Clinical effects of ursodeoxycholic acid in COVID-19 infection: a systematic review and dose–response meta-analysis
et al., Frontiers in Pharmacology, doi:10.3389/fphar.2026.1719144, PROSPERO CRD420251019195, Mar 2026
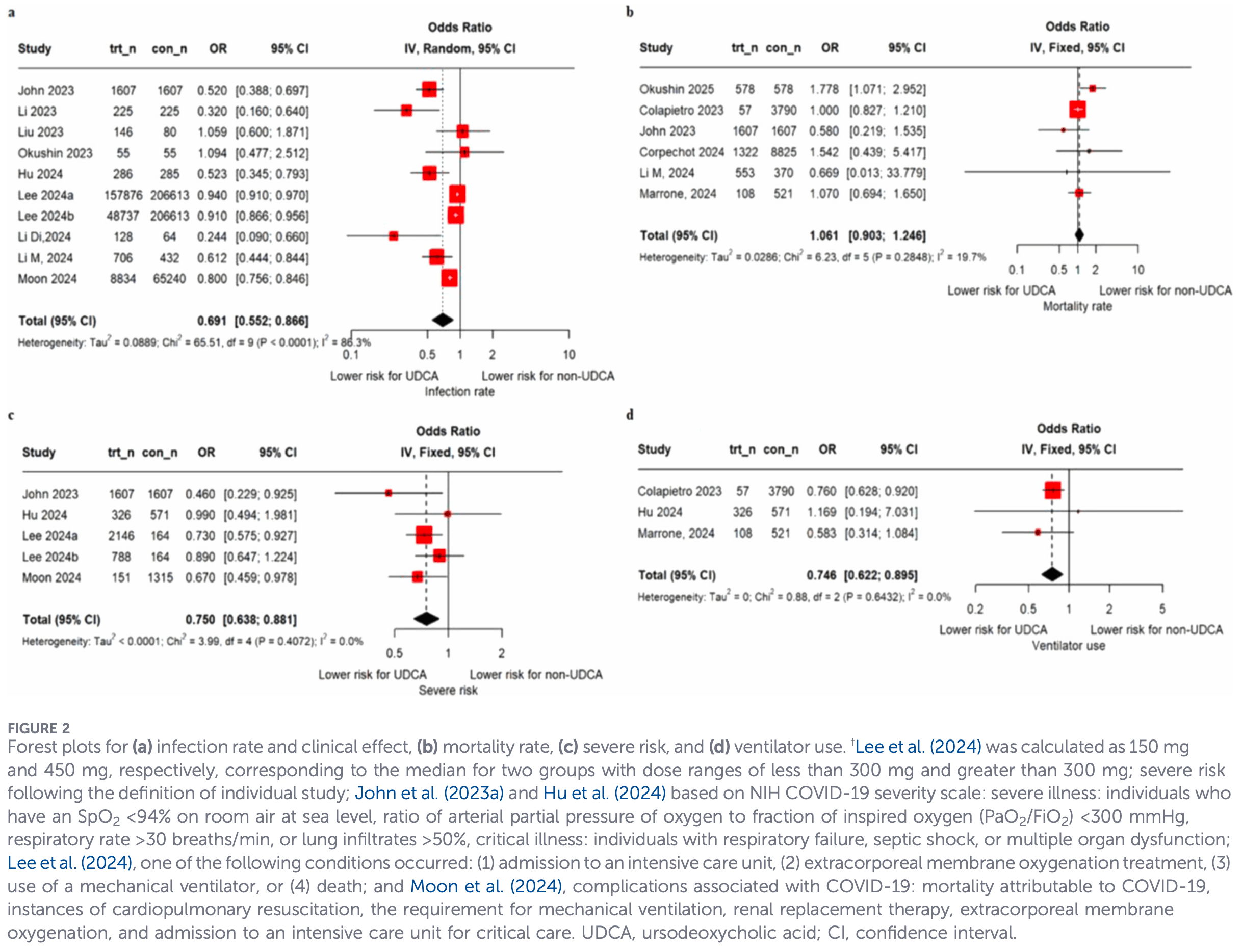
Systematic review and meta-analysis of 15 studies with 716,310 participants showing lower COVID-19 infection risk, severe infection risk, and ventilator use with ursodeoxycholic acid (UDCA) treatment. A dose-response relationship was observed, with increasing UDCA doses associated with progressively lower infection risk. No significant differences were found for mortality, hospitalization, ICU admission, or recovery time.
2 meta-analyses show significant improvements with ursodeoxycholic acid for mechanical ventilation1,
severity1,2, and
cases1.
Currently there are 22 ursodeoxycholic acid for COVID-19 studies, showing 7% lower mortality [-24‑30%], 6% lower ventilation [-293‑78%], 4% lower ICU admission [-66‑44%], 7% lower hospitalization [-9‑20%], and 17% fewer cases [11‑23%].
Song et al., 31 Mar 2026, China, peer-reviewed, 9 authors, trial PROSPERO CRD420251019195.
Contact: sungryul.shim@gmail.com, kchuh2020@hanmail.net.
Abstract:
OPEN ACCESS
EDITED BY Ranjan K. Mohapatra, Government College of Engineering, Keonjhar, India
REVIEWED BY
Bijaya Kumar Padhi, Post Graduate Institute of Medical Education and Research (PGIMER), India Kokouvi Kassegne,
Shanghai Jiao Tong University, China sungryul.shim@gmail.com
*CORRESPONDENCE Sung Ryul Shim, Kyu Chan Huh, kchuh2020@hanmail.net
RECEIVED 03 November 2025 REVISED 05 February 2026 ACCEPTED 20 February 2026 PUBLISHED 01 April 2026
CITATION
Song JH, Shim SR, Shin J, Choe WH, Park J, Lee TH, Kang S, Rhee TG and Huh KC (2026) Clinical effects of ursodeoxycholic acid in COVID-19 infection: a systematic review and dose-response meta-analysis. Front. Pharmacol. 17:1719144. doi: 10.3389/fphar.2026.1719144
COPYRIGHT
© 2026 Song, Shim, Shin, Choe, Park, Lee, Kang, Rhee and Huh. This is an openaccess article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
[Clinical effects of ursodeoxycholic acid in COVID-19 infection: a systematic review and dose-response meta-analysis](https://www.frontiersin.org/articles/10.3389/fphar.2026.1719144/full)
Joo Hye Song 1 , Sung Ryul Shim 2,3 *, Jieun Shin 2,3 , Won Hyeok Choe 1 , Jiho Park 4 , Tae Hee Lee 5 , Seonghui Kang 6 , Taeho Greg Rhee 7,8 and Kyu Chan Huh 5 *
1 Division of Gastroenterology, Department of Internal Medicine, Konkuk University Medical Center, Konkuk University School of Medicine, Seoul, Republic of Korea, 2 Department of Biomedical Informatics, College of Medicine, Konyang University, Daejeon, Republic of Korea, 3 Evidence Based Research Center, Konyang University Hospital, Daejeon, Republic of Korea, 4 Division of Infectious Diseases, Department of Internal Medicine, Konkuk University of Medical Center, Konkuk University School of Medicine, Seoul, Republic of Korea, 5 Division of Gastroenterology, Department of Internal Medicine, Konyang University College of Medicine, Daejeon, Republic of Korea, 6 Division of Infectious Diseases, Department of Internal Medicine, Konyang University College of Medicine, Daejeon, Republic of Korea, 7 Department of Psychiatry, Yale University School of Medicine, New Haven, CT, United States, 8 Department of Public Health Sciences, University of Connecticut School of Medicine, Farmington, CT, United States
Objectives: Previous studies have shown that ursodeoxycholic acid (UDCA) reduces COVID-19 infection by inhibiting farnesoid X receptor activity, a direct regulator of ACE2. Even though UDCA, an easily accessible medication with few side effects, could be considered for administration to prevent infection and relieve symptoms for COVID-19 infection, there are limited supporting studies with a high-level of evidence and recommendations for the exact dosage of UDCA. We conducted a systematic review and dose-response meta-analysis to evaluate the clinical effect of UDCA in COVID-19 infection.
Methods: Studies were identified through a literature search: PubMed, Embase, and Cochrane from inception to March 2025. We included research related to COVID-19 infection and UDCA. Primary outcomes were COVID-19 infection rate, mortality rate, COVID-19..
DOI record:
{
"DOI": "10.3389/fphar.2026.1719144",
"ISSN": [
"1663-9812"
],
"URL": "http://dx.doi.org/10.3389/fphar.2026.1719144",
"abstract": "<jats:sec>\n <jats:title>Objectives</jats:title>\n <jats:p>Previous studies have shown that ursodeoxycholic acid (UDCA) reduces COVID-19 infection by inhibiting farnesoid X receptor activity, a direct regulator of ACE2. Even though UDCA, an easily accessible medication with few side effects, could be considered for administration to prevent infection and relieve symptoms for COVID-19 infection, there are limited supporting studies with a high-level of evidence and recommendations for the exact dosage of UDCA. We conducted a systematic review and dose–response meta-analysis to evaluate the clinical effect of UDCA in COVID-19 infection.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Methods</jats:title>\n <jats:p>Studies were identified through a literature search: PubMed, Embase, and Cochrane from inception to March 2025. We included research related to COVID-19 infection and UDCA. Primary outcomes were COVID-19 infection rate, mortality rate, COVID-19 severe infection risk, ventilator use, hospitalization, ICU hospitalization, and recovery time between UDCA group and controls. The secondary outcome was UDCA dose–response association regarding infection risk. We analyzed for odds ratios (ORs), including infection rate, mortality rate, severe infection risk, ventilator use, hospitalization, and intensive care unit hospitalization, and for standardized mean difference (SMD), including recovery time between UDCA groups and controls. Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) was used to evaluate bias risk.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>Of 188 articles, 15 cohort studies with 716,310 participants (control = 495,276; UDCA treatment = 221,034) were included. The level of risk of bias was seven studies at low, four at moderate, and four at serious. UDCA showed association with a lower risk of infection (OR, 0.69; 95% CI, 0.55–0.86), lower severe infection risk (OR, 0.75; 95% CI, 0.64–0.89), and ventilator use (OR, 0.75; 95% CI, 0.62–0.90) compared to controls.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusion</jats:title>\n <jats:p>The findings support evidence for the clinical effects of UDCA for COVID-19 infection. There is a need for randomized trials to evaluate UDCA as a potential prophylactic agent against COVID-19.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Systematic Review Registration</jats:title>\n <jats:p>\n <jats:ext-link>https://www.crd.york.ac.uk/PROSPERO/view/CRD420251019195</jats:ext-link>\n , identifier #CRD420251019195.\n </jats:p>\n </jats:sec>",
"alternative-id": [
"10.3389/fphar.2026.1719144"
],
"article-number": "1719144",
"author": [
{
"affiliation": [
{
"name": "Division of Gastroenterology, Department of Internal Medicine, Konkuk University Medical Center, Konkuk University School of Medicine",
"place": [
"Seoul, Republic of Korea"
]
}
],
"family": "Song",
"given": "Joo Hye",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Department of Biomedical Informatics, College of Medicine, Konyang University",
"place": [
"Daejeon, Republic of Korea"
]
},
{
"name": "Evidence Based Research Center, Konyang University Hospital",
"place": [
"Daejeon, Republic of Korea"
]
}
],
"family": "Shim",
"given": "Sung Ryul",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Biomedical Informatics, College of Medicine, Konyang University",
"place": [
"Daejeon, Republic of Korea"
]
},
{
"name": "Evidence Based Research Center, Konyang University Hospital",
"place": [
"Daejeon, Republic of Korea"
]
}
],
"family": "Shin",
"given": "Jieun",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Gastroenterology, Department of Internal Medicine, Konkuk University Medical Center, Konkuk University School of Medicine",
"place": [
"Seoul, Republic of Korea"
]
}
],
"family": "Choe",
"given": "Won Hyeok",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Infectious Diseases, Department of Internal Medicine, Konkuk University of Medical Center, Konkuk University School of Medicine",
"place": [
"Seoul, Republic of Korea"
]
}
],
"family": "Park",
"given": "Jiho",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Gastroenterology, Department of Internal Medicine, Konyang University College of Medicine",
"place": [
"Daejeon, Republic of Korea"
]
}
],
"family": "Lee",
"given": "Tae Hee",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Infectious Diseases, Department of Internal Medicine, Konyang University College of Medicine",
"place": [
"Daejeon, Republic of Korea"
]
}
],
"family": "Kang",
"given": "Seonghui",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Psychiatry, Yale University School of Medicine",
"place": [
"New Haven, CT, United States"
]
},
{
"name": "Department of Public Health Sciences, University of Connecticut School of Medicine",
"place": [
"Farmington, CT, United States"
]
}
],
"family": "Rhee",
"given": "Taeho Greg",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Division of Gastroenterology, Department of Internal Medicine, Konyang University College of Medicine",
"place": [
"Daejeon, Republic of Korea"
]
}
],
"family": "Huh",
"given": "Kyu Chan",
"sequence": "additional"
}
],
"container-title": "Frontiers in Pharmacology",
"container-title-short": "Front. Pharmacol.",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2026,
4,
1
]
],
"date-time": "2026-04-01T05:39:40Z",
"timestamp": 1775021980000
},
"deposited": {
"date-parts": [
[
2026,
4,
1
]
],
"date-time": "2026-04-01T05:39:42Z",
"timestamp": 1775021982000
},
"funder": [
{
"DOI": "10.13039/501100004470",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100004470",
"id-type": "DOI"
}
],
"name": "Daewoong Pharmaceutical Company"
}
],
"indexed": {
"date-parts": [
[
2026,
4,
1
]
],
"date-time": "2026-04-01T08:29:13Z",
"timestamp": 1775032153185,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
4,
1
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
4,
1
]
],
"date-time": "2026-04-01T00:00:00Z",
"timestamp": 1775001600000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fphar.2026.1719144/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2026,
4,
1
]
]
},
"published-online": {
"date-parts": [
[
2026,
4,
1
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"author": "Alert",
"key": "B1",
"volume-title": "Guidance for industry estimating the maximum safe starting dose in initial clinical trials for therapeutics in adult healthy volunteers",
"year": "2005"
},
{
"DOI": "10.1136/bmj.n949",
"article-title": "Prophylaxis against covid-19: living systematic review and network meta-analysis",
"author": "Bartoszko",
"doi-asserted-by": "publisher",
"first-page": "373",
"journal-title": "BMJ",
"key": "B2",
"year": "2021"
},
{
"DOI": "10.2307/2533446",
"article-title": "Operating characteristics of a rank correlation test for publication bias",
"author": "Begg",
"doi-asserted-by": "crossref",
"first-page": "1088",
"journal-title": "Biometrics",
"key": "B3",
"volume": "50",
"year": "1994"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the treatment of Covid-19 - Final report",
"author": "Beigel",
"doi-asserted-by": "publisher",
"first-page": "1813",
"journal-title": "N. Engl. J. Med.",
"key": "B4",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1016/j.atherosclerosis.2008.08.001",
"article-title": "Efficacy and safety of ursodeoxycholic acid in primary, type IIa or IIb hypercholesterolemia: a multicenter, randomized, double-blind clinical trial",
"author": "Braga",
"doi-asserted-by": "publisher",
"first-page": "479",
"journal-title": "Atherosclerosis",
"key": "B5",
"volume": "203",
"year": "2009"
},
{
"DOI": "10.1038/s41586-022-05594-0",
"article-title": "FXR inhibition may protect from SARS-CoV-2 infection by reducing ACE2",
"author": "Brevini",
"doi-asserted-by": "publisher",
"first-page": "134",
"journal-title": "Nature",
"key": "B6",
"volume": "615",
"year": "2023"
},
{
"DOI": "10.1038/s41586-021-04385-3",
"article-title": "Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies",
"author": "Cao",
"doi-asserted-by": "publisher",
"first-page": "657",
"journal-title": "Nature",
"key": "B7",
"volume": "602",
"year": "2022"
},
{
"DOI": "10.3390/v15081738",
"article-title": "Ursodeoxycholic acid does not improve COVID-19 outcome in hospitalized patients",
"author": "Colapietro",
"doi-asserted-by": "publisher",
"first-page": "1738",
"journal-title": "Viruses",
"key": "B8",
"volume": "15",
"year": "2023"
},
{
"DOI": "10.2903/j.efsa.2012.2579",
"article-title": "Guidance on selected default values to be used by the EFSA scientific committee, scientific panels and units in the absence of actual measured data",
"author": "Committee",
"doi-asserted-by": "publisher",
"first-page": "2579",
"journal-title": "EFSA J.",
"key": "B9",
"volume": "10",
"year": "2012"
},
{
"DOI": "10.1038/s41564-020-0695-z",
"article-title": "The species severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2",
"doi-asserted-by": "publisher",
"first-page": "536",
"journal-title": "Nat. Microbiol.",
"key": "B49",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.1002/jmv.29418",
"article-title": "Exploring the impact of ursodeoxycholic acid therapy on COVID-19 in a real-word setting",
"author": "Corpechot",
"doi-asserted-by": "publisher",
"first-page": "e29418",
"journal-title": "J. Med. Virol.",
"key": "B10",
"volume": "96",
"year": "2024"
},
{
"DOI": "10.1136/bmj.316.7124.61",
"article-title": "Bias in location and selection of studies",
"author": "Egger",
"doi-asserted-by": "publisher",
"first-page": "61",
"journal-title": "Bmj",
"key": "B11",
"volume": "316",
"year": "1998"
},
{
"DOI": "10.1007/s10620-022-07644-z",
"article-title": "Viral vector vaccines are victorious against COVID-19 in patients with cirrhosis",
"author": "Ferreira",
"doi-asserted-by": "publisher",
"first-page": "349",
"journal-title": "Dig. Dis. Sci.",
"key": "B12",
"volume": "68",
"year": "2023"
},
{
"DOI": "10.1038/s41591-021-01310-z",
"article-title": "Actionable druggable genome-wide Mendelian randomization identifies repurposing opportunities for COVID-19",
"author": "Gaziano",
"doi-asserted-by": "publisher",
"first-page": "668",
"journal-title": "Nat. Med.",
"key": "B13",
"volume": "27",
"year": "2021"
},
{
"DOI": "10.1038/s41579-021-00573-0",
"article-title": "SARS-CoV-2 variants, spike mutations and immune escape",
"author": "Harvey",
"doi-asserted-by": "publisher",
"first-page": "409",
"journal-title": "Nat. Rev. Microbiol.",
"key": "B14",
"volume": "19",
"year": "2021"
},
{
"DOI": "10.1016/j.jhep.2017.03.02",
"article-title": "EASL clinical practice guidelines: the diagnosis and management of patients with primary biliary cholangitis",
"author": "Hirschfield",
"doi-asserted-by": "publisher",
"first-page": "145",
"journal-title": "J. Hepatology",
"key": "B15",
"volume": "67",
"year": "2017"
},
{
"DOI": "10.1093/qjmed/hcad254",
"article-title": "Effect of ursodeoxycholic acid on preventing SARS-CoV-2 infection in patients with liver transplantation: a multicenter retrospective cohort study",
"author": "Hu",
"doi-asserted-by": "publisher",
"first-page": "339",
"journal-title": "Qjm",
"key": "B16",
"volume": "117",
"year": "2024"
},
{
"DOI": "10.3389/fmed.2025.1494248",
"article-title": "Ursodeoxycholic acid relieves clinical severity of COVID-19 in patients with chronic liver diseases. Article",
"author": "Hu",
"doi-asserted-by": "publisher",
"journal-title": "Front. Med.",
"key": "B17",
"year": "2025"
},
{
"DOI": "10.1371/journal.pone.0280691",
"article-title": "Efficacy and safety of ursodeoxycholic acid in children with cholestasis: a systematic review and meta-analysis",
"author": "Huang",
"doi-asserted-by": "publisher",
"first-page": "e0280691",
"journal-title": "PLoS One",
"key": "B18",
"volume": "18",
"year": "2023"
},
{
"DOI": "10.1001/jamainternmed.2021.4325",
"article-title": "Association of BNT162b2 mRNA and mRNA-1273 vaccines with COVID-19 infection and hospitalization among patients with cirrhosis",
"author": "John",
"doi-asserted-by": "publisher",
"first-page": "1306",
"journal-title": "JAMA Intern. Med.",
"key": "B19",
"volume": "181",
"year": "2021"
},
{
"DOI": "10.1016/j.cgh.2022.05.038",
"article-title": "Effectiveness of COVID-19 viral vector Ad.26.COV2.S vaccine and comparison with mRNA vaccines in cirrhosis",
"author": "John",
"doi-asserted-by": "publisher",
"first-page": "2405",
"journal-title": "Clin. Gastroenterol. Hepatol.",
"key": "B20",
"volume": "20",
"year": "2022"
},
{
"DOI": "10.1111/joim.13630",
"article-title": "Ursodeoxycholic acid is associated with a reduction in SARS-CoV-2 infection and reduced severity of COVID-19 in patients with cirrhosis",
"author": "John",
"doi-asserted-by": "publisher",
"first-page": "636",
"journal-title": "J. Intern Med.",
"key": "B21",
"volume": "293",
"year": ""
},
{
"DOI": "10.1136/gutjnl-2022-327799",
"article-title": "COVID-19 vaccine effectiveness and community prevalence of alpha, Delta and omicron variants in patients with cirrhosis",
"author": "John",
"doi-asserted-by": "publisher",
"first-page": "1800",
"journal-title": "Gut",
"key": "B22",
"volume": "72",
"year": ""
},
{
"author": "Kawano-Dourado",
"first-page": "81",
"key": "B23",
"volume-title": "Therapeutics and COVID-19: living guideline",
"year": "2021"
},
{
"DOI": "10.1046/j.1440-1746.2001.02376.x",
"article-title": "Use of ursodeoxycholic acid in liver diseases",
"author": "Kumar",
"doi-asserted-by": "publisher",
"first-page": "3",
"journal-title": "J. Gastroenterology Hepatology",
"key": "B24",
"volume": "16",
"year": "2001"
},
{
"DOI": "10.2196/59274",
"article-title": "Association between ursodeoxycholic acid and clinical outcomes in patients with COVID-19 infection: population-based cohort study",
"author": "Lee",
"doi-asserted-by": "publisher",
"first-page": "e59274",
"journal-title": "JMIR Public Health Surveill.",
"key": "B25",
"volume": "10",
"year": "2024"
},
{
"DOI": "10.3389/fcimb.2023.1178590",
"article-title": "Protective effect of ursodeoxycholic acid on COVID-19 in patients with chronic liver disease",
"author": "Li",
"doi-asserted-by": "publisher",
"first-page": "1178590",
"journal-title": "Front. Cell Infect. Microbiol.",
"key": "B26",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1016/j.bsheal.2024.04.004",
"article-title": "Impact of ursodeoxycholic acid therapy in autoimmune liver disease patients with COVID-19 and its clinical prognosis. Article",
"author": "Li",
"doi-asserted-by": "publisher",
"first-page": "165",
"journal-title": "Biosaf. Health",
"key": "B27",
"volume": "6",
"year": "2024"
},
{
"DOI": "10.3389/fphar.2024.1381830",
"article-title": "Evaluating the protective effectiveness and risk factors of ursodeoxycholic acid on COVID-19 among outpatients. Article",
"author": "Li",
"doi-asserted-by": "publisher",
"journal-title": "Front. Pharmacol.",
"key": "B28",
"year": "2024"
},
{
"DOI": "10.1111/liv.15660",
"article-title": "Ursodeoxycholic acid administration did not reduce susceptibility to SARS-CoV-2 infection in children",
"author": "Liu",
"doi-asserted-by": "publisher",
"first-page": "1950",
"journal-title": "Liver Int.",
"key": "B29",
"volume": "43",
"year": "2023"
},
{
"DOI": "10.1016/j.clinre.2020.05.020",
"article-title": "Ursodeoxycholic acid therapy throughout pregnancy in women affected with chronic cholestasis of childhood: no evidence for teratogenicity",
"author": "Lykavieris",
"doi-asserted-by": "publisher",
"first-page": "101472",
"journal-title": "Clin. Res. Hepatol. Gastroenterol.",
"key": "B30",
"volume": "45",
"year": "2021"
},
{
"DOI": "10.1111/liv.15736",
"article-title": "Ursodeoxycholic acid does not affect the clinical outcome of SARS-CoV-2 infection: a retrospective study of propensity score-matched cohorts",
"author": "Marrone",
"doi-asserted-by": "publisher",
"first-page": "83",
"journal-title": "Liver Int.",
"key": "B31",
"volume": "44",
"year": "2024"
},
{
"DOI": "10.1371/journal.ppat.1008762",
"article-title": "COVID-19 pandemic: insights into structure, function, and hACE2 receptor recognition by SARS-CoV-2",
"author": "Mittal",
"doi-asserted-by": "publisher",
"first-page": "e1008762",
"journal-title": "PLoS Pathogens",
"key": "B32",
"volume": "16",
"year": "2020"
},
{
"DOI": "10.1371/journal.pmed.1000097",
"article-title": "Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement",
"author": "Moher",
"doi-asserted-by": "publisher",
"first-page": "e1000097",
"journal-title": "PLoS Med.",
"key": "B33",
"volume": "6",
"year": "2009"
},
{
"DOI": "10.1038/s41409-019-0705-z",
"article-title": "Prophylactic, preemptive, and curative treatment for sinusoidal obstruction syndrome/veno-occlusive disease in adult patients: a position statement from an international expert group",
"author": "Mohty",
"doi-asserted-by": "publisher",
"first-page": "485",
"journal-title": "Bone Marrow Transpl.",
"key": "B34",
"volume": "55",
"year": "2020"
},
{
"DOI": "10.1186/s12985-024-02464-1",
"article-title": "Association between ursodeoxycholic acid use and COVID-19 in individuals with chronic liver disease: a nationwide case-control study in South Korea",
"author": "Moon",
"doi-asserted-by": "publisher",
"first-page": "202",
"journal-title": "Virol. J.",
"key": "B35",
"volume": "21",
"year": "2024"
},
{
"DOI": "10.1111/joim.13704",
"article-title": "Ursodeoxycholic acid for coronavirus disease 2019 prevention",
"author": "Okushin",
"doi-asserted-by": "publisher",
"first-page": "106",
"journal-title": "J. Intern. Med.",
"key": "B36",
"volume": "295",
"year": "2024"
},
{
"DOI": "10.2169/internalmedicine.4856-24",
"article-title": "Use of ursodeoxycholic acid and the risk of severe coronavirus disease 2019 in elderly patients with viral hepatitis",
"author": "Okushin",
"doi-asserted-by": "publisher",
"first-page": "2301",
"journal-title": "Intern. Med. Tokyo, Jpn.",
"key": "B37",
"volume": "64",
"year": "2025"
},
{
"DOI": "10.1186/s12864-022-08358-2",
"article-title": "Pango lineage designation and assignment using SARS-CoV-2 spike gene nucleotide sequences",
"author": "O’Toole",
"doi-asserted-by": "publisher",
"first-page": "121",
"journal-title": "BMC Genomics",
"key": "B38",
"volume": "23",
"year": "2022"
},
{
"DOI": "10.1038/s41598-021-01705-5",
"article-title": "Interaction of spike protein and lipid membrane of SARS-CoV-2 with ursodeoxycholic acid, an in-silico analysis",
"author": "Rodal",
"doi-asserted-by": "publisher",
"first-page": "22288",
"journal-title": "Sci. Rep.",
"key": "B39",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1042/cs20110184",
"article-title": "Ursodeoxycholic acid in cholestasis: linking action mechanisms to therapeutic applications",
"author": "Roma",
"doi-asserted-by": "publisher",
"first-page": "523",
"journal-title": "Clin. Sci. (Lond).",
"key": "B40",
"volume": "121",
"year": "2011"
},
{
"DOI": "10.1111/j.1572-0241.2006.00634.x",
"article-title": "Long-term effects of mid-dose ursodeoxycholic acid in primary biliary cirrhosis: a meta-analysis of randomized controlled trials",
"author": "Shi",
"doi-asserted-by": "publisher",
"first-page": "1529",
"journal-title": "Am. J. Gastroenterol.",
"key": "B41",
"volume": "101",
"year": "2006"
},
{
"DOI": "10.4178/epih.e2019008",
"article-title": "Intervention meta-analysis: application and practice using R software",
"author": "Shim",
"doi-asserted-by": "publisher",
"first-page": "e2019008",
"journal-title": "Epidemiol. Health",
"key": "B42",
"volume": "41",
"year": "2019"
},
{
"DOI": "10.4178/epih.e2019006",
"article-title": "Dose-response meta-analysis: application and practice using the R software",
"author": "Shim",
"doi-asserted-by": "publisher",
"first-page": "e2019006",
"journal-title": "Epidemiol. Health",
"key": "B43",
"volume": "41",
"year": "2019"
},
{
"DOI": "10.1001/jamanetworkopen.2021.25072",
"article-title": "Cancer risk after radioactive iodine treatment for hyperthyroidism: a systematic review and meta-analysis",
"author": "Shim",
"doi-asserted-by": "publisher",
"first-page": "e2125072",
"journal-title": "JAMA Netw. Open",
"key": "B44",
"volume": "4",
"year": "2021"
},
{
"DOI": "10.1099/jgv.0.001664",
"article-title": "A natural variant in ANP32B impairs influenza virus replication in human cells",
"author": "Staller",
"doi-asserted-by": "publisher",
"first-page": "001664",
"journal-title": "J. Gen. Virol.",
"key": "B45",
"volume": "102",
"year": "2021"
},
{
"DOI": "10.1136/bmj.i4919",
"article-title": "ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions",
"author": "Sterne",
"doi-asserted-by": "publisher",
"first-page": "i4919",
"journal-title": "Bmj",
"key": "B46",
"volume": "355",
"year": "2016"
},
{
"DOI": "10.1016/j.cgh.2013.11.031",
"article-title": "Ursodeoxycholic acid and diets higher in fat prevent gallbladder stones during weight loss: a meta-analysis of randomized controlled trials",
"author": "Stokes",
"doi-asserted-by": "publisher",
"first-page": "1090",
"journal-title": "Clin. Gastroenterol. Hepatol.",
"key": "B47",
"volume": "12",
"year": "2014"
},
{
"DOI": "10.1001/jama.283.15.2008",
"article-title": "Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group",
"author": "Stroup",
"doi-asserted-by": "publisher",
"first-page": "2008",
"journal-title": "Jama",
"key": "B48",
"volume": "283",
"year": "2000"
},
{
"DOI": "10.1016/j.immuni.2020.05.002",
"article-title": "Immunology of COVID-19: current state of the science",
"author": "Vabret",
"doi-asserted-by": "publisher",
"first-page": "910",
"journal-title": "Immunity",
"key": "B50",
"volume": "52",
"year": "2020"
},
{
"DOI": "10.1002/jrsm.1164",
"article-title": "Methods to estimate the between-study variance and its uncertainty in meta-analysis",
"author": "Veroniki",
"doi-asserted-by": "publisher",
"first-page": "55",
"journal-title": "Res. Synth. Methods",
"key": "B51",
"volume": "7",
"year": "2016"
},
{
"DOI": "10.1002/14651858.CD000493.pub3",
"article-title": "Pharmacological interventions for treating intrahepatic cholestasis of pregnancy",
"author": "Walker",
"doi-asserted-by": "publisher",
"first-page": "CD000493",
"journal-title": "Cochrane Database Syst. Rev.",
"key": "B52",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1053/j.gastro.2022.09.008",
"article-title": "Minimal risk of drug-induced liver injury with molnupiravir and ritonavir-boosted nirmatrelvir",
"author": "Wong",
"doi-asserted-by": "publisher",
"first-page": "151",
"journal-title": "Gastroenterology",
"key": "B53",
"volume": "164",
"year": "2023"
},
{
"DOI": "10.1126/sciadv.abe5575",
"article-title": "Conformational dynamics of SARS-CoV-2 trimeric spike glycoprotein in complex with receptor ACE2 revealed by cryo-EM",
"author": "Xu",
"doi-asserted-by": "publisher",
"journal-title": "Sci. Adv.",
"key": "B54",
"volume": "7",
"year": "2021"
},
{
"article-title": "Red and processed meat consumption and the risk of lung cancer: a dose-response meta-analysis of 33 published studies",
"author": "Xue",
"first-page": "1542",
"journal-title": "Int. J. Clin. Exp. Med.",
"key": "B55",
"volume": "7",
"year": "2014"
},
{
"DOI": "10.1080/14787210.2024.2376153",
"article-title": "Ursodeoxycholic acid and COVID-19 outcomes: a cohort study and data synthesis of state-of-art evidence",
"author": "Yu",
"doi-asserted-by": "publisher",
"first-page": "1239",
"journal-title": "Expert Rev. Anti Infect. Ther.",
"key": "B56",
"volume": "22",
"year": "2024"
}
],
"reference-count": 56,
"references-count": 56,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fphar.2026.1719144/full"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Clinical effects of ursodeoxycholic acid in COVID-19 infection: a systematic review and dose–response meta-analysis",
"type": "journal-article",
"update-policy": "https://doi.org/10.3389/crossmark-policy",
"volume": "17"
}