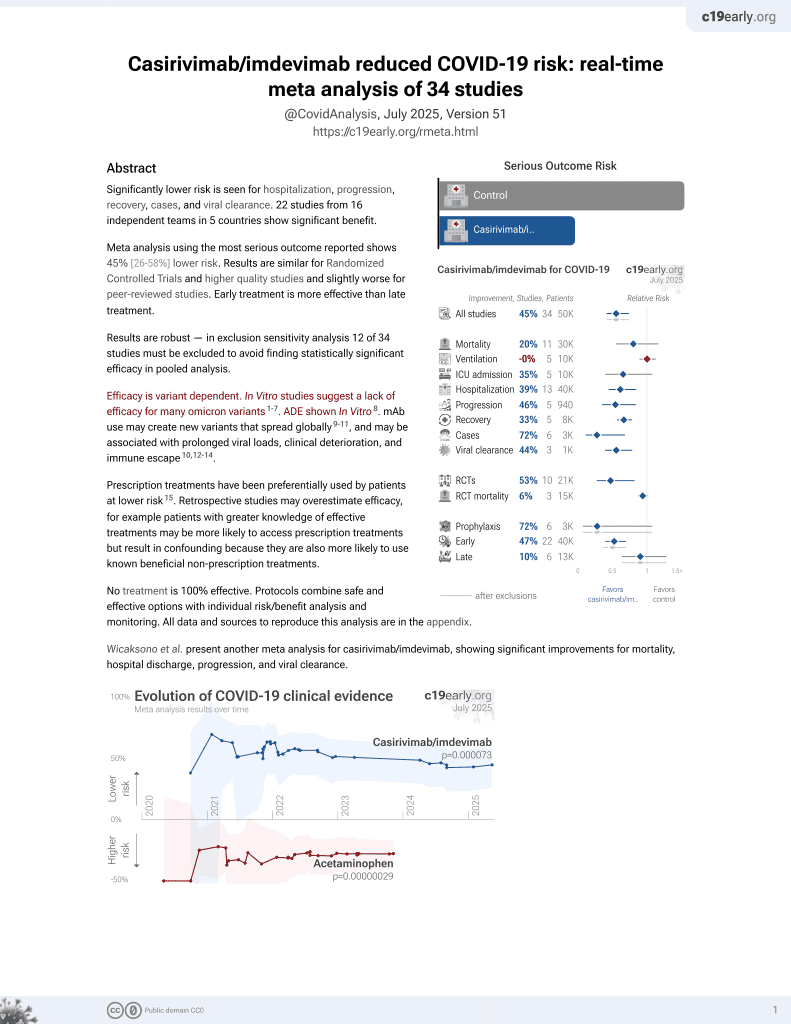
March 2021, now with p = 0.000095 from 34 studies, recognized in 52 countries.
Efficacy is variant dependent.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT 4,057 outpatients with ≥1 risk factor for severe disease, showing significantly lower combined hospitalization/death, and significantly faster recovery with treatment. Median time from onset of symptoms 3 days.
Efficacy is variant dependent. In Vitro research suggests a lack of efficacy for many omicron variants1-7.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments8.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of death, 50.0% lower, RR 0.50, p = 0.45, treatment 2 of 2,091 (0.1%), control 4 of 2,089 (0.2%), NNT 1044, Table S9.
|
|
risk of death, 67.0% lower, RR 0.33, p = 0.37, treatment 1 of 1,355 (0.1%), control 3 of 1,341 (0.2%), NNT 667, 2400mg,Table S9.
|
|
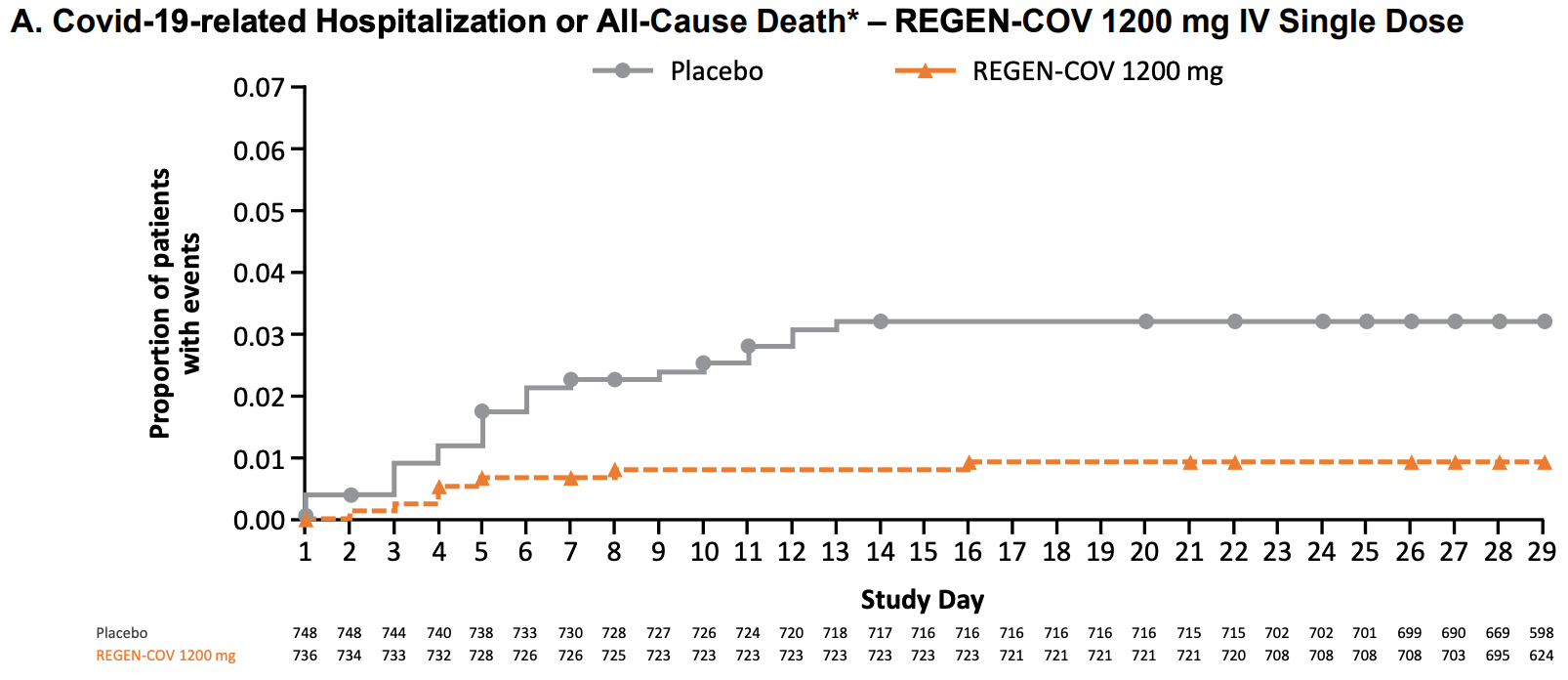
risk of death, 1.6% higher, RR 1.02, p = 1.00, treatment 1 of 736 (0.1%), control 1 of 748 (0.1%), 1200mg,Table S9.
|
|
risk of death/hospitalization, 71.3% lower, RR 0.29, p < 0.001, treatment 18 of 1,355 (1.3%), control 62 of 1,341 (4.6%), NNT 30, 2400mg.
|
|
risk of death/hospitalization, 70.4% lower, RR 0.30, p = 0.002, treatment 7 of 736 (1.0%), control 24 of 748 (3.2%), NNT 44, 1200mg.
|
|
recovery time, 28.6% lower, relative time 0.71, p < 0.001, treatment 1,355, control 1,341, 2400mg.
|
|
recovery time, 28.6% lower, relative time 0.71, p < 0.001, treatment 736, control 748, 1200mg.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
1.
Liu et al., Striking Antibody Evasion Manifested by the Omicron Variant of SARS-CoV-2, bioRxiv, doi:10.1101/2021.12.14.472719.
2.
Sheward et al., Variable loss of antibody potency against SARS-CoV-2 B.1.1.529 (Omicron), bioRxiv, doi:10.1101/2021.12.19.473354.
3.
VanBlargan et al., An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by several therapeutic monoclonal antibodies, bioRxiv, doi:10.1101/2021.12.15.472828.
4.
Tatham et al., Lack of Ronapreve (REGN-CoV; casirivimab and imdevimab) virological efficacy against the SARS-CoV 2 Omicron variant (B.1.1.529) in K18-hACE2 mice, bioRxiv, doi:10.1101/2022.01.23.477397.
5.
Pochtovyi et al., In Vitro Efficacy of Antivirals and Monoclonal Antibodies against SARS-CoV-2 Omicron Lineages XBB.1.9.1, XBB.1.9.3, XBB.1.5, XBB.1.16, XBB.2.4, BQ.1.1.45, CH.1.1, and CL.1, Vaccines, doi:10.3390/vaccines11101533.
6.
Haars et al., Prevalence of SARS-CoV-2 Omicron Sublineages and Spike Protein Mutations Conferring Resistance against Monoclonal Antibodies in a Swedish Cohort during 2022–2023, Microorganisms, doi:10.3390/microorganisms11102417.
Weinreich et al., 21 May 2021, Randomized Controlled Trial, USA, peer-reviewed, 40 authors, study period 24 September, 2020 - 17 January, 2021, average treatment delay 3.0 days, trial NCT04425629 (history).
REGEN-COV Antibody Combination and Outcomes in Outpatients with Covid-19
New England Journal of Medicine, doi:10.1056/nejmoa2108163
BACKGROUND In the phase 1-2 portion of an adaptive trial, REGEN-COV, a combination of the monoclonal antibodies casirivimab and imdevimab, reduced the viral load and number of medical visits in patients with coronavirus disease 2019 (Covid-19). REGEN-COV has activity in vitro against current severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variants of concern.
METHODS In the phase 3 portion of an adaptive trial, we randomly assigned outpatients with Covid-19 and risk factors for severe disease to receive various doses of intravenous REGEN-COV or placebo. Patients were followed through day 29. A prespecified hierarchical analysis was used to assess the end points of hospitalization or death and the time to resolution of symptoms. Safety was also evaluated.
RESULTS Covid-19-related hospitalization or death from any cause occurred in 18 of 1355 patients in the REGEN-COV 2400-mg group (1.3%) and in 62 of 1341 patients in the placebo group who underwent randomization concurrently (4.6%) (relative risk reduction [1 minus the relative risk], 71.3%; P<0.001); these outcomes occurred in 7 of 736 patients in the REGEN-COV 1200-mg group (1.0%) and in 24 of 748 patients in the placebo group who underwent randomization concurrently (3.2%) (relative risk reduction, 70.4%; P = 0.002). The median time to resolution of symptoms was 4 days shorter with each REGEN-COV dose than with placebo (10 days vs. 14 days; P<0.001 for both comparisons). REGEN-COV was efficacious across various subgroups, including patients who were SARS-CoV-2 serum antibody-positive at baseline. Both REGEN-COV doses reduced viral load faster than placebo; the least-squares mean difference in viral load from baseline through day 7 was −0.71 log 10 copies per milliliter (95% confidence interval [CI], −0.90 to −0.53) in the 1200-mg group and −0.86 log 10 copies per milliliter (95% CI, −1.00 to −0.72) in the 2400-mg group. Serious adverse events occurred more frequently in the placebo group (4.0%) than in the 1200-mg group (1.1%) and the 2400-mg group (1.3%); infusion-related reactions of grade 2 or higher occurred in less than 0.3% of the patients in all groups.
CONCLUSIONS REGEN-COV reduced the risk of Covid-19-related hospitalization or death from any cause, and it resolved symptoms and reduced the SARS-CoV-2 viral load more rapidly than placebo. (Funded by Regeneron Pharmaceuticals and others; ClinicalTrials .gov number, NCT04425629.
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org. A data sharing statement provided by the authors is available with the full text of this article at NEJM.org. We thank the trial participants; their families; the investigational site members involved in this trial (principal and subprincipal investigators, listed in the Supplementary Appendix); the Regeneron trial team (members listed in the Supplementary Appendix); the members of the independent data and safety monitoring committee; Brian Head, Ph.D., Caryn Trbovic, Ph.D., and S. Balachandra Dass, Ph.D., of Regeneron Pharmaceuticals for assistance with development of an earlier version of the manuscript; and Prime for assistance with the formatting and copy editing of an earlier version of the manuscript.
Appendix The authors' full names and academic degrees are as follows: David M. Weinreich
References
Baum, Ajithdoss, Copin, REGN-COV2 antibodies prevent and treat SARS-CoV-2 infection in rhesus macaques and hamsters, Science
Baum, Fulton, Wloga, Antibody cocktail to SARS-CoV-2 spike protein prevents rapid mutational escape seen with individual antibodies, Science
Challen, Brooks-Pollock, Read, Dyson, Tsaneva-Atanasova et al., Risk of mortality in patients infected with SARS-CoV-2 variant of concern 202012/1: matched cohort study, BMJ
Copin, Baum, Wloga, The monoclonal antibody combination REGEN-COV protects against SARS-CoV-2 mutational escape in preclinical and human studies, Cell
Davies, Abbott, Barnard, Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England, Science
Del Rio, Collins, Malani, Longterm health consequences of COVID-19, JAMA
Farrington, Manning, Test statistics and sample size formulae for comparative binomial trials with null hypothesis of non-zero risk difference or non-unity relative risk, Stat Med
Hansen, Baum, Pascal, Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail, Science
Logue, Franko, Mcculloch, Sequelae in adults at 6 months after COVID-19 infection, JAMA Netw Open
Nalbandian, Sehgal, Gupta, Post-acute COVID-19 syndrome, Nat Med
Onder, Rezza, Brusaferro, Casefatality rate and characteristics of patients dying in relation to COVID-19 in Italy, JAMA
Rofail, Mcgale, Im, Development and content validation of the Symptoms Evolution of COVID-19: a patient-reported electronic daily diary in clinical and real-world studies, doi:10.1101/2021.07.06.21259654v1
Stokes, Zambrano, Anderson, Coronavirus disease 2019 case surveillance -United States, January 22, MMWR Morb Mortal Wkly Rep
Wang, Nair, Liu, Antibody resistance of SARS-CoV-2 variants B.1.351 and B.1.1.7, Nature
Weinreich, Sivapalasingam, Norton, REGEN-COV antibody cocktail in outpatients with Covid-19, doi:10.1101/2021.06.09.21257915v1
Weinreich, Sivapalasingam, Norton, REGN-COV2, a neutralizing antibody cocktail, in outpatients with Covid-19, N Engl J Med
Williamson, Walker, Bhaskaran, Factors associated with COVID-19-related death using OpenSAFELY, Nature
Wu, Mcgoogan, Characteristics of and important lessons from the Coronavirus Disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention, JAMA
Xie, Liu, Liu, Neutralization of SARS-CoV-2 spike 69/70 deletion, E484K and N501Y variants by BNT162b2 vaccineelicited sera, Nat Med
DOI record:
{
"DOI": "10.1056/nejmoa2108163",
"ISSN": [
"0028-4793",
"1533-4406"
],
"URL": "http://dx.doi.org/10.1056/NEJMoa2108163",
"alternative-id": [
"10.1056/NEJMoa2108163"
],
"author": [
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Weinreich",
"given": "David M.",
"sequence": "first"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Sivapalasingam",
"given": "Sumathi",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Norton",
"given": "Thomas",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Ali",
"given": "Shazia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Gao",
"given": "Haitao",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Bhore",
"given": "Rafia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Xiao",
"given": "Jing",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Hooper",
"given": "Andrea T.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Hamilton",
"given": "Jennifer D.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Musser",
"given": "Bret J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Rofail",
"given": "Diana",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Hussein",
"given": "Mohamed",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Im",
"given": "Joseph",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Atmodjo",
"given": "Dominique Y.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Perry",
"given": "Christina",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Pan",
"given": "Cynthia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Mahmood",
"given": "Adnan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Hosain",
"given": "Romana",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6282-1670",
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"authenticated-orcid": false,
"family": "Davis",
"given": "John D.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Turner",
"given": "Kenneth C.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Baum",
"given": "Alina",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Kyratsous",
"given": "Christos A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Kim",
"given": "Yunji",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Cook",
"given": "Amanda",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Kampman",
"given": "Wendy",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Roque-Guerrero",
"given": "Lilia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Acloque",
"given": "Gerard",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Aazami",
"given": "Hessam",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Cannon",
"given": "Kevin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Simón-Campos",
"given": "J. Abraham",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Bocchini",
"given": "Joseph A.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Kowal",
"given": "Bari",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "DiCioccio",
"given": "A. Thomas",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Soo",
"given": "Yuhwen",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Geba",
"given": "Gregory P.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Stahl",
"given": "Neil",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Lipsich",
"given": "Leah",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Braunstein",
"given": "Ned",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Herman",
"given": "Gary",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "From Regeneron Pharmaceuticals, Tarrytown (D.M.W., T.N., S.A., H.G., R.B., J.X., A.T.H., J.D.H., B.J.M., D.R., M.H., J.I., D.Y.A., C. Perry, C. Pan, A.M., R.H., J.D.D., K.C.T., A.B., C.A.K., Y.K., A.C., W.K., B.K., A.T.D., Y. S., G.P.G., N.S., L.L., N.B., G.H., G.D.Y.) and Excision BioTherapeutics, New York (S.S.) — both in New York; Bio-Medical Research (L.R.-G.) and Universal Medical and Research Center (G.A.) — both in Miami; Hope Clinical Research, Canoga Park, CA (H.A.); PMG Research of Wilmington,..."
}
],
"family": "Yancopoulos",
"given": "George D.",
"sequence": "additional"
}
],
"container-title": "New England Journal of Medicine",
"container-title-short": "N Engl J Med",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
9,
29
]
],
"date-time": "2021-09-29T21:02:54Z",
"timestamp": 1632949374000
},
"deposited": {
"date-parts": [
[
2022,
10,
20
]
],
"date-time": "2022-10-20T18:07:05Z",
"timestamp": 1666289225000
},
"funder": [
{
"DOI": "10.13039/100009857",
"award": [
"Not applicable"
],
"doi-asserted-by": "publisher",
"name": "Regeneron Pharmaceuticals"
},
{
"DOI": "10.13039/100012399",
"award": [
"Not applicable"
],
"doi-asserted-by": "publisher",
"name": "Biomedical Advanced Research and Development Authority of the Department of Health and Human Services"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
9
]
],
"date-time": "2024-04-09T11:57:56Z",
"timestamp": 1712663876898
},
"is-referenced-by-count": 445,
"issue": "23",
"issued": {
"date-parts": [
[
2021,
12,
2
]
]
},
"journal-issue": {
"issue": "23",
"published-print": {
"date-parts": [
[
2021,
12,
2
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://www.nejmgroup.org/legal/terms-of-use.htm",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
12,
2
]
],
"date-time": "2021-12-02T00:00:00Z",
"timestamp": 1638403200000
}
}
],
"link": [
{
"URL": "http://www.nejm.org/doi/pdf/10.1056/NEJMoa2108163",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "150",
"original-title": [],
"page": "e81",
"prefix": "10.1056",
"published": {
"date-parts": [
[
2021,
12,
2
]
]
},
"published-print": {
"date-parts": [
[
2021,
12,
2
]
]
},
"publisher": "Massachusetts Medical Society",
"reference": [
{
"author": "Onder G",
"first-page": "1775",
"journal-title": "JAMA",
"key": "r3",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.15585/mmwr.mm6924e2",
"doi-asserted-by": "publisher",
"key": "r4"
},
{
"DOI": "10.1038/s41586-020-2521-4",
"doi-asserted-by": "publisher",
"key": "r5"
},
{
"DOI": "10.1001/jama.2020.2648",
"doi-asserted-by": "publisher",
"key": "r6"
},
{
"DOI": "10.1136/bmj.n579",
"doi-asserted-by": "publisher",
"key": "r7"
},
{
"DOI": "10.1126/science.abg3055",
"doi-asserted-by": "publisher",
"key": "r8"
},
{
"DOI": "10.1038/s41586-021-03398-2",
"doi-asserted-by": "publisher",
"key": "r9"
},
{
"DOI": "10.1038/s41591-021-01270-4",
"doi-asserted-by": "publisher",
"key": "r10"
},
{
"DOI": "10.1126/science.abd0827",
"doi-asserted-by": "publisher",
"key": "r11"
},
{
"DOI": "10.1126/science.abd0831",
"doi-asserted-by": "publisher",
"key": "r12"
},
{
"DOI": "10.1126/science.abe2402",
"doi-asserted-by": "publisher",
"key": "r13"
},
{
"DOI": "10.1016/j.cell.2021.06.002",
"doi-asserted-by": "publisher",
"key": "r14"
},
{
"DOI": "10.1056/NEJMoa2035002",
"doi-asserted-by": "publisher",
"key": "r16"
},
{
"DOI": "10.1002/sim.4780091208",
"doi-asserted-by": "publisher",
"key": "r18"
},
{
"DOI": "10.1001/jama.2020.19719",
"doi-asserted-by": "publisher",
"key": "r19"
},
{
"DOI": "10.1001/jamanetworkopen.2021.0830",
"doi-asserted-by": "publisher",
"key": "r20"
},
{
"DOI": "10.1038/s41591-021-01283-z",
"doi-asserted-by": "publisher",
"key": "r21"
}
],
"reference-count": 17,
"references-count": 17,
"relation": {
"has-preprint": [
{
"asserted-by": "object",
"id": "10.1101/2021.05.19.21257469",
"id-type": "doi"
}
]
},
"resource": {
"primary": {
"URL": "http://www.nejm.org/doi/10.1056/NEJMoa2108163"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": "REGEN-COV Antibody Combination and Outcomes in Outpatients with Covid-19",
"type": "journal-article",
"volume": "385"
}