Health-related quality of life in immunocompromised adults with mild–moderate COVID-19 treated with nirmatrelvir-ritonavir: results from the randomized, double-blinded EPIC-IC trial
et al., Health and Quality of Life Outcomes, doi:10.1186/s12955-026-02518-8, EPIC-IC, NCT05438602, Mar 2026
RCT 150 immunocompromised adults with mild-to-moderate COVID-19 comparing 5, 10, or 15 days of nirmatrelvir/ritonavir. There was no control group. No dose-response relationship was observed: HRQoL recovery patterns were statistically indistinguishable across the three treatment durations in the overall sample and in both severe and non-severe immunocompromised subgroups. The single nominally significant finding - higher day 10 physical health scores in the 10-day versus 5-day arm among severely immunocompromised participants arose from a small, post hoc subgroup analysis with notable baseline imbalances between arms, and the absence of any further improvement in the 15-day arm argues against a genuine treatment effect.
Mokgokong et al., 19 Mar 2026, Double Blind Randomized Controlled Trial, multiple countries, peer-reviewed, 5 authors, study period 3 August, 2022 - 13 November, 2023, trial NCT05438602 (history) (EPIC-IC).
Contact: ruth.mokgokong@pfizer.com.
Health-related quality of life in immunocompromised adults with mild–moderate COVID-19 treated with nirmatrelvir-ritonavir: results from the randomized, double-blinded EPIC-IC trial
Health and Quality of Life Outcomes, doi:10.1186/s12955-026-02518-8
Background: Little is known about health-related quality of life (HRQoL) in immunocompromised people during and after COVID-19 illness. We describe HRQoL outcomes from the EPIC-IC trial, which included participants with immunocompromise and mild-moderate COVID-19. Methods: EPIC-IC was a randomized, double-blind trial. Participants were assigned 1:1:1 to 5-day, 10-day, or 15-day nirmatrelvir-ritonavir (NMV/r) and completed the SF-36 and EQ-5D-5L through Week (W)24. HRQoL was analyzed across and by treatment arms for the evaluable population (N=150) and in post hoc subpopulations with severe (n=57) and non-severe (n=93) immunocompromise. Mixed-effects longitudinal models compared 5-day vs. 10-day or 15-day NMV/r.
Results: In the overall sample, mean baseline SF-36 domain scores (norm-based; mean=50, SD=10) ranged from 35.7-44.1, mean Physical Component Summary (PCS) score was 37.9, and mean Mental Component Summary (MCS) score was 41.5 -all substantially worse than cancer comparator norms and age-matched general population (AMGP) norms. Baseline mean EQ-5D-5L Index score was 0.65; participants reported problems with pain/discomfort (90% of participants), usual activities (73%), mobility (50%), anxiety/depression (48%), and self-care (29%). These proportions improved through Day (D)15 (change from baseline [CFB]: -49%points for pain/discomfort, -38%-points usual activities, -22%-points mobility, -24%points anxiety/depression, -19%-points self-care) and then stabilized. From the earliest post-baseline assessment (EQ-5D-5L: D5, SF-36: D10), all overall-sample outcomes improved significantly (p<0.05), with all mean improvements exceeding published minimum important difference thresholds (SF-36: 2-4-point; EQ-5D-5L
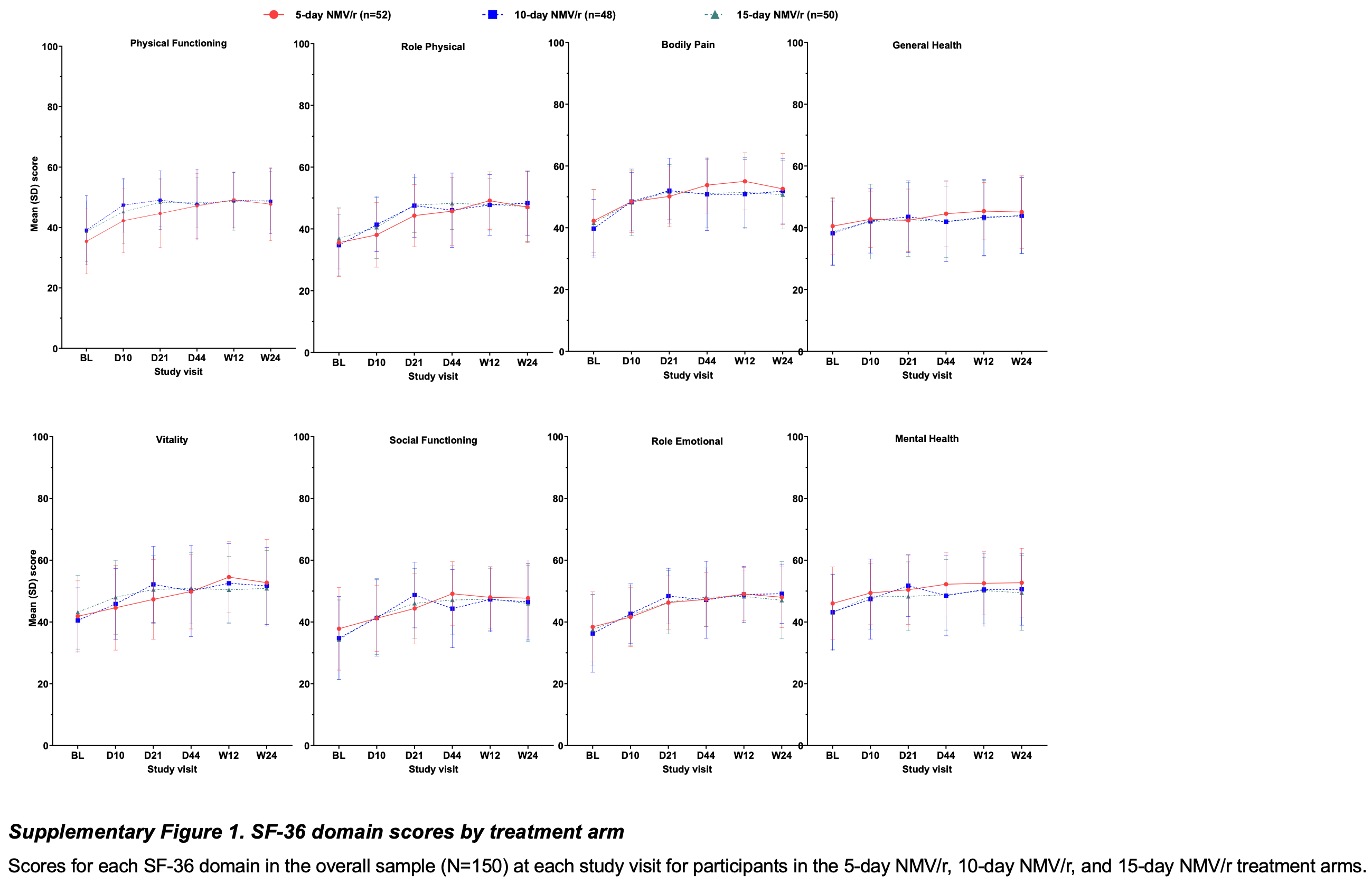
SF-36 completion and outcomes The SF-36 was completed by 51% of participants at baseline and by 83-90% of participants across post-baseline timepoints (Supplementary Table 1 ; Supplementary results).
SF-36 domain scores
Overall sample For all domains, baseline scores appeared to be similar across treatment arms (Supplementary Figure 1 ; Supplementary Tables 2 3 4 5 6 7 8 9 ). Across arms, mean baseline domain scores were lowest (i.e., indicating worse HRQoL) in Social Functioning (35.7 , norm-based with a mean of 50 and SD of 10) and Role-Physical (35.7) domains and highest in the Mental Health (44.1) domain (Supplementary Tables 2 3 4 5 6 7 8 9 ). For each domain, mean baseline scores in the overall sample were substantially lower (i.e., worse) than US general population norm scores for ages 55-64 [24] , population norm scores for ages 55-64 provided in the SF-36 User Manual [26] , and comparator scores for people with cancer (all cancers except skin cancer) [26] . Post-baseline improvements in the 5-day arm generally appeared similar to those in 10-day and 15-day arms (Supplementary Figure 1 ; Supplementary Tables 2 3 4 5 6 7 8 9 ). Consequently, scores are summarized below across treatment arms. For all domains, scores were significantly improved from baseline at each postbaseline visit (all p<0.05; Figure 3 ; Supplementary Tables 2 3 4 5 6 7 8 9 ). Mean improvements at each post-baseline visit were numerically greater than published MID values
List..
References
Ansari, Coetzer, Gebo, Ren, Scott et al., Patient-Reported Outcomes of Nirmatrelvir Treatment for High-Risk, Nonhospitalized Adults With Symptomatic COVID-19, Open Forum Infect Dis, doi:10.1093/ofid/ofaf449
Aranda, Oriol, Martín, Feria, Vázquez et al., Long-term impact of COVID-19 associated acute respiratory distress syndrome, J Infect, doi:10.1016/j.jinf.2021.08.018
Bartoletti, Azap, Barac, Selma, Ergonul et al., Management of COVID-19 in immunocompromised patients: an European Society of Clinical Microbiology and Infectious Diseases consensus document, Clin Microbiol Infect, doi:10.1016/j.cmi.2025.05.032
Beyer, Haufe, Meike, Scharbau, Lampe et al., Post-COVID-19 syndrome: Physical capacity, fatigue and quality of life, PLoS One, doi:10.1371/journal.pone.0292928
Bourke, Bennett, Oluboyede, Li, Longworth et al., Estimating the minimally important difference for the EQ-5D-5L and EORTC QLQ-C30 in cancer, Health Qual Life Outcomes, doi:10.1186/s12955-024-02294-3
Caso, Fernández-Ruiz, López-Medrano, Teller, Lizasoain et al., Nirmatrelvir/ritonavir for the treatment of immunocompromised adult patients with early-stage symptomatic COVID-19: A real-life experience, J Med Virol, doi:10.1002/jmv.29082
Cheng, Chen, Cheng, Herdman, Luo, Systematic review reveals that EQ-5D minimally important differences vary with treatment type and may decrease with increasing baseline score, J Clin Epidemiol, doi:10.1016/j.jclinepi.2024.111487
Devlin, Parkin, Janssen, Methods for Analysing and Reporting EQ-5D Data, Cham
Evans, Dube, Lu, Yates, Arnetorp et al., Impact of COVID-19 on immunocompromised populations during the Omicron era: insights from the observational population-based INFORM study, Lancet Reg Health Eur, doi:10.1016/j.lanepe.2023.100747
Fitzmaurice, Laird, Ware, Applied Longitudinal Analysis
Food, Administration, HIGHLIGHTS OF PRESCRIBING INFORMATION: PAXLOVID™ (nirmatrelvir tablets; ritonavir tablets), co-packaged for oral use
Fung, Babik, COVID-19 in Immunocompromised Hosts: What We Know So Far, Clinical Infectious Diseases, doi:10.1093/cid/ciaa863
Gidey, Niriayo, Asgedom, Lubetkin, Health-related quality of life in COVID-19 patients: a systematic review and meta-analysis of EQ-5D studies, Health and Quality of Life Outcomes, doi:10.1186/s12955-025-02421-8
Götz, Mathé, Agarwal, Hornuss, Pfau et al., Clinical phenotype and outcome of persistent SARS-CoV-2 replication in immunocompromised hosts: a retrospective observational study in the Omicron era, Infection, doi:10.1007/s15010-023-02138-0
Hammond, Leister-Tebbe, Gardner, Abreu, Wisemandle, Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19, N Engl J Med, doi:10.1056/NEJMoa2118542
Herdman, Gudex, Lloyd, Janssen, Kind et al., Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L), Qual Life Res, doi:10.1007/s11136-011-9903-x
Jiang, Janssen, Pickard, US population norms for the EQ-5D-5L and comparison of norms from face-to-face and online samples, Qual Life Res, doi:10.1007/s11136-020-02650-y
Li, Choudhary, Regan, Boucau, Nathan et al., SARS-CoV-2 viral clearance and evolution varies by type and severity of immunodeficiency, Sci Transl Med, doi:10.1126/scitranslmed.adk1599
Liu, Zhang, Yang, Zhang, Li et al., Respiratory rehabilitation in elderly patients with COVID-19: A randomized controlled study, Complement Ther Clin Pract, doi:10.1016/j.ctcp.2020.101166
Loubet, Benotmane, Fourati, Malard, Vuotto et al., Risk of Severe COVID-19 in Four Immunocompromised Populations: A French Expert Perspective, Infect Dis Ther, doi:10.1007/s40121-025-01124-3
Lubetkin, Long, Haagsma, Janssen, Bonsel, Health inequities as measured by the EQ-5D-5L during COVID-19: Results from New York in healthy and diseased persons, PLoS One, doi:10.1371/journal.pone.0272252
Maervoet, Bergemann, Potential Impact of EQ-5D-5L Mapping Function Recently Recommended by the National Institute for Health and Care Excellence on Utility Values and Decision Making, Value in Health, doi:10.1016/j.jval.2025.03.015
Maglinte, Hays, Kaplan, US general population norms for telephone administration of the SF-36v2, J Clin Epidemiol, doi:10.1016/j.jclinepi.2011.09.008
Meyyappan, Prasad, Kushwaha, Patel, Behera et al., Health-Related Quality of Life Score and Outcomes in Living Donor Renal Transplant Recipients With COVID-19, Exp Clin Transplant, doi:10.6002/ect.2021.0332
Mokgokong, Cislo, Tudone, Weinstein, Cappelleri, Symptom Alleviation/Resolution and Returns to Usual Health/Activities in Immunocompromised Adults with COVID-19 Treated with Nirmatrelvir-Ritonavir: Results from the EPIC-IC Trial, Infect Dis Ther, doi:10.1007/s40121-025-01228-w
Moser, Li, Eron, Aga, Daar et al., Predictors of SARS-CoV-2 RNA From Nasopharyngeal Swabs and Concordance With Other Compartments in Nonhospitalized Adults With Mild to Moderate COVID-19, Open Forum Infect Dis, doi:10.1093/ofid/ofac618
Mulhern, Feng, Shah, Janssen, Herdman et al., Comparing the UK EQ-5D-3L and English EQ-5D-5L Value Sets, Pharmacoeconomics, doi:10.1007/s40273-018-0628-3
Pickard, Law, Jiang, Pullenayegum, Shaw et al., United States Valuation of EQ-5D-5L Health States Using an International Protocol, Value in Health, doi:10.1016/j.jval.2019.02.009
Pinheiro, Hammerle, Sales, Freitas, Da et al., Assessment of health-related quality of life after mild COVID-19 infection, Acta Psychol (Amst), doi:10.1016/j.actpsy.2024.104422
Poudel, Zhu, Cooper, Roderick, Alwan et al., Impact of Covid-19 on health-related quality of life of patients: A structured review, PLoS One, doi:10.1371/journal.pone.0259164
Quint, Dube, Carty, Yokota, Bell et al., Immunocompromised individuals remain at risk of COVID-19: 2023 results from the observational INFORM study, J Infect, doi:10.1016/j.jinf.2025.106432
Rodríguez-Galán, Albaladejo-Blázquez, Ruiz-Robledillo, Pascual-Lledó, Ferrer-Cascales et al., Impact of COVID-19 on Health-Related Quality of Life: A Longitudinal Study in a Spanish Clinical Sample, Int J Environ Res Public Health, doi:10.3390/ijerph191610421
Roudijk, Ludwig, Devlin, EQ-5D-5L Value Set Summaries
Soare, Ansari, Nguyen, Mendes, Ahmed et al., Healthrelated quality of life in mild-to-moderate COVID-19 in the UK: a cross-sectional study from pre-to post-infection, Health Qual Life Outcomes, doi:10.1186/s12955-024-02230-5
Spinner, Bell, Einsele, Tremblay, Goldman et al., Is COVID-19 Still a Threat? An Expert Opinion Review on the Continued Healthcare Burden in Immunocompromised Individuals, Adv Ther, doi:10.1007/s12325-024-03043-0
Sun, Fusco, Puzniak, Coetzer, Zamparo et al., Assessment of retrospective collection of EQ-5D-5L in a US COVID-19 population, Health and Quality of Life Outcomes, doi:10.1186/s12955-023-02187-x
Trøseid, Hentzien, Ader, Cardoso, Arribas et al., Immunocompromised patients have been neglected in COVID-19 trials: a call for action, Clin Microbiol Infect, doi:10.1016/j.cmi.2022.05.005
Van Hout, Janssen, Feng, Kohlmann, Busschbach et al., Interim Scoring for the EQ-5D-5L: Mapping the EQ-5D-5L to EQ-5D-3L Value Sets, Value in Health, doi:10.1016/j.jval.2012.02.008
Verveen, Wynberg, Van Willigen, Davidovich, Lok et al., Health-related quality of life among persons with initial mild, moderate, and severe or critical COVID-19 at 1 and 12 months after infection: a prospective cohort study, BMC Med, doi:10.1186/s12916-022-02615-7
Wang, Luo, Wang, A comparative analysis of EQ-5D-5L general population norms across 23 countries: Gender and age disparities, Pharmacoecon Policy, doi:10.1016/j.pharp.2025.03.001
Ware, Kosinski, Turner-Bowker, Gandek, Maruish, User's Manual for the SF-36v2® Health Survey
Weinstein, Paredes, Gardner, Almas, Baniecki et al., Extended nirmatrelvir-ritonavir treatment durations for immunocompromised patients with COVID-19 (EPIC-IC): a placebo-controlled, randomised, double-blind, phase 2 trial, Lancet Infect Dis, doi:10.1016/S1473-3099(25)00221-X
Zhou, Guan, Wang, Zhang, Rui et al., Health-Related Quality of Life in Patients With Different Diseases Measured With the EQ-5D-5L: A Systematic Review, Front Public Health, doi:10.3389/fpubh.2021.675523
DOI record:
{
"DOI": "10.1186/s12955-026-02518-8",
"ISSN": [
"1477-7525"
],
"URL": "http://dx.doi.org/10.1186/s12955-026-02518-8",
"alternative-id": [
"2518"
],
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "12 November 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "12 March 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "19 March 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "Participants provided written informed consent before enrolling in the EPIC-IC trial [\n \n ]. Ethics approval for the protocol, amendments, and other relevant documents was granted by each institution’s review boards and ethics committees. Investigator names and approving ethics committees are listed in Weinstein, et al., 2025 [\n \n ]. The study was conducted in accordance with consensus ethical principles, including those in the Declaration of Helsinki and the Council for International Organizations of Medical Sciences (CIOMS) International Ethical Guidelines; with applicable International Council for Harmonisation of Technical Requirements of Pharmaceuticals for Human Use (ICH) guidelines for Good Clinical Practice; and with applicable laws and regulations, including applicable privacy laws."
},
{
"group": {
"label": "Consent for publication",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "Not applicable."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 4,
"value": "RM, PC, ET, EW, and JCC are employees and minority shareholders of Pfizer, which funded this study."
}
],
"author": [
{
"ORCID": "https://orcid.org/0000-0002-6278-4969",
"affiliation": [],
"authenticated-orcid": false,
"family": "Mokgokong",
"given": "Ruth",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0009-0004-3130-2818",
"affiliation": [],
"authenticated-orcid": false,
"family": "Cislo",
"given": "Paul",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0005-0406-7565",
"affiliation": [],
"authenticated-orcid": false,
"family": "Tudone",
"given": "Elena",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Weinstein",
"given": "Edward",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-9586-0748",
"affiliation": [],
"authenticated-orcid": false,
"family": "Cappelleri",
"given": "Joseph C.",
"sequence": "additional"
}
],
"container-title": "Health and Quality of Life Outcomes",
"container-title-short": "Health Qual Life Outcomes",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
19
]
],
"date-time": "2026-03-19T04:47:21Z",
"timestamp": 1773895641000
},
"deposited": {
"date-parts": [
[
2026,
3,
19
]
],
"date-time": "2026-03-19T04:47:30Z",
"timestamp": 1773895650000
},
"funder": [
{
"DOI": "10.13039/100004319",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/100004319",
"id-type": "DOI"
}
],
"name": "Pfizer"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
19
]
],
"date-time": "2026-03-19T09:14:32Z",
"timestamp": 1773911672247,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
19
]
]
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
19
]
],
"date-time": "2026-03-19T00:00:00Z",
"timestamp": 1773878400000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "am",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
19
]
],
"date-time": "2026-03-19T00:00:00Z",
"timestamp": 1773878400000
}
}
],
"link": [
{
"URL": "https://link.springer.com/article/10.1186/s12955-026-02518-8",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1186",
"published": {
"date-parts": [
[
2026,
3,
19
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
19
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1016/j.jinf.2021.08.018",
"author": "J Aranda",
"doi-asserted-by": "publisher",
"first-page": "581",
"issue": "5",
"journal-title": "J Infect",
"key": "2518_CR1",
"unstructured": "Aranda J, Oriol I, Martín M, Feria L, Vázquez N, Rhyman N, et al. Long-term impact of COVID-19 associated acute respiratory distress syndrome. J Infect. 2021;83(5):581–8. https://doi.org/10.1016/j.jinf.2021.08.018.",
"volume": "83",
"year": "2021"
},
{
"DOI": "10.1371/journal.pone.0292928",
"author": "S Beyer",
"doi-asserted-by": "publisher",
"first-page": "e0292928",
"issue": "10",
"journal-title": "PLoS ONE",
"key": "2518_CR2",
"unstructured": "Beyer S, Haufe S, Meike D, Scharbau M, Lampe V, Dopfer-Jablonka A, et al. Post-COVID-19 syndrome: Physical capacity, fatigue and quality of life. PLoS ONE. 2023;18(10):e0292928. https://doi.org/10.1371/journal.pone.0292928.",
"volume": "18",
"year": "2023"
},
{
"DOI": "10.1016/j.actpsy.2024.104422",
"author": "PG Pinheiro",
"doi-asserted-by": "publisher",
"first-page": "104422",
"journal-title": "Acta Psychol (Amst)",
"key": "2518_CR3",
"unstructured": "Pinheiro PG, Hammerle MB, Sales DS, Freitas DV, da Silva Souza R, Herzog ACF, et al. Assessment of health-related quality of life after mild COVID-19 infection. Acta Psychol (Amst). 2024;248:104422. https://doi.org/10.1016/j.actpsy.2024.104422.",
"volume": "248",
"year": "2024"
},
{
"DOI": "10.1371/journal.pone.0259164",
"author": "AN Poudel",
"doi-asserted-by": "publisher",
"first-page": "e0259164",
"issue": "10",
"journal-title": "PLoS ONE",
"key": "2518_CR4",
"unstructured": "Poudel AN, Zhu S, Cooper N, Roderick P, Alwan N, Tarrant C, et al. Impact of Covid-19 on health-related quality of life of patients: A structured review. PLoS ONE. 2021;16(10):e0259164. https://doi.org/10.1371/journal.pone.0259164.",
"volume": "16",
"year": "2021"
},
{
"DOI": "10.3390/ijerph191610421",
"doi-asserted-by": "publisher",
"key": "2518_CR5",
"unstructured": "Rodríguez-Galán I, Albaladejo-Blázquez N, Ruiz-Robledillo N, Pascual-Lledó JF, Ferrer-Cascales R, Gil-Carbonell J. Impact of COVID-19 on Health-Related Quality of Life: A Longitudinal Study in a Spanish Clinical Sample. Int J Environ Res Public Health. 2022;19(16). https://doi.org/10.3390/ijerph191610421."
},
{
"DOI": "10.1371/journal.pone.0272252",
"author": "EI Lubetkin",
"doi-asserted-by": "publisher",
"first-page": "e0272252",
"issue": "7",
"journal-title": "PLoS ONE",
"key": "2518_CR6",
"unstructured": "Lubetkin EI, Long D, Haagsma JA, Janssen MF, Bonsel GJ. Health inequities as measured by the EQ-5D-5L during COVID-19: Results from New York in healthy and diseased persons. PLoS ONE. 2022;17(7):e0272252. https://doi.org/10.1371/journal.pone.0272252.",
"volume": "17",
"year": "2022"
},
{
"DOI": "10.1186/s12955-024-02230-5",
"author": "IA Soare",
"doi-asserted-by": "publisher",
"first-page": "12",
"issue": "1",
"journal-title": "Health Qual Life Outcomes",
"key": "2518_CR7",
"unstructured": "Soare IA, Ansari W, Nguyen JL, Mendes D, Ahmed W, Atkinson J, et al. Health-related quality of life in mild-to-moderate COVID-19 in the UK: a cross-sectional study from pre- to post-infection. Health Qual Life Outcomes. 2024;22(1):12. https://doi.org/10.1186/s12955-024-02230-5.",
"volume": "22",
"year": "2024"
},
{
"DOI": "10.1186/s12916-022-02615-7",
"author": "A Verveen",
"doi-asserted-by": "publisher",
"first-page": "422",
"issue": "1",
"journal-title": "BMC Med",
"key": "2518_CR8",
"unstructured": "Verveen A, Wynberg E, van Willigen HDG, Davidovich U, Lok A, van Moll EP, et al. Health-related quality of life among persons with initial mild, moderate, and severe or critical COVID-19 at 1 and 12 months after infection: a prospective cohort study. BMC Med. 2022;20(1):422. https://doi.org/10.1186/s12916-022-02615-7.",
"volume": "20",
"year": "2022"
},
{
"DOI": "10.1007/s12325-024-03043-0",
"author": "CD Spinner",
"doi-asserted-by": "publisher",
"first-page": "666",
"issue": "2",
"journal-title": "Adv Ther",
"key": "2518_CR9",
"unstructured": "Spinner CD, Bell S, Einsele H, Tremblay C, Goldman M, Chagla Z, et al. Is COVID-19 Still a Threat? An Expert Opinion Review on the Continued Healthcare Burden in Immunocompromised Individuals. Adv Ther. 2025;42(2):666–719. https://doi.org/10.1007/s12325-024-03043-0.",
"volume": "42",
"year": "2025"
},
{
"DOI": "10.1016/j.cmi.2022.05.005",
"author": "M Trøseid",
"doi-asserted-by": "publisher",
"first-page": "1182",
"issue": "9",
"journal-title": "Clin Microbiol Infect",
"key": "2518_CR10",
"unstructured": "Trøseid M, Hentzien M, Ader F, Cardoso SW, Arribas JR, Molina J-M, et al. Immunocompromised patients have been neglected in COVID-19 trials: a call for action. Clin Microbiol Infect. 2022;28(9):1182–3. https://doi.org/10.1016/j.cmi.2022.05.005.",
"volume": "28",
"year": "2022"
},
{
"DOI": "10.1126/scitranslmed.adk1599",
"author": "Y Li",
"doi-asserted-by": "publisher",
"first-page": "eadk1599",
"issue": "731",
"journal-title": "Sci Transl Med",
"key": "2518_CR11",
"unstructured": "Li Y, Choudhary MC, Regan J, Boucau J, Nathan A, Speidel T, et al. SARS-CoV-2 viral clearance and evolution varies by type and severity of immunodeficiency. Sci Transl Med. 2024;16(731):eadk1599. https://doi.org/10.1126/scitranslmed.adk1599.",
"volume": "16",
"year": "2024"
},
{
"DOI": "10.1007/s40121-025-01124-3",
"author": "P Loubet",
"doi-asserted-by": "publisher",
"first-page": "671",
"issue": "4",
"journal-title": "Infect Dis Ther",
"key": "2518_CR12",
"unstructured": "Loubet P, Benotmane I, Fourati S, Malard F, Vuotto F, Blanchard E, et al. Risk of Severe COVID-19 in Four Immunocompromised Populations: A French Expert Perspective. Infect Dis Ther. 2025;14(4):671–733. https://doi.org/10.1007/s40121-025-01124-3.",
"volume": "14",
"year": "2025"
},
{
"DOI": "10.1016/j.jinf.2025.106432",
"author": "JK Quint",
"doi-asserted-by": "publisher",
"first-page": "106432",
"issue": "3",
"journal-title": "J Infect",
"key": "2518_CR13",
"unstructured": "Quint JK, Dube S, Carty L, Yokota R, Bell S, Turtle L, et al. Immunocompromised individuals remain at risk of COVID-19: 2023 results from the observational INFORM study. J Infect. 2025;90(3):106432. https://doi.org/10.1016/j.jinf.2025.106432.",
"volume": "90",
"year": "2025"
},
{
"DOI": "10.1056/NEJMoa2118542",
"author": "J Hammond",
"doi-asserted-by": "publisher",
"first-page": "1397",
"issue": "15",
"journal-title": "N Engl J Med",
"key": "2518_CR14",
"unstructured": "Hammond J, Leister-Tebbe H, Gardner A, Abreu P, Bao W, Wisemandle W, et al. Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with Covid-19. N Engl J Med. 2022;386(15):1397–408. https://doi.org/10.1056/NEJMoa2118542.",
"volume": "386",
"year": "2022"
},
{
"key": "2518_CR15",
"unstructured": "U.S. Food and Drug Administration. HIGHLIGHTS OF PRESCRIBING INFORMATION: PAXLOVID™ (nirmatrelvir tablets; ritonavir tablets), co-packaged for oral use. 2025. Updated 12-Feb-2025; Accessed 19-Feb-2025. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/217188s010lbl.pdf"
},
{
"DOI": "10.6002/ect.2021.0332",
"author": "J Meyyappan",
"doi-asserted-by": "publisher",
"first-page": "42",
"issue": "1",
"journal-title": "Exp Clin Transpl",
"key": "2518_CR16",
"unstructured": "Meyyappan J, Prasad N, Kushwaha R, Patel M, Behera M, Bhadauria D, et al. Health-Related Quality of Life Score and Outcomes in Living Donor Renal Transplant Recipients With COVID-19. Exp Clin Transpl. 2022;20(1):42–51. https://doi.org/10.6002/ect.2021.0332.",
"volume": "20",
"year": "2022"
},
{
"DOI": "10.1007/s40121-025-01228-w",
"author": "R Mokgokong",
"doi-asserted-by": "publisher",
"journal-title": "Infect Dis Ther",
"key": "2518_CR17",
"unstructured": "Mokgokong R, Cislo P, Tudone E, Weinstein E, Cappelleri JC. Symptom Alleviation/Resolution and Returns to Usual Health/Activities in Immunocompromised Adults with COVID-19 Treated with Nirmatrelvir-Ritonavir: Results from the EPIC-IC Trial. Infect Dis Ther. 2025. https://doi.org/10.1007/s40121-025-01228-w.",
"year": "2025"
},
{
"DOI": "10.1016/S1473-3099(25)00221-X",
"author": "E Weinstein",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Infect Dis",
"key": "2518_CR18",
"unstructured": "Weinstein E, Paredes R, Gardner A, Almas M, Baniecki ML, Guan S, et al. Extended nirmatrelvir–ritonavir treatment durations for immunocompromised patients with COVID-19 (EPIC-IC): a placebo-controlled, randomised, double-blind, phase 2 trial. Lancet Infect Dis. 2025. https://doi.org/10.1016/S1473-3099(25)00221-X.",
"year": "2025"
},
{
"key": "2518_CR19",
"unstructured": "Infectious Diseases Society of America. IDSA Guidelines on the Treatment and Management of Patients with COVID-19. 2025. Updated 14 October 2025; Accessed 11 February 2026. Available from: https://www.idsociety.org/globalassets/idsa/practice-guidelines/covid-19/treatment/antiviral-treatment-for-mild-to-moderate-covid-19-in-adults.pdf; https://www.idsociety.org/COVID19guidelines"
},
{
"DOI": "10.1016/j.cmi.2025.05.032",
"author": "M Bartoletti",
"doi-asserted-by": "publisher",
"first-page": "1655",
"issue": "10",
"journal-title": "Clin Microbiol Infect",
"key": "2518_CR20",
"unstructured": "Bartoletti M, Azap O, Barac A, Ben Selma M, Ergonul O, Gkrania-Klotsas E, et al. Management of COVID-19 in immunocompromised patients: an European Society of Clinical Microbiology and Infectious Diseases consensus document. Clin Microbiol Infect. 2025;31(10):1655–66. https://doi.org/10.1016/j.cmi.2025.05.032.",
"volume": "31",
"year": "2025"
},
{
"key": "2518_CR21",
"unstructured": "National Institute for Health and Care Excellence (NICE). Nirmatrelvir plus ritonavir, sotrovimab and tocilizumab for treating COVID-19: Technology appraisal guidance. 2025. Updated 01 May 2025; Accessed 13 February 2026. Available from: https://www.nice.org.uk/guidance/ta878/chapter/1-Recommendations"
},
{
"DOI": "10.1002/jmv.29082",
"author": "JM Caso",
"doi-asserted-by": "publisher",
"first-page": "e29082",
"issue": "9",
"journal-title": "J Med Virol",
"key": "2518_CR22",
"unstructured": "Caso JM, Fernández-Ruiz M, López-Medrano F, Caro-Teller JM, Lizasoain M, San-Juan R, et al. Nirmatrelvir/ritonavir for the treatment of immunocompromised adult patients with early-stage symptomatic COVID-19: A real-life experience. J Med Virol. 2023;95(9):e29082. https://doi.org/10.1002/jmv.29082.",
"volume": "95",
"year": "2023"
},
{
"DOI": "10.1007/s15010-023-02138-0",
"author": "V Götz",
"doi-asserted-by": "publisher",
"first-page": "923",
"issue": "3",
"journal-title": "Infection",
"key": "2518_CR23",
"unstructured": "Götz V, Mathé P, Agarwal P, Hornuss D, Pfau S, Panning M, et al. Clinical phenotype and outcome of persistent SARS-CoV-2 replication in immunocompromised hosts: a retrospective observational study in the Omicron era. Infection. 2024;52(3):923–33. https://doi.org/10.1007/s15010-023-02138-0.",
"volume": "52",
"year": "2024"
},
{
"DOI": "10.1016/j.jclinepi.2011.09.008",
"author": "GA Maglinte",
"doi-asserted-by": "publisher",
"first-page": "497",
"issue": "5",
"journal-title": "J Clin Epidemiol",
"key": "2518_CR24",
"unstructured": "Maglinte GA, Hays RD, Kaplan RM. US general population norms for telephone administration of the SF-36v2. J Clin Epidemiol. 2012;65(5):497–502. https://doi.org/10.1016/j.jclinepi.2011.09.008.",
"volume": "65",
"year": "2012"
},
{
"DOI": "10.1007/978-3-030-47622-9",
"doi-asserted-by": "crossref",
"key": "2518_CR25",
"unstructured": "Devlin N, Parkin D, Janssen B. Methods for analysing and reporting EQ-5D Data. Cham (CH); 2020."
},
{
"key": "2518_CR26",
"unstructured": "Ware JE Jr., Kosinski M, B JB, Turner-Bowker DM, Gandek B, Maruish ME. User’s Manual for the SF-36v2® Health Survey. 2nd ed. In: QualityMetric Incorporated; Lincoln, RI;2007."
},
{
"DOI": "10.1186/s12955-024-02294-3",
"author": "S Bourke",
"doi-asserted-by": "publisher",
"first-page": "81",
"issue": "1",
"journal-title": "Health Qual Life Outcomes",
"key": "2518_CR27",
"unstructured": "Bourke S, Bennett B, Oluboyede Y, Li T, Longworth L, O’Sullivan SB, et al. Estimating the minimally important difference for the EQ-5D-5L and EORTC QLQ-C30 in cancer. Health Qual Life Outcomes. 2024;22(1):81. https://doi.org/10.1186/s12955-024-02294-3.",
"volume": "22",
"year": "2024"
},
{
"DOI": "10.1007/s11136-011-9903-x",
"author": "M Herdman",
"doi-asserted-by": "publisher",
"first-page": "1727",
"issue": "10",
"journal-title": "Qual Life Res",
"key": "2518_CR28",
"unstructured": "Herdman M, Gudex C, Lloyd A, Janssen MF, Kind P, Parkin D, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res. 2011;20(10):1727–36. https://doi.org/10.1007/s11136-011-9903-x.",
"volume": "20",
"year": "2011"
},
{
"DOI": "10.1007/s11136-020-02650-y",
"author": "R Jiang",
"doi-asserted-by": "publisher",
"first-page": "803",
"issue": "3",
"journal-title": "Qual Life Res",
"key": "2518_CR29",
"unstructured": "Jiang R, Janssen MFB, Pickard AS. US population norms for the EQ-5D-5L and comparison of norms from face-to-face and online samples. Qual Life Res. 2021;30(3):803–16. https://doi.org/10.1007/s11136-020-02650-y.",
"volume": "30",
"year": "2021"
},
{
"DOI": "10.1007/978-3-030-89289-0_4",
"doi-asserted-by": "crossref",
"key": "2518_CR30",
"unstructured": "Roudijk B, Ludwig K, Devlin N. EQ-5D-5L Value Set Summaries. In: Devlin N, Roudijk B, Ludwig K, editors. Value Sets for EQ-5D-5L: A compendium, comparative review & user guide. Cham (CH); 2022. pp. 55–212."
},
{
"DOI": "10.1016/j.pharp.2025.03.001",
"author": "Z Wang",
"doi-asserted-by": "publisher",
"first-page": "5",
"issue": "1",
"journal-title": "Pharmacoecon Policy",
"key": "2518_CR31",
"unstructured": "Wang Z, Luo N, Wang P. A comparative analysis of EQ-5D-5L general population norms across 23 countries: Gender and age disparities. Pharmacoecon Policy. 2025;1(1):5–14. https://doi.org/10.1016/j.pharp.2025.03.001.",
"volume": "1",
"year": "2025"
},
{
"DOI": "10.3389/fpubh.2021.675523",
"doi-asserted-by": "publisher",
"key": "2518_CR32",
"unstructured": "Zhou T, Guan H, Wang L, Zhang Y, Rui M, Ma A. Health-related quality of life in patients with different diseases measured with the EQ-5D-5L: a systematic review. Front Public Health. 2021;9–2021. https://doi.org/10.3389/fpubh.2021.675523."
},
{
"DOI": "10.1186/s12955-025-02421-8",
"author": "K Gidey",
"doi-asserted-by": "publisher",
"first-page": "97",
"issue": "1",
"journal-title": "Health Qual Life Outcomes",
"key": "2518_CR33",
"unstructured": "Gidey K, Niriayo YL, Asgedom SW, Lubetkin E. Health-related quality of life in COVID-19 patients: a systematic review and meta-analysis of EQ-5D studies. Health Qual Life Outcomes. 2025;23(1):97. https://doi.org/10.1186/s12955-025-02421-8.",
"volume": "23",
"year": "2025"
},
{
"key": "2518_CR34",
"unstructured": "U.S. Food and Drug Administration. Assessing COVID-19-Related Symptoms in Outpatient Adult and Adolescent Subjects in Clinical Trials of Drugs and Biological Products for COVID-19 Prevention or Treatment: Guidance for Industry. 2024. Updated February 2024; Accessed 30 January 2026."
},
{
"key": "2518_CR35",
"unstructured": "US Centers for Disease Control and Prevention. Underlying Conditions and the Higher Risk for Severe COVID-19. 2025. Updated 06-Feb-2025; Accessed 12-Jun-2025. Available from: https://www.cdc.gov/covid/hcp/clinical-care/underlying-conditions.html"
},
{
"DOI": "10.1016/j.jval.2012.02.008",
"author": "B van Hout",
"doi-asserted-by": "publisher",
"first-page": "708",
"issue": "5",
"journal-title": "Value Health",
"key": "2518_CR36",
"unstructured": "van Hout B, Janssen MF, Feng Y-S, Kohlmann T, Busschbach J, Golicki D, et al. Interim Scoring for the EQ-5D-5L: Mapping the EQ-5D-5L to EQ-5D-3L Value Sets. Value Health. 2012;15(5):708–15. https://doi.org/10.1016/j.jval.2012.02.008.",
"volume": "15",
"year": "2012"
},
{
"key": "2518_CR37",
"unstructured": "National Institute for Health and Care Excellence (NICE). Use of the EQ-5D-5L value set for England: NICE position statement. 2019. Updated 31 October 2019; Accessed 10 February 2026. Available from: https://www.nice.org.uk/corporate/ecd16/chapter/position-statement"
},
{
"DOI": "10.1002/9781119513469",
"doi-asserted-by": "crossref",
"key": "2518_CR38",
"unstructured": "Fitzmaurice GM, Laird NM, Ware JH. Applied longitudinal analysis. 2nd ed. In: John Wiley & Sons, Inc.; 2011."
},
{
"DOI": "10.1007/s40273-018-0628-3",
"author": "B Mulhern",
"doi-asserted-by": "publisher",
"first-page": "699",
"issue": "6",
"journal-title": "PharmacoEconomics",
"key": "2518_CR39",
"unstructured": "Mulhern B, Feng Y, Shah K, Janssen MF, Herdman M, van Hout B, Devlin N. Comparing the UK EQ-5D-3L and English EQ-5D-5L Value Sets. PharmacoEconomics. 2018;36(6):699–713. https://doi.org/10.1007/s40273-018-0628-3.",
"volume": "36",
"year": "2018"
},
{
"DOI": "10.1016/j.jval.2019.02.009",
"author": "AS Pickard",
"doi-asserted-by": "publisher",
"first-page": "931",
"issue": "8",
"journal-title": "Value Health",
"key": "2518_CR40",
"unstructured": "Pickard AS, Law EH, Jiang R, Pullenayegum E, Shaw JW, Xie F, et al. United States Valuation of EQ-5D-5L Health States Using an International Protocol. Value Health. 2019;22(8):931–41. https://doi.org/10.1016/j.jval.2019.02.009.",
"volume": "22",
"year": "2019"
},
{
"DOI": "10.1016/j.jclinepi.2024.111487",
"author": "LJ Cheng",
"doi-asserted-by": "publisher",
"first-page": "111487",
"journal-title": "J Clin Epidemiol",
"key": "2518_CR41",
"unstructured": "Cheng LJ, Chen LA, Cheng JY, Herdman M, Luo N. Systematic review reveals that EQ-5D minimally important differences vary with treatment type and may decrease with increasing baseline score. J Clin Epidemiol. 2024;174:111487. https://doi.org/10.1016/j.jclinepi.2024.111487.",
"volume": "174",
"year": "2024"
},
{
"DOI": "10.1016/j.jval.2025.03.015",
"author": "J Maervoet",
"doi-asserted-by": "publisher",
"first-page": "1038",
"issue": "7",
"journal-title": "Value Health",
"key": "2518_CR42",
"unstructured": "Maervoet J, Bergemann R, the National Institute for Health and Care Excellence on Utility Values and Decision Making. Potential Impact of EQ-5D-5L Mapping Function Recently Recommended by. Value Health. 2025;28(7):1038–45. https://doi.org/10.1016/j.jval.2025.03.015.",
"volume": "28",
"year": "2025"
},
{
"DOI": "10.1016/j.ctcp.2020.101166",
"author": "K Liu",
"doi-asserted-by": "publisher",
"first-page": "101166",
"journal-title": "Complement Ther Clin Pract",
"key": "2518_CR43",
"unstructured": "Liu K, Zhang W, Yang Y, Zhang J, Li Y, Chen Y. Respiratory rehabilitation in elderly patients with COVID-19: A randomized controlled study. Complement Ther Clin Pract. 2020;39:101166. https://doi.org/10.1016/j.ctcp.2020.101166.",
"volume": "39",
"year": "2020"
},
{
"DOI": "10.1093/ofid/ofaf449",
"author": "W Ansari",
"doi-asserted-by": "publisher",
"journal-title": "Open Forum Infect Dis",
"key": "2518_CR44",
"unstructured": "Ansari W, Coetzer H, Gebo KA, Ren J, Scott A, Cappelleri JC, et al. Patient-Reported Outcomes of Nirmatrelvir Treatment for High-Risk, Nonhospitalized Adults With Symptomatic COVID-19. Open Forum Infect Dis. 2025. https://doi.org/10.1093/ofid/ofaf449.",
"year": "2025"
},
{
"DOI": "10.1186/s12955-023-02187-x",
"author": "X Sun",
"doi-asserted-by": "publisher",
"first-page": "103",
"issue": "1",
"journal-title": "Health Qual Life Outcomes",
"key": "2518_CR45",
"unstructured": "Sun X, Di Fusco M, Puzniak L, Coetzer H, Zamparo JM, Tabak YP, Cappelleri JC. Assessment of retrospective collection of EQ-5D-5L in a US COVID-19 population. Health Qual Life Outcomes. 2023;21(1):103. https://doi.org/10.1186/s12955-023-02187-x.",
"volume": "21",
"year": "2023"
},
{
"key": "2518_CR46",
"unstructured": "US Food and Drug Administration. Assessing COVID-19-Related Symptoms in Outpatient Adult and Adolescent Subjects in Clinical Trials of Drugs and Biological Products for COVID-19 Prevention or Treatment: Guidance for Industry. 2024. Accessed 05-Aug-2024. Available from: https://www.fda.gov/media/167275/download"
},
{
"key": "2518_CR47",
"unstructured": "EuroQol Research Foundation, EQ-5D-5L User Guide. 2025. Updated Aug-2025; Accessed 23-Oct-2025. Available from: https://euroqol.org/information-and-support/documentation/user-guides/"
},
{
"DOI": "10.1093/ofid/ofac618",
"author": "C Moser",
"doi-asserted-by": "publisher",
"first-page": "ofac618",
"issue": "11",
"journal-title": "Open Forum Infect Dis",
"key": "2518_CR48",
"unstructured": "Moser C, Li JZ, Eron JJ, Aga E, Daar ES, Wohl DA, et al. Predictors of SARS-CoV-2 RNA From Nasopharyngeal Swabs and Concordance With Other Compartments in Nonhospitalized Adults With Mild to Moderate COVID-19. Open Forum Infect Dis. 2022;9(11):ofac618. https://doi.org/10.1093/ofid/ofac618.",
"volume": "9",
"year": "2022"
},
{
"DOI": "10.1016/j.lanepe.2023.100747",
"doi-asserted-by": "publisher",
"key": "2518_CR49",
"unstructured": "Evans RA, Dube S, Lu Y, Yates M, Arnetorp S, Barnes E, et al. Impact of COVID-19 on immunocompromised populations during the Omicron era: insights from the observational population-based INFORM study. Lancet Reg Health Eur. 2023;35. https://doi.org/10.1016/j.lanepe.2023.100747."
},
{
"DOI": "10.1093/cid/ciaa863",
"author": "M Fung",
"doi-asserted-by": "publisher",
"first-page": "340",
"issue": "2",
"journal-title": "Clin Infect Dis",
"key": "2518_CR50",
"unstructured": "Fung M, Babik JM. COVID-19 in Immunocompromised Hosts: What We Know So Far. Clin Infect Dis. 2020;72(2):340–50. https://doi.org/10.1093/cid/ciaa863.",
"volume": "72",
"year": "2020"
}
],
"reference-count": 50,
"references-count": 50,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1186/s12955-026-02518-8"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Health-related quality of life in immunocompromised adults with mild–moderate COVID-19 treated with nirmatrelvir-ritonavir: results from the randomized, double-blinded EPIC-IC trial",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy"
}