Risk Factors Associated With In-Hospital Mortality in a US National Sample of Patients With COVID-19
et al., JAMA Network Open, doi:10.1001/jamanetworkopen.2020.29058, Dec 2020
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
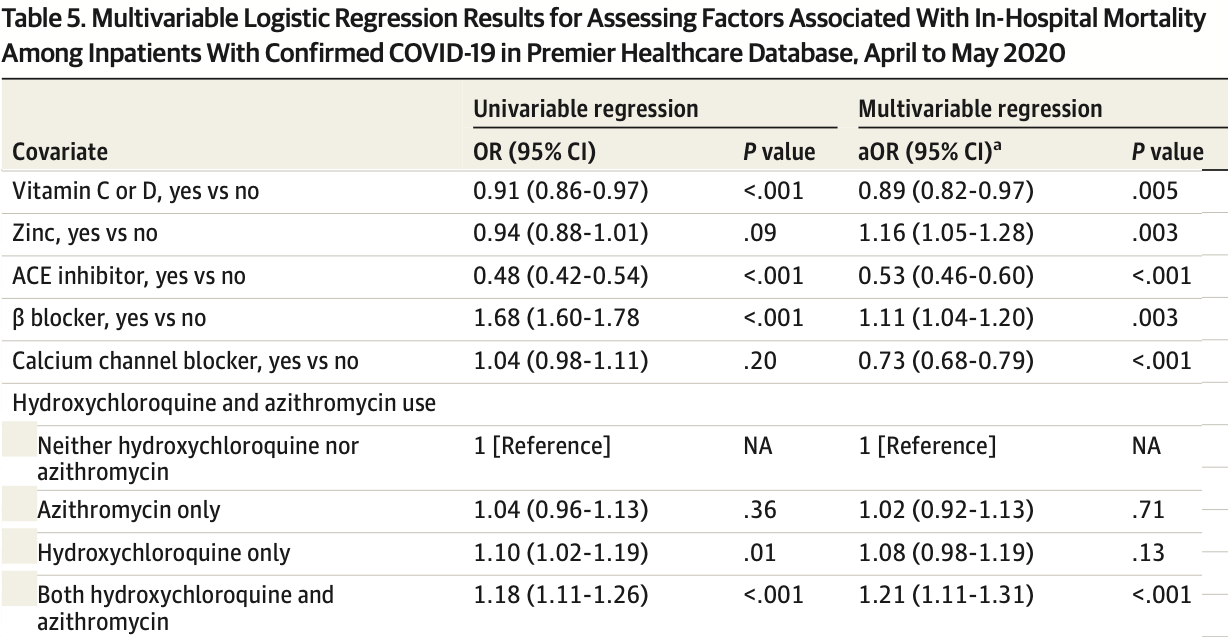
Retrospective database analysis of 64,781 hospitalized patients in the USA, showing lower mortality with vitamin C or vitamin D (authors do not distinguish between the two), and higher mortality with zinc and HCQ, statistically significant for zinc. Authors excluded hospital-based outpatient visits, without explanation. Confounding by indication is likely, adjustments do not appear to include any information on COVID-19 severity at baseline.
Cholecalciferol was used in this study.
Meta-analysis shows that late stage treatment with calcitriol / calcifediol (or
paricalcitol, alfacalcidol, etc.) is more effective than cholecalciferol: 66% [47‑78%] lower risk vs. 43% [32‑52%] lower risk.
Cholecalciferol requires two hydroxylation steps to become activated - first
in the liver to calcifediol, then in the kidney to calcitriol. Calcitriol,
paricalcitol, and alfacalcidol are active vitamin D analogs that do not
require conversion. This allows them to have more rapid onset of action
compared to cholecalciferol. The time delay for cholecalciferol to increase
serum calcifediol levels can be 2-3 days, and the delay for converting
calcifediol to active calcitriol can be up to 7 days.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
This study is excluded in meta-analysis:
results are only provided for use of one or more treatments within a class of treatments, results for each treatment are not provided; confounding by indication is likely and adjustments do not consider COVID-19 severity at baseline.
|
risk of death, 11.0% lower, OR 0.89, p = 0.005, adjusted per study, vitamin C or vitamin D, multivariable, RR approximated with OR.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Rosenthal et al., 10 Dec 2020, retrospective, database analysis, USA, peer-reviewed, 5 authors, dosage not specified.
Risk Factors Associated With In-Hospital Mortality in a US National Sample of Patients With COVID-19
JAMA Network Open, doi:10.1001/jamanetworkopen.2020.29058
IMPORTANCE Coronavirus disease 2019 (COVID-19) has infected more than 8.1 million US residents and killed more than 221 000. There is a dearth of research on epidemiology and clinical outcomes in US patients with COVID-19. OBJECTIVES To characterize patients with COVID-19 treated in US hospitals and to examine risk factors associated with in-hospital mortality. DESIGN, SETTING, AND PARTICIPANTS This cohort study was conducted using Premier Healthcare Database, a large geographically diverse all-payer hospital administrative database including 592 acute care hospitals in the United States. Inpatient and hospital-based outpatient visits with a principal or secondary discharge diagnosis of COVID-19 (International Classification of Diseases, Tenth Revision, Clinical Modification diagnosis code, U07.1) between April 1 and May 31, 2020, were included. EXPOSURES Characteristics of patients were reported by inpatient/outpatient and survival status. Risk factors associated with death examined included patient characteristics, acute complications, comorbidities, and medications. MAIN OUTCOMES AND MEASURES In-hospital mortality, intensive care unit (ICU) admission, use of invasive mechanical ventilation, total hospital length of stay (LOS), ICU LOS, acute complications, and treatment patterns. RESULTS Overall, 64 781 patients with COVID-19 (29 479 [45.5%] outpatients; 35 302 [54.5%] inpatients) were analyzed. The median (interquartile range [IQR]) age was 46 (33-59) years for outpatients and 65 (52-77) years for inpatients; 31 968 (49.3%) were men, 25 841 (39.9%) were White US residents, and 14 340 (22.1%) were Black US residents. In-hospital mortality was 20.3% among inpatients (7164 patients). A total of 5625 inpatients (15.9%) received invasive mechanical ventilation, and 6849 (19.4%) were admitted to the ICU. Median (IQR) inpatient LOS was 6 (3-10) days. Median (IQR) ICU LOS was 5 (2-10) days. Common acute complications among inpatients included acute respiratory failure (19 706 [55.8%]), acute kidney failure (11 971 [33.9%]), and sepsis (11 910 [33.7%]). Older age was the risk factor most strongly associated with death (eg, age Ն80 years vs 18-34 years: odds ratio [OR], 16.20; 95% CI, 11.58-22.67; P < .001). Receipt of statins (OR, 0.60; 95% CI, 0.56-0.65; P < .001), angiotensin-converting enzyme inhibitors (OR, 0.53; 95% CI, 0.46-0.60; P < .001), and calcium channel blockers (OR, 0.73; 95% CI, 0.68-0.79; P < .001) was associated with decreased odds of death. Compared with patients with no hydroxychloroquine or azithromycin, patients with both azithromycin and hydroxychloroquine had increased odds of death (OR, 1.21; 95% CI, 1.11-1.31; P < .001). (continued) Key Points Question What are the epidemiologic characteristics of patients with coronavirus disease 2019 (COVID-19) treated in US hospitals, and what risk factors are associated with mortality? Findings In this cohort study of 64 781 patients with COVID-19 treated in 592 US hospitals..
Additional Contributions: The authors would like to thank Bernadette Johnson Flavors, MBA, John House, MS, Teresa Davis, BS, Umang Patel, MS, and the entire PHD development team for making the data available on time for the analysis. The authors would also like to thank Denise Juliano, MS, Myla Maloney, MBA, BCMAS, Carol Cohen, BA, and the Premier Applied Sciences COVID-19 Task Force for their support with the analysis. All of the above individuals are employees of Premier Inc. No payment in addition to their regular salary was provided for their support. Dr Rosenthal has obtained written permission to include the names of individuals in this article.
References
Aggarwal, Garcia-Telles, Aggarwal, Lavie, Lippi et al., Clinical features, laboratory characteristics, and outcomes of patients hospitalized with coronavirus disease 2019 (COVID-19): early report from the United States, Diagnosis (Berl), doi:10.1515/dx-2020-0046
Boulware, Pullen, Bangdiwala, A randomized trial of hydroxychloroquine as postexposure prophylaxis for COVID-19, N Engl J Med, doi:10.1056/NEJMoa2016638
Brufsky, Hyperglycemia, hydroxychloroquine, and the COVID-19 pandemic, J Med Virol, doi:10.1002/jmv.25887
Burton, Fort, Seoane, Hospitalization and mortality among Black patients and White patients with COVID-19, N Engl J Med, doi:10.1056/NEJMsa2011686
Cavalcanti, Zampieri, Rosa, Coalition Covid-19 Brazil I Investigators. Hydroxychloroquine with or without azithromycin in mild-to-moderate COVID-19, N Engl J Med, doi:10.1056/NEJMoa2019014
Cdc Covid-, 19 Response Team. Severe outcomes among patients with coronavirus disease 2019 (COVID-19)-United States, MMWR Morb Mortal Wkly Rep, doi:10.15585/mmwr.mm6912e2
Cdc Covid-, Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019-United States, February 12, MMWR Morb Mortal Wkly Rep, doi:10.15585/mmwr.mm6913e2
Chary, Barbuto, Izadmehr, Hayes, Burns, COVID-19: therapeutics and their toxicities, J Med Toxicol, doi:10.1007/s13181-020-00777-5
Deyo, Cherkin, Ciol, Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases, J Clin Epidemiol, doi:10.1016/0895-4356(92)90133-8
Guzik, Mohiddin, Dimarco, COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options, Cardiovasc Res, doi:10.1093/cvr/cvaa106
Holshue, Debolt, Lindquist, Washington State 2019-nCoV Case Investigation Team. First case of 2019 novel coronavirus in the United States, N Engl J Med, doi:10.1056/NEJMoa2001191
Horby, Mafham, Linsell, Effect of hydroxychloroquine in hospitalized patients with COVID-19, N Engl J Med
Imam, Odish, Gill, Older age and comorbidity are independent mortality predictors in a large cohort of 1305 COVID-19 patients in Michigan, United States, J Intern Med, doi:10.1111/joim.13119
Lu, Chen, Lee, Chang, Potential therapeutic agents against COVID-19: what we know so far, J Chin Med Assoc, doi:10.1097/JCMA.0000000000000318
Piva, Filippini, Turla, Clinical presentation and initial management critically ill patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in Brescia, Italy, J Crit Care, doi:10.1016/j.jcrc.2020.04.004
Premier, Premier Healthcare Database: data that informs and performs
Rentsch, Kidwai-Khan, Tate, COVID-19 by race and ethnicity: a national cohort study of 6 million United States veterans. medRxiv, doi:10.1101/2020.05.12.20099135
Richardson, Hirsch, Narasimhan, the Northwell COVID-19 Research Consortium. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area, JAMA, doi:https://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.2020.6775&utm_campaign=articlePDF%26utm_medium=articlePDFlink%26utm_source=articlePDF%26utm_content=jamanetworkopen.2020.29058
Rubino, Kelvin, Bermejo-Martin, As COVID-19 cases, deaths and fatality rates surge in Italy, underlying causes require investigation, J Infect Dev Ctries, doi:10.3855/jidc.12734
Scuccimarri, Sutton, Fitzcharles, Hydroxychloroquine: a potential ethical dilemma for rheumatologists during the COVID-19 pandemic, J Rheumatol, doi:10.3899/jrheum.200369
Singh, Singh, Shaikh, Singh, Misra, Chloroquine and hydroxychloroquine in the treatment of COVID-19 with or without diabetes: a systematic search and a narrative review with a special reference to India and other developing countries, Diabetes Metab Syndr, doi:10.1016/j.dsx.2020.03.011
Xia, Wu, Liu, Xia, Jia et al., Epidemiological and initial clinical characteristics of patients with family aggregation of COVID-19, J Clin Virol, doi:10.1016/j.jcv.2020.104360
Zhai, Ding, Wu, Long, Zhong et al., The epidemiology, diagnosis and treatment of COVID-19, Int J Antimicrob Agents, doi:10.1016/j.ijantimicag.2020.105955
Zhou, Yu, Du, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study, Lancet, doi:10.1016/S0140-6736(20)30566-3
DOI record:
{
"DOI": "10.1001/jamanetworkopen.2020.29058",
"ISSN": [
"2574-3805"
],
"URL": "http://dx.doi.org/10.1001/jamanetworkopen.2020.29058",
"author": [
{
"affiliation": [
{
"name": "Premier Applied Sciences, Premier Inc, Charlotte, North Carolina"
}
],
"family": "Rosenthal",
"given": "Ning",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Premier Applied Sciences, Premier Inc, Charlotte, North Carolina"
}
],
"family": "Cao",
"given": "Zhun",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Premier Applied Sciences, Premier Inc, Charlotte, North Carolina"
}
],
"family": "Gundrum",
"given": "Jake",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Premier Applied Sciences, Premier Inc, Charlotte, North Carolina"
}
],
"family": "Sianis",
"given": "Jim",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Premier Applied Sciences, Premier Inc, Charlotte, North Carolina"
}
],
"family": "Safo",
"given": "Stella",
"sequence": "additional"
}
],
"container-title": [
"JAMA Network Open"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2020,
12,
10
]
],
"date-time": "2020-12-10T16:34:06Z",
"timestamp": 1607618046000
},
"deposited": {
"date-parts": [
[
2021,
1,
14
]
],
"date-time": "2021-01-14T16:14:01Z",
"timestamp": 1610640841000
},
"indexed": {
"date-parts": [
[
2022,
4,
12
]
],
"date-time": "2022-04-12T09:52:32Z",
"timestamp": 1649757152363
},
"is-referenced-by-count": 172,
"issn-type": [
{
"type": "electronic",
"value": "2574-3805"
}
],
"issue": "12",
"issued": {
"date-parts": [
[
2020,
12,
10
]
]
},
"journal-issue": {
"issue": "12",
"published-print": {
"date-parts": [
[
2020,
12,
1
]
]
}
},
"language": "en",
"link": [
{
"URL": "https://jamanetwork.com/journals/jamanetworkopen/articlepdf/2773971/rosenthal_2020_oi_200925_1610392201.69457.pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "10",
"original-title": [],
"page": "e2029058",
"prefix": "10.1001",
"published": {
"date-parts": [
[
2020,
12,
10
]
]
},
"published-online": {
"date-parts": [
[
2020,
12,
10
]
]
},
"publisher": "American Medical Association (AMA)",
"reference": [
{
"DOI": "10.1056/NEJMoa2001191",
"article-title": "First case of 2019 novel coronavirus in the United States.",
"author": "Holshue",
"doi-asserted-by": "crossref",
"first-page": "929",
"issue": "10",
"journal-title": "N Engl J Med",
"key": "zoi200925r2",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/j.jcrc.2020.04.004",
"article-title": "Clinical presentation and initial management critically ill patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in Brescia, Italy.",
"author": "Piva",
"doi-asserted-by": "crossref",
"first-page": "29",
"journal-title": "J Crit Care",
"key": "zoi200925r3",
"volume": "58",
"year": "2020"
},
{
"DOI": "10.3855/jidc.12734",
"article-title": "As COVID-19 cases, deaths and fatality rates surge in Italy, underlying causes require investigation.",
"author": "Rubino",
"doi-asserted-by": "crossref",
"first-page": "265",
"issue": "3",
"journal-title": "J Infect Dev Ctries",
"key": "zoi200925r4",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/j.jcv.2020.104360",
"article-title": "Epidemiological and initial clinical characteristics of patients with family aggregation of COVID-19.",
"author": "Xia",
"doi-asserted-by": "crossref",
"journal-title": "J Clin Virol",
"key": "zoi200925r5",
"volume": "127",
"year": "2020"
},
{
"DOI": "10.1016/j.ijantimicag.2020.105955",
"article-title": "The epidemiology, diagnosis and treatment of COVID-19.",
"author": "Zhai",
"doi-asserted-by": "crossref",
"issue": "5",
"journal-title": "Int J Antimicrob Agents",
"key": "zoi200925r6",
"volume": "55",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"article-title": "Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study.",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "1054",
"issue": "10229",
"journal-title": "Lancet",
"key": "zoi200925r7",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.15585/mmwr.mm6913e2",
"article-title": "Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019—United States, February 12-March 28, 2020.",
"author": "CDC COVID-19 Response Team",
"doi-asserted-by": "crossref",
"first-page": "382",
"issue": "13",
"journal-title": "MMWR Morb Mortal Wkly Rep",
"key": "zoi200925r8",
"volume": "69",
"year": "2020"
},
{
"DOI": "10.15585/mmwr.mm6912e2",
"article-title": "Severe outcomes among patients with coronavirus disease 2019 (COVID-19)—United States, February 12-March 16, 2020.",
"author": "CDC COVID-19 Response Team",
"doi-asserted-by": "crossref",
"first-page": "343",
"issue": "12",
"journal-title": "MMWR Morb Mortal Wkly Rep",
"key": "zoi200925r9",
"volume": "69",
"year": "2020"
},
{
"DOI": "10.1515/dx-2020-0046",
"article-title": "Clinical features, laboratory characteristics, and outcomes of patients hospitalized with coronavirus disease 2019 (COVID-19): early report from the United States.",
"author": "Aggarwal",
"doi-asserted-by": "crossref",
"first-page": "91",
"issue": "2",
"journal-title": "Diagnosis (Berl)",
"key": "zoi200925r10",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.6775",
"article-title": "Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area.",
"author": "Richardson",
"doi-asserted-by": "crossref",
"first-page": "2052",
"issue": "20",
"journal-title": "JAMA",
"key": "zoi200925r11",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1016/0895-4356(92)90133-8",
"article-title": "Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases.",
"author": "Deyo",
"doi-asserted-by": "crossref",
"first-page": "613",
"issue": "6",
"journal-title": "J Clin Epidemiol",
"key": "zoi200925r14",
"volume": "45",
"year": "1992"
},
{
"DOI": "10.1101/2020.05.12.20099135",
"article-title": "COVID-19 by race and ethnicity: a national cohort study of 6 million United States veterans.",
"author": "Rentsch",
"doi-asserted-by": "crossref",
"key": "zoi200925r15",
"year": "2020"
},
{
"DOI": "10.1056/NEJMsa2011686",
"article-title": "Hospitalization and mortality among Black patients and White patients with COVID-19.",
"author": "Price-Haywood",
"doi-asserted-by": "crossref",
"first-page": "2534",
"issue": "26",
"journal-title": "N Engl J Med",
"key": "zoi200925r16",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1111/joim.13119",
"article-title": "Older age and comorbidity are independent mortality predictors in a large cohort of 1305 COVID-19 patients in Michigan, United States.",
"author": "Imam",
"doi-asserted-by": "crossref",
"first-page": "469",
"issue": "4",
"journal-title": "J Intern Med",
"key": "zoi200925r17",
"volume": "288",
"year": "2020"
},
{
"DOI": "10.1002/jmv.25887",
"article-title": "Hyperglycemia, hydroxychloroquine, and the COVID-19 pandemic.",
"author": "Brufsky",
"doi-asserted-by": "crossref",
"first-page": "770",
"issue": "7",
"journal-title": "J Med Virol",
"key": "zoi200925r18",
"volume": "92",
"year": "2020"
},
{
"DOI": "10.1007/s13181-020-00777-5",
"article-title": "COVID-19: therapeutics and their toxicities.",
"author": "Chary",
"doi-asserted-by": "crossref",
"first-page": "284",
"issue": "3",
"journal-title": "J Med Toxicol",
"key": "zoi200925r19",
"volume": "16",
"year": "2020"
},
{
"DOI": "10.1093/cvr/cvaa106",
"article-title": "COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options.",
"author": "Guzik",
"doi-asserted-by": "crossref",
"first-page": "1666",
"issue": "10",
"journal-title": "Cardiovasc Res",
"key": "zoi200925r20",
"volume": "116",
"year": "2020"
},
{
"DOI": "10.1097/JCMA.0000000000000318",
"article-title": "Potential therapeutic agents against COVID-19: what we know so far.",
"author": "Lu",
"doi-asserted-by": "crossref",
"first-page": "534",
"issue": "6",
"journal-title": "J Chin Med Assoc",
"key": "zoi200925r21",
"volume": "83",
"year": "2020"
},
{
"DOI": "10.3899/jrheum.200369",
"article-title": "Hydroxychloroquine: a potential ethical dilemma for rheumatologists during the COVID-19 pandemic.",
"author": "Scuccimarri",
"doi-asserted-by": "crossref",
"first-page": "783",
"issue": "6",
"journal-title": "J Rheumatol",
"key": "zoi200925r22",
"volume": "47",
"year": "2020"
},
{
"DOI": "10.1016/j.dsx.2020.03.011",
"article-title": "Chloroquine and hydroxychloroquine in the treatment of COVID-19 with or without diabetes: a systematic search and a narrative review with a special reference to India and other developing countries.",
"author": "Singh",
"doi-asserted-by": "crossref",
"first-page": "241",
"issue": "3",
"journal-title": "Diabetes Metab Syndr",
"key": "zoi200925r23",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2019014",
"article-title": "Hydroxychloroquine with or without azithromycin in mild-to-moderate COVID-19.",
"author": "Cavalcanti",
"doi-asserted-by": "crossref",
"journal-title": "N Engl J Med",
"key": "zoi200925r24",
"year": "2020"
},
{
"article-title": "Effect of hydroxychloroquine in hospitalized patients with COVID-19.",
"author": "Horby",
"journal-title": "N Engl J Med",
"key": "zoi200925r25",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2016638",
"article-title": "A randomized trial of hydroxychloroquine as postexposure prophylaxis for COVID-19.",
"author": "Boulware",
"doi-asserted-by": "crossref",
"first-page": "517",
"issue": "6",
"journal-title": "N Engl J Med",
"key": "zoi200925r26",
"volume": "383",
"year": "2020"
},
{
"key": "zoi200925r1",
"unstructured": "US Centers for Disease Control and Prevention. CDC COVID data tracker. Accessed on November 19, 2020. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html"
},
{
"key": "zoi200925r12",
"unstructured": "Premier. Premier Healthcare Database: data that informs and performs. Published March 2, 2020. Accessed November 6, 2020. https://learn.premierinc.com/white-papers/premier-healthcare-database-whitepaper"
},
{
"key": "zoi200925r13",
"unstructured": "US Centers for Disease Control and Prevention. ICD-10-CM official coding and reporting guidelines, April 1, 2020 through September 30, 2020. Accessed on May 31, 2020. 2020. https://www.cdc.gov/nchs/data/icd/COVID-19-guidelines-final.pdf"
}
],
"reference-count": 26,
"references-count": 26,
"relation": {},
"resource": {
"primary": {
"URL": "https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2773971"
}
},
"score": 1,
"short-container-title": [
"JAMA Netw Open"
],
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": [
"Risk Factors Associated With In-Hospital Mortality in a US National Sample of Patients With COVID-19"
],
"type": "journal-article",
"volume": "3"
}
rosenthal
