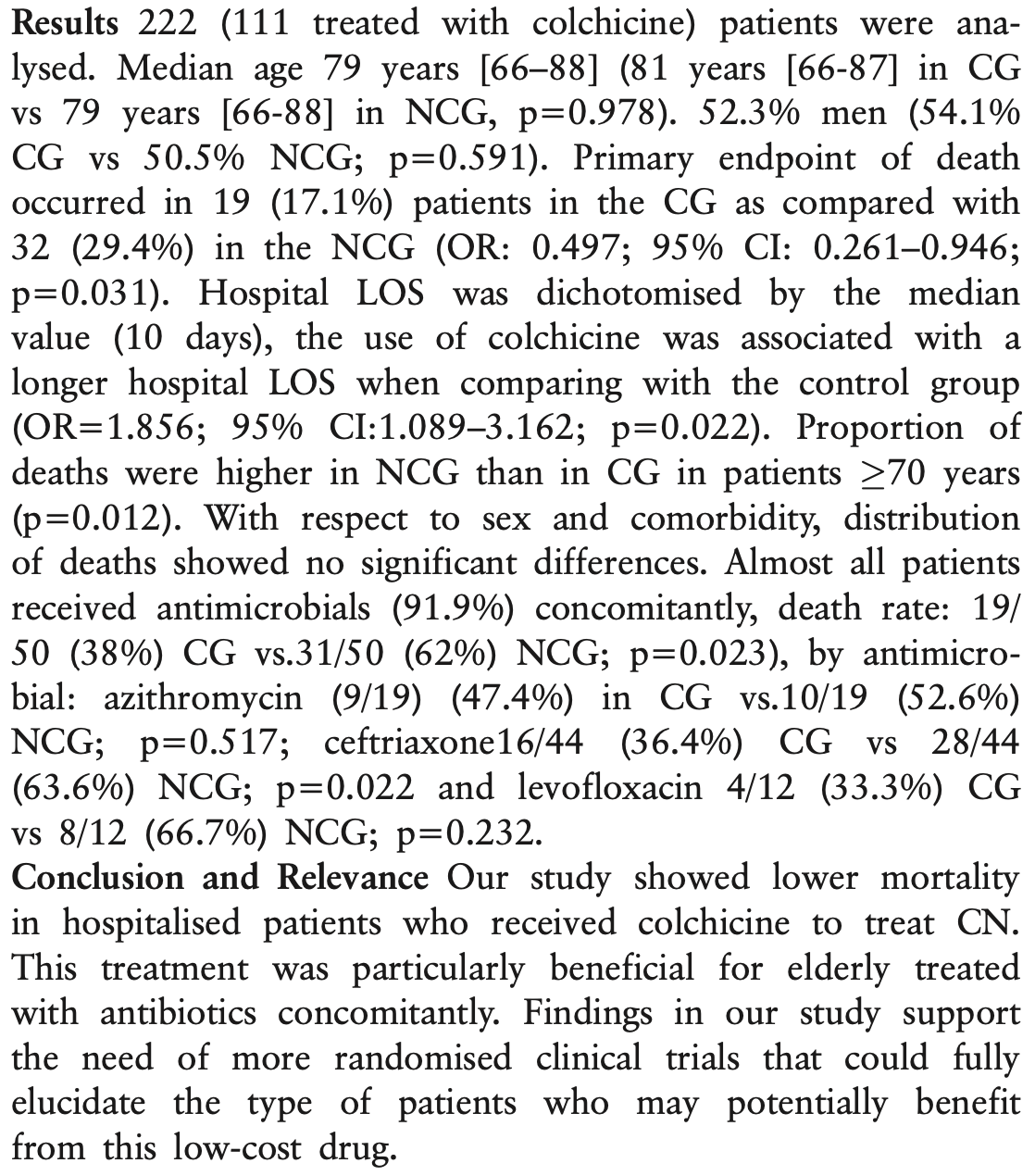
Targeting patients with pneumonia by COVID-19 that could be beneficiated by colchicine
et al., Eur. J. Hosp. Pharm., doi:10.1136/ejhpharm-2023-eahp.56, Mar 2023
Colchicine for COVID-19
5th treatment shown to reduce risk in
September 2020, now with p = 0.0000049 from 54 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Retrospective 111 hospitalized COVID-19 pneumonia patients treated with colchicine and 111 matched controls, showing lower mortality with colchicine treatment.
|
risk of death, 41.9% lower, RR 0.58, p = 0.03, treatment 19 of 111 (17.1%), control 32 of 111 (28.8%), NNT 8.5, odds ratio converted to relative risk.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Villamañán et al., 23 Mar 2023, retrospective, Spain, peer-reviewed, median age 79.0, 10 authors, study period March 2020 - June 2020.
Abstract: 4CPS-007 TARGETING PATIENTS WITH PNEUMONIA BY COVID-
4CPS-008 DOES COMORBIDITY AFFECT ADHERENCE TO
19 THAT COULD BE BENEFICIATED BY COLCHICINE
1
1
1
1
1
1
INHALERS IN SEVERE ASTHMA PATIENTS TREATED
WITH BIOLOGICS?
2
E Villamañán*, C Sobrino, C Mateos, V Collada, A Hoyo, S Mallón, J Pavón,
I Jiménez, 3Y Larrubia, 1A Herrero. 1Hospital Universitario la Paz, Pharmacy, Madrid, Spain;
2
Hospital Universitario la Paz, Pneumology, Madrid, Spain; 3Hospital Infanta Sofía,
Pharmacy, Madrid, Spain
1
1
10.1136/ejhpharm-2023-eahp.56
10.1136/ejhpharm-2023-eahp.57
Background and Importance Available data reported different
results about the effect of colchicine in patients with COVID19 pneumonia (CN) proving the need for more analysis. Currently, many of these patients are treated with high-cost new
drugs with poor results.
Aim and Objectives To evaluate whether treatment with colchicine added to the standard therapy for CN was related to
deaths reduction. Secondary objectives: to analyse differences
in length of stay (LOS) and combination of drugs in treatment
protocols with better results.
Material and Methods Multicentre, real-world, controlled, retrospective cohort study(March-June2020). Inclusion criteria:
hospitalised adult patients with CN. Admitted to critical care
units were excluded. Experimental group: Patients treated with
colchicine who met inclusion criteria (colchicine therapy group
[CG]). Control group: those who met inclusion criteria and
did not received colchicine (non-colchicine therapy group
[NCG]). Patients were matched 1:1 by age ( ± 2years), sex,
severity of the disease and comorbidity. To select controls, we
chose the consecutively next admitted patient after one treated
with colchicine. This allowed us to select control subjects at a
close time and place to cases, that is, under similar circumstances in terms of patient care protocols.
Results 222 (111 treated with colchicine) patients were analysed. Median age 79 years [66–88] (81 years [66-87] in CG
vs 79 years [66-88] in NCG, p=0.978). 52.3% men (54.1%
CG vs 50.5% NCG; p=0.591). Primary endpoint of death
occurred in 19 (17.1%) patients in the CG as compared with
32 (29.4%) in the NCG (OR: 0.497; 95% CI: 0.261–0.946;
p=0.031). Hospital LOS was dichotomised by the median
value (10 days), the use of colchicine was associated with a
longer hospital LOS when comparing with the control group
(OR=1.856; 95% CI:1.089–3.162; p=0.022). Proportion of
deaths were higher in NCG than in CG in patients 70 years
(p=0.012). With respect to sex and comorbidity, distribution
of deaths showed no significant differences. Almost all patients
received antimicrobials (91.9%) concomitantly, death rate: 19/
50 (38%) CG vs.31/50 (62%) NCG; p=0.023), by antimicrobial: azithromycin (9/19) (47.4%) in CG vs.10/19 (52.6%)
NCG; p=0.517; ceftriaxone16/44 (36.4%) CG vs 28/44
(63.6%) NCG; p=0.022 and levofloxacin 4/12 (33.3%) CG
vs 8/12 (66.7%) NCG; p=0.232.
Conclusion and Relevance Our study showed lower mortality
in hospitalised patients who received colchicine to treat CN.
This treatment was particularly beneficial for elderly treated
with antibiotics concomitantly. Findings in our study support
the need of more randomised clinical trials that could fully
elucidate the type of patients who may potentially benefit
from this low-cost drug.
Background and Importance Comorbidities are often associated with severe asthma including those patients treated with
biologics. That often contributes to..
DOI record:
{
"DOI": "10.1136/ejhpharm-2023-eahp.56",
"URL": "http://dx.doi.org/10.1136/ejhpharm-2023-eahp.56",
"alternative-id": [
"10.1136/ejhpharm-2023-eahp.56"
],
"author": [
{
"affiliation": [],
"family": "Villamañán",
"given": "E",
"sequence": "first"
},
{
"affiliation": [],
"family": "Sobrino",
"given": "C",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mateos",
"given": "C",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Collada",
"given": "V",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hoyo",
"given": "A",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mallón",
"given": "S",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pavón",
"given": "J",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jiménez",
"given": "I",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Larrubia",
"given": "Y",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Herrero",
"given": "A",
"sequence": "additional"
}
],
"container-title": "Section 4: Clinical pharmacy services",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"bmj.com"
]
},
"created": {
"date-parts": [
[
2023,
3,
23
]
],
"date-time": "2023-03-23T09:35:38Z",
"timestamp": 1679564138000
},
"deposited": {
"date-parts": [
[
2023,
3,
23
]
],
"date-time": "2023-03-23T09:40:21Z",
"timestamp": 1679564421000
},
"event": "27th EAHP Congress, Lisbon, Portugal, 22-23-24 March 2023",
"indexed": {
"date-parts": [
[
2023,
3,
24
]
],
"date-time": "2023-03-24T04:50:42Z",
"timestamp": 1679633442670
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2023,
3
]
]
},
"link": [
{
"URL": "https://syndication.highwire.org/content/doi/10.1136/ejhpharm-2023-eahp.56",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "239",
"original-title": [],
"prefix": "10.1136",
"published": {
"date-parts": [
[
2023,
3
]
]
},
"published-online": {
"date-parts": [
[
2023,
3,
23
]
]
},
"published-print": {
"date-parts": [
[
2023,
3
]
]
},
"publisher": "British Medical Journal Publishing Group",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://ejhp.bmj.com/lookup/doi/10.1136/ejhpharm-2023-eahp.56"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "4CPS-007 Targeting patients with pneumonia by COVID-19 that could be beneficiated by colchicine",
"type": "proceedings-article",
"update-policy": "http://dx.doi.org/10.1136/crossmarkpolicy"
}
