Positive impact of oral hydroxychloroquine and povidone-iodine throat spray for COVID-19 prophylaxis: an open-label randomized trial
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2021.04.035, NCT04446104, Apr 2021
PVP-I for COVID-19
14th treatment shown to reduce risk in
February 2021, now with p = 0.0000000037 from 21 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
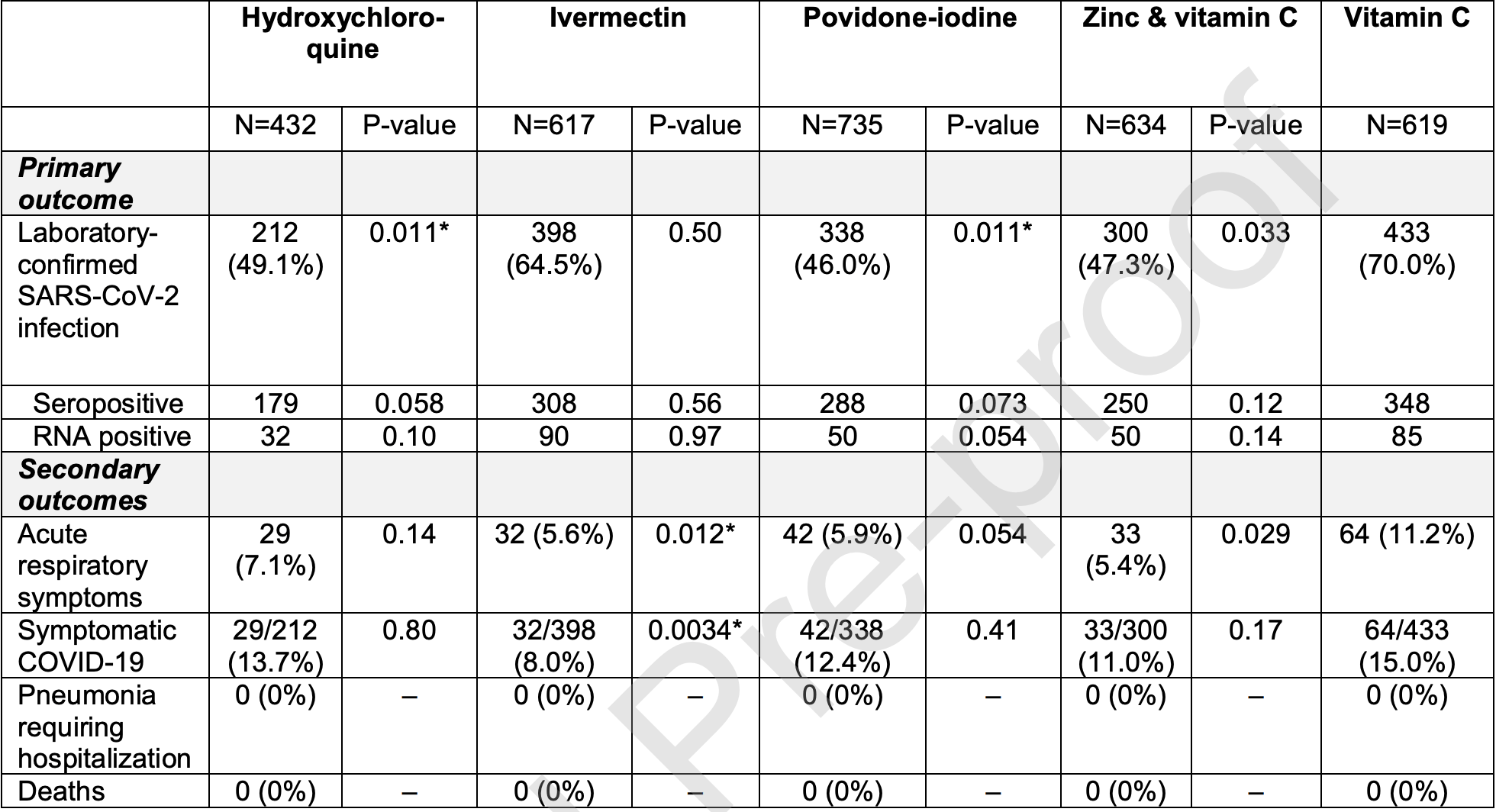
Prophylaxis RCT in Singapore with 3,037 low risk patients, showing lower serious cases, lower symptomatic cases, and lower confirmed cases of COVID-19 with all treatments (ivermectin, HCQ, PVP-I, and Zinc + vitamin C) compared to vitamin C.
Meta-analysis of vitamin C in 6 previous trials shows a benefit of 16%, so the actual benefit of ivermectin, HCQ, and PVP-I may be higher. Cluster RCT with 40 clusters.
There were no hospitalizations and no deaths.
Targeted administration to the respiratory tract provides treatment directly
to the typical source of initial SARS-CoV-2 infection and replication, and
allows for rapid onset of action, higher local drug concentration, and reduced systemic side effects.
|
risk of symptomatic case, 44.7% lower, RR 0.55, p = 0.002, treatment 42 of 735 (5.7%), control 64 of 619 (10.3%), NNT 22.
|
|
risk of case, 31.1% lower, RR 0.69, p = 0.01, treatment 338 of 735 (46.0%), control 433 of 619 (70.0%), NNT 4.2, adjusted per study, odds ratio converted to relative risk, model 6.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Seet et al., 14 Apr 2021, Cluster Randomized Controlled Trial, Singapore, peer-reviewed, 15 authors, study period 13 May, 2020 - 31 August, 2020, this trial compares with another treatment - results may be better when compared to placebo, trial NCT04446104 (history).
COVID-19 chemoprevention
International Journal of Infectious Diseases, doi:10.1016/j.ijid.2021.04.045
Background: We examined whether existing licensed pharmacotherapies could reduce the spread of coronavirus disease 2019 . Methods: An open-label parallel randomized controlled trial was performed among healthy migrant workers quarantined in a large multi-storey dormitory in Singapore. Forty clusters (each defined as individual floors of the dormitory) were randomly assigned to receive a 42-day prophylaxis regimen of either oral hydroxychloroquine (400 mg once, followed by 200 mg/day), oral ivermectin (12 mg once), povidone-iodine throat spray (3 times/day, 270 mg/day), oral zinc (80 mg/day)/vitamin C (500 mg/day) combination, or oral vitamin C, 500 mg/day. The primary outcome was laboratory evidence of SARS-CoV-2 infection as shown by either: (1) a positive serologic test for SARS-CoV-2 antibody on day 42, or (2) a positive PCR test for SARS-CoV-2 at any time between baseline and day 42. Results: A total of 3037 asymptomatic participants (mean age, 33.0 years; all men) who were seronegative to SARS-CoV-2 at baseline were included in the primary analysis. Follow-up was nearly complete (99.6%). Compared with vitamin C, significant absolute risk reductions (%, 98.75% confidence interval) were observed for oral hydroxychloroquine (21%, 2-42%) and povidone-iodine throat spray (24%, 7-39%). No statistically significant differences were observed with oral zinc/vitamin C combination (23%, -5 to +41%) and ivermectin (5%, -10 to +22%). Interruptions due to side effects were highest among participants who received zinc/vitamin C combination (6.9%), followed by vitamin C (4.7%), povidone-iodine (2.0%), and hydroxychloroquine (0.7%). Conclusions: Chemoprophylaxis with either oral hydroxychloroquine or povidone-iodine throat spray was superior to oral vitamin C in reducing SARS-CoV-2 infection in young and healthy men.
Authors contributions All authors have made substantial contributions to the publication. RSCS, QMLA, PAT, and MH contributed to the conception and design of the study and acquisition of data, drafted the article, revised it critically for important intellectual content, and approved the final version for submission. DSQO, SS, SRL, CYK, JBYS, BCG, KSL, DF, and HLT have contributed to the conception and design of the study and acquisition of data, revised it critically for important intellectual content, and approved the final version for submission. JS and ARC have contributed to statistical analysis, revised the manuscript for important intellectual content, and approved the final version for submission.
Conflicts of interest Dr. Seet reported receiving grants from the National Medical Research Council and Temasek Foundation, Singapore. Dr. Tambyah reported receiving grants from Johnson and Johnson, GlaxoSmithKline, and Roche.
Data availability statement The data underlying this article will be shared on reasonable request to the corresponding author. Data suppression rules apply to ensure the anonymity of the study participants.
ClinicalTrials.gov number
NCT04446104 Appendix A. Supplementary data Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.ijid.2021.04.035.
References
Abella, Jolkovsky, Biney, Uspal, Hyman et al., Efficacy and safety of hydroxychloroquine vs Placebo for pre-exposure SARS-CoV-2 prophylaxis among health care workers: a randomized clinical trial, JAMA Intern Med
Anderson, Sivalingam, Kang, Ananthanarayanan, Arumugam et al., Povidone-iodine demonstrates rapid in vitro virucidal activity against SARS-CoV-2, the virus causing COVID-19 disease, Infect Dis Ther
Boulware, Pullen, Bangdiwala, Pastick, Lofgren et al., A randomized trial of hydroxychloroquine as postexposure prophylaxis for Covid-19, N Engl J Med
Caly, Druce, Catton, Jans, Wagstaff, The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro, Antiviral Res, doi:10.1016/j.antiviral.2020
Chen, Yap, Hsu, Teo, COVID-19 and Singapore: from early response to circuit breaker, Ann Acad Med Singap
Core, R: A language and environment for statistical computing
Dong, Zhou, Niu, Wang, Pan et al., Highly accurate and sensitive diagnostic detection of SARS-CoV-2 by digital PCR, Talanta
Feyaerts, Luyten, Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19?, Nutrition, doi:10.1016/j.nut.2020.110948
Frank, Brown, Capriotti, Westover, Pelletier et al., In vitro efficacy of a povidone-iodine nasal antiseptic for rapid inactivation of SARS-CoV-2, JAMA Otolaryngol Head Neck Surg
Goldman, Bomze, Dankner, Hod, Meirson et al., Cardiovascular adverse events associated with hydroxychloroquine and chloroquine: a comprehensive pharmacovigilance analysis of pre-COVID-19 reports, Br J Clin Pharmacol
Hoffmann, Mösbauer, Hofmann-Winkler, Kaul, Kleine-Weber et al., Chloroquine does not inhibit infection of human lung cells with SARS-CoV-2, Nature
Hunter, Arentz, Goldenberg, Yang, Beardsley et al., Rapid review protocol: Zinc for the prevention or treatment of COVID-19 and other coronavirus-related respiratory tract infections, Integr Med Res, doi:10.1016/j.imr.2020.100457
Koh, Migrant workers and COVID-19, Occup Environ Med
Lee, Chiew, Khong, Interrupting transmission of COVID-19: lessons from containment efforts in Singapore, J Travel Med, doi:10.1093/jtm/taaa039
Li, Thoon, Chong, Maiwald, Kam et al., Comparative analysis of symptomatic and asymptomatic SARS-CoV-2 infection in children, Ann Acad Med Singap
Liu, Cao, Xu, Wang, Zhang et al., Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro, Cell Discov, doi:10.1038/s41421-020-0156-0
Maisonnasse, Guedj, Contreras, Behillil, Solas et al., Hydroxychloroquine use against SARS-CoV-2 infection in non-human primates, Nature
Mehra, Desai, Ruschitzka, Patel, RETRACTED: Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis, Lancet, doi:10.1016/S0140-6736(20)31180-6
Mitjà, Corbacho-Monné, Ubals, Alemany, Suñer et al., A clusterrandomized trial of hydroxychloroquine for prevention of Covid-19, N Engl J Med
Nikolai, Meyer, Kremsner, Velavan, Asymptomatic SARS Coronavirus 2 infection: invisible yet invincible, Int J Infect Dis
Rakedzon, Neuberger, Domb, Petersiel, Schwartz, From hydroxychloroquine to ivermectin: what are the anti-viral properties of antiparasitic drugs to combat SARS-CoV-2?, J Travel Med, doi:10.1093/jtm/taab005
Rosenberg, Dufort, Udo, Wilberschied, Kumar et al., Association of treatment with hydroxychloroquine or azithromycin with inhospital mortality in patients with COVID-19 in New York State, JAMA
Stevenson, Kirresh, Conway, White, Ahmad et al., Hydroxychloroquine use in COVID-19: is the risk of cardiovascular toxicity justified?, Open Heart, doi:10.1136/openhrt-2020-001362
Sun, Koh, Marimuthu, Ng, Young et al., Epidemiological and clinical predictors of COVID-19, Clin Infect Dis
Tan, Cook, Logan, Rozanova, Wilder-Smith, Singapore's pandemic preparedness: an overview of the first wave of COVID-19, Int J Environ Res Public Health 2020a, doi:10.3390/ijerph18010252
Tan, Toh, Vasoo, Lye, Ang et al., Coronavirus disease 2019 (COVID-19): the Singapore Experience. A review of the first eight months, Ann Acad Med Singap
Ternovoi, Lutkovsky, Ponomareva, Gladysheva, Chub et al., Detection of SARS-CoV-2 RNA in nasopharyngeal swabs from COVID-19 patients and asymptomatic cases of infection by real-time and digital PCR, Klin Lab Diagn
Trial, Neha Burla, Zhiyong Blok, Jie, Ching Wan et al., were instrumental to the successful recruitment and follow-up of all participants
Velthuis, Van Den Worm, Sims, Baric, Snijder et al., Zn(2 +) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture, PLoS Pathog, doi:10.1371/journal.ppat.1001176
Wiersinga, Rhodes, Cheng, Peacock, Prescott, Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review, JAMA
Yao, Ye, Zhang, Cui, Huang et al., In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), Clin Infect Dis
Yi, Ng, Farwin, Low, Chang et al., Health equity considerations in COVID-19: geospatial network analysis of the COVID-19 outbreak in the migrant population in Singapore, J Travel Med, doi:10.1093/jtm/taaa159
Young, Ong, Ng, Anderson, Chia et al., Viral dynamics and immune correlates of COVID-19 disease severity, Clin Infect Dis, doi:10.1093/cid/ciaa1280
DOI record:
{
"DOI": "10.1016/j.ijid.2021.04.035",
"ISSN": [
"1201-9712"
],
"URL": "http://dx.doi.org/10.1016/j.ijid.2021.04.035",
"alternative-id": [
"S1201971221003453"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Positive impact of oral hydroxychloroquine and povidone-iodine throat spray for COVID-19 prophylaxis: An open-label randomized trial"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "International Journal of Infectious Diseases"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.ijid.2021.04.035"
},
{
"label": "CrossRef DOI link to the associated document",
"name": "associatedlink",
"value": "https://doi.org/10.1016/j.ijid.2021.04.045"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 The Author(s). Published by Elsevier Ltd on behalf of International Society for Infectious Diseases."
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0001-8946-3578",
"affiliation": [],
"authenticated-orcid": false,
"family": "Seet",
"given": "Raymond Chee Seong",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-0569-0472",
"affiliation": [],
"authenticated-orcid": false,
"family": "Quek",
"given": "Amy May Lin",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8677-0126",
"affiliation": [],
"authenticated-orcid": false,
"family": "Ooi",
"given": "Delicia Shu Qin",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8706-8355",
"affiliation": [],
"authenticated-orcid": false,
"family": "Sengupta",
"given": "Sharmila",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1454-3503",
"affiliation": [],
"authenticated-orcid": false,
"family": "Lakshminarasappa",
"given": "Satish Ramapatna",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6051-3807",
"affiliation": [],
"authenticated-orcid": false,
"family": "Koo",
"given": "Chieh Yang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "So",
"given": "Jimmy Bok Yan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Goh",
"given": "Boon Cher",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Loh",
"given": "Kwok Seng",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Fisher",
"given": "Dale",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Teoh",
"given": "Hock Luen",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-5264-4428",
"affiliation": [],
"authenticated-orcid": false,
"family": "Sun",
"given": "Jie",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cook",
"given": "Alex R.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tambyah",
"given": "Paul Anantharajah",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5726-9965",
"affiliation": [],
"authenticated-orcid": false,
"family": "Hartman",
"given": "Mikael",
"sequence": "additional"
}
],
"container-title": "International Journal of Infectious Diseases",
"container-title-short": "International Journal of Infectious Diseases",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.fr",
"clinicalkey.jp",
"clinicalkey.es",
"clinicalkey.com.au",
"clinicalkey.com",
"ijidonline.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2021,
4,
21
]
],
"date-time": "2021-04-21T03:12:44Z",
"timestamp": 1618974764000
},
"deposited": {
"date-parts": [
[
2022,
11,
18
]
],
"date-time": "2022-11-18T09:17:53Z",
"timestamp": 1668763073000
},
"funder": [
{
"DOI": "10.13039/501100001349",
"doi-asserted-by": "publisher",
"name": "National Medical Research Council"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
8
]
],
"date-time": "2024-04-08T20:35:29Z",
"timestamp": 1712608529855
},
"is-referenced-by-count": 66,
"issued": {
"date-parts": [
[
2021,
5
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
5,
1
]
],
"date-time": "2021-05-01T00:00:00Z",
"timestamp": 1619827200000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
4,
10
]
],
"date-time": "2021-04-10T00:00:00Z",
"timestamp": 1618012800000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971221003453?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971221003453?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "314-322",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
5
]
]
},
"published-print": {
"date-parts": [
[
2021,
5
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1001/jamainternmed.2020.6319",
"article-title": "Efficacy and safety of hydroxychloroquine vs Placebo for pre-exposure SARS-CoV-2 prophylaxis among health care workers: a randomized clinical trial",
"author": "Abella",
"doi-asserted-by": "crossref",
"first-page": "195",
"journal-title": "JAMA Intern Med",
"key": "10.1016/j.ijid.2021.04.035_bib0005",
"volume": "181",
"year": "2020"
},
{
"DOI": "10.1007/s40121-020-00316-3",
"article-title": "Povidone-iodine demonstrates rapid in vitro virucidal activity against SARS-CoV-2, the virus causing COVID-19 disease",
"author": "Anderson",
"doi-asserted-by": "crossref",
"first-page": "669",
"journal-title": "Infect Dis Ther",
"key": "10.1016/j.ijid.2021.04.035_bib0010",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2016638",
"article-title": "A randomized trial of hydroxychloroquine as postexposure prophylaxis for Covid-19",
"author": "Boulware",
"doi-asserted-by": "crossref",
"first-page": "517",
"journal-title": "N Engl J Med",
"key": "10.1016/j.ijid.2021.04.035_bib0015",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1016/j.antiviral.2020.104787",
"article-title": "The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro",
"author": "Caly",
"doi-asserted-by": "crossref",
"journal-title": "Antiviral Res",
"key": "10.1016/j.ijid.2021.04.035_bib0020",
"volume": "178",
"year": "2020"
},
{
"DOI": "10.47102/annals-acadmedsg.2020239",
"article-title": "COVID-19 and Singapore: from early response to circuit breaker",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "561",
"journal-title": "Ann Acad Med Singap",
"key": "10.1016/j.ijid.2021.04.035_bib0025",
"volume": "49",
"year": "2020"
},
{
"DOI": "10.1016/j.talanta.2020.121726",
"article-title": "Highly accurate and sensitive diagnostic detection of SARS-CoV-2 by digital PCR",
"author": "Dong",
"doi-asserted-by": "crossref",
"journal-title": "Talanta",
"key": "10.1016/j.ijid.2021.04.035_bib0030",
"volume": "224",
"year": "2021"
},
{
"article-title": "Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19?",
"author": "Feyaerts",
"journal-title": "Nutrition",
"key": "10.1016/j.ijid.2021.04.035_bib0035",
"volume": "79–80",
"year": "2020"
},
{
"DOI": "10.1001/jamaoto.2020.3053",
"article-title": "In vitro efficacy of a povidone-iodine nasal antiseptic for rapid inactivation of SARS-CoV-2",
"author": "Frank",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "JAMA Otolaryngol Head Neck Surg",
"key": "10.1016/j.ijid.2021.04.035_bib0040",
"volume": "146",
"year": "2020"
},
{
"DOI": "10.1111/bcp.14546",
"article-title": "Cardiovascular adverse events associated with hydroxychloroquine and chloroquine: a comprehensive pharmacovigilance analysis of pre-COVID-19 reports",
"author": "Goldman",
"doi-asserted-by": "crossref",
"first-page": "1432",
"journal-title": "Br J Clin Pharmacol",
"key": "10.1016/j.ijid.2021.04.035_bib0045",
"volume": "87",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2575-3",
"article-title": "Chloroquine does not inhibit infection of human lung cells with SARS-CoV-2",
"author": "Hoffmann",
"doi-asserted-by": "crossref",
"first-page": "588",
"journal-title": "Nature",
"key": "10.1016/j.ijid.2021.04.035_bib0050",
"volume": "585",
"year": "2020"
},
{
"DOI": "10.1016/j.imr.2020.100457",
"article-title": "Rapid review protocol: Zinc for the prevention or treatment of COVID-19 and other coronavirus-related respiratory tract infections",
"author": "Hunter",
"doi-asserted-by": "crossref",
"journal-title": "Integr Med Res",
"key": "10.1016/j.ijid.2021.04.035_bib0055",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1136/oemed-2020-106626",
"article-title": "Migrant workers and COVID-19",
"author": "Koh",
"doi-asserted-by": "crossref",
"first-page": "634",
"journal-title": "Occup Environ Med",
"key": "10.1016/j.ijid.2021.04.035_bib0060",
"volume": "77",
"year": "2020"
},
{
"DOI": "10.1093/jtm/taaa039",
"article-title": "Interrupting transmission of COVID-19: lessons from containment efforts in Singapore",
"author": "Lee",
"doi-asserted-by": "crossref",
"journal-title": "J Travel Med",
"key": "10.1016/j.ijid.2021.04.035_bib0065",
"volume": "27",
"year": "2020"
},
{
"DOI": "10.47102/annals-acadmedsg.2020257",
"article-title": "Comparative analysis of symptomatic and asymptomatic SARS-CoV-2 infection in children",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "530",
"journal-title": "Ann Acad Med Singap",
"key": "10.1016/j.ijid.2021.04.035_bib0070",
"year": "2020"
},
{
"DOI": "10.1038/s41421-020-0156-0",
"article-title": "Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "16",
"journal-title": "Cell Discov",
"key": "10.1016/j.ijid.2021.04.035_bib0075",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2558-4",
"article-title": "Hydroxychloroquine use against SARS-CoV-2 infection in non-human primates",
"author": "Maisonnasse",
"doi-asserted-by": "crossref",
"first-page": "584",
"journal-title": "Nature",
"key": "10.1016/j.ijid.2021.04.035_bib0080",
"volume": "585",
"year": "2020"
},
{
"article-title": "RETRACTED: Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis",
"author": "Mehra",
"journal-title": "Lancet",
"key": "10.1016/j.ijid.2021.04.035_bib0085",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2021801",
"article-title": "A cluster-randomized trial of hydroxychloroquine for prevention of Covid-19",
"author": "Mitjà",
"doi-asserted-by": "crossref",
"first-page": "417",
"journal-title": "N Engl J Med",
"key": "10.1016/j.ijid.2021.04.035_bib0090",
"volume": "384",
"year": "2020"
},
{
"DOI": "10.1016/j.ijid.2020.08.076",
"article-title": "Asymptomatic SARS Coronavirus 2 infection: invisible yet invincible",
"author": "Nikolai",
"doi-asserted-by": "crossref",
"first-page": "112",
"journal-title": "Int J Infect Dis",
"key": "10.1016/j.ijid.2021.04.035_bib0095",
"volume": "100",
"year": "2020"
},
{
"author": "R Core Team",
"key": "10.1016/j.ijid.2021.04.035_bib0100",
"series-title": "R: A language and environment for statistical computing",
"year": "2020"
},
{
"DOI": "10.1093/jtm/taab005",
"article-title": "From hydroxychloroquine to ivermectin: what are the anti-viral properties of anti-parasitic drugs to combat SARS-CoV-2?",
"author": "Rakedzon",
"doi-asserted-by": "crossref",
"journal-title": "J Travel Med",
"key": "10.1016/j.ijid.2021.04.035_bib0105",
"year": "2021"
},
{
"DOI": "10.1001/jama.2020.8630",
"article-title": "Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York State",
"author": "Rosenberg",
"doi-asserted-by": "crossref",
"first-page": "2493",
"journal-title": "JAMA",
"key": "10.1016/j.ijid.2021.04.035_bib0110",
"volume": "323",
"year": "2020"
},
{
"key": "10.1016/j.ijid.2021.04.035_bib0115",
"unstructured": "Singapore Ministry of Health 2020. https://www.moh.gov.sg/covid-19 [Accessed 5 March 2021]."
},
{
"DOI": "10.1136/openhrt-2020-001362",
"article-title": "Hydroxychloroquine use in COVID-19: is the risk of cardiovascular toxicity justified?",
"author": "Stevenson",
"doi-asserted-by": "crossref",
"journal-title": "Open Heart",
"key": "10.1016/j.ijid.2021.04.035_bib0120",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1093/cid/ciaa322",
"article-title": "Epidemiological and clinical predictors of COVID-19",
"author": "Sun",
"doi-asserted-by": "crossref",
"first-page": "786",
"journal-title": "Clin Infect Dis",
"key": "10.1016/j.ijid.2021.04.035_bib0125",
"volume": "71",
"year": "2020"
},
{
"DOI": "10.3390/ijerph18010252",
"article-title": "Singapore’s pandemic preparedness: an overview of the first wave of COVID-19",
"author": "Tan",
"doi-asserted-by": "crossref",
"first-page": "252",
"journal-title": "Int J Environ Res Public Health",
"key": "10.1016/j.ijid.2021.04.035_bib0130",
"volume": "18",
"year": "2020"
},
{
"DOI": "10.47102/annals-acadmedsg.2020306",
"article-title": "Coronavirus disease 2019 (COVID-19): the Singapore Experience. A review of the first eight months",
"author": "Tan",
"doi-asserted-by": "crossref",
"first-page": "764",
"journal-title": "Ann Acad Med Singap",
"key": "10.1016/j.ijid.2021.04.035_bib0135",
"volume": "49",
"year": "2020"
},
{
"DOI": "10.1371/journal.ppat.1001176",
"article-title": "Zn(2+) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture",
"author": "te Velthuis",
"doi-asserted-by": "crossref",
"journal-title": "PLoS Pathog",
"key": "10.1016/j.ijid.2021.04.035_bib0140",
"volume": "6",
"year": "2010"
},
{
"DOI": "10.18821/0869-2084-2020-65-12-785-792",
"article-title": "Detection of SARS-CoV-2 RNA in nasopharyngeal swabs from COVID-19 patients and asymptomatic cases of infection by real-time and digital PCR",
"author": "Ternovoi",
"doi-asserted-by": "crossref",
"first-page": "785",
"journal-title": "Klin Lab Diagn",
"key": "10.1016/j.ijid.2021.04.035_bib0145",
"volume": "65",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.12839",
"article-title": "Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review",
"author": "Wiersinga",
"doi-asserted-by": "crossref",
"first-page": "782",
"journal-title": "JAMA",
"key": "10.1016/j.ijid.2021.04.035_bib0150",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1093/cid/ciaa237",
"article-title": "In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)",
"author": "Yao",
"doi-asserted-by": "crossref",
"first-page": "732",
"journal-title": "Clin Infect Dis",
"key": "10.1016/j.ijid.2021.04.035_bib0155",
"volume": "71",
"year": "2020"
},
{
"article-title": "Health equity considerations in COVID-19: geospatial network analysis of the COVID-19 outbreak in the migrant population in Singapore",
"author": "Yi",
"journal-title": "J Travel Med",
"key": "10.1016/j.ijid.2021.04.035_bib0160",
"year": "2020"
},
{
"article-title": "Viral dynamics and immune correlates of COVID-19 disease severity",
"author": "Young",
"journal-title": "Clin Infect Dis",
"key": "10.1016/j.ijid.2021.04.035_bib0165",
"year": "2020"
}
],
"reference-count": 33,
"references-count": 33,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S1201971221003453"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)",
"General Medicine"
],
"subtitle": [],
"title": "Positive impact of oral hydroxychloroquine and povidone-iodine throat spray for COVID-19 prophylaxis: An open-label randomized trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "106"
}
seet
