Effectiveness of a sustained-release ammonium chloride formulation in reducing the viral load of patients with COVID-19 or influenza: A prospective, randomized, double-blind, placebo-controlled study
et al., Research Square, doi:10.21203/rs.3.rs-9009421/v1, NCT07254052, Mar 2026
RCT 32 outpatients (28 with COVID-19) showing potential virological benefit with ammonium chloride. All participants experienced mild illness with no progression to severe disease, hospitalization, or death in either cohort. There was a statistically significant reduction in viral load by day 10-11 in the treatment group. The reliability of these results is weakened by the following issues:
Retrospective clinical trial registration: the trial was registered in ClinicalTrials.gov and ISRCTN in October and November 2025, which is months after the reported completion of the study in April 2025. Retrospective registration allows for selective reporting and endpoint switching.
Data duplication: in the VDF (placebo) group, the data for Participant 17 (P17) and Participant 19 (P19) are identical across all three timepoints down to two decimal places. In real-world biological PCR testing, two different humans having the exact same cycle thresholds to the hundredth decimal place on three separate days is statistically very unlikely.
Two authors (Dr. Drakoulis and Dr. Tsirikos-Karapanos) hold direct intellectual property and financial interests in the proprietary intervention evaluated in the study. The study was also partially funded by Metron Nutraceuticals, the manufacturer.
There is a large baseline imbalance in comorbidities between the ACF group (68.8%) and the VDF group (25.0%) with a p-value of 0.02.
The registry lists symptom duration/severity and several severe outcomes as secondary endpoints, however authors report only a narrative statement that no severe outcomes occurred and no symptom results.
Authors repeatedly describes the Day 10-11 Ct result as statistically significant, however Table 3 reports FDR-adjusted q-values of about 0.098 to 0.10 for the adjusted analyses, indicating that the evidence is less robust than the narrative suggests.
Inclusion of an "already cleared" patient: participant P14 in the ACF treatment group was enrolled with a day 1 baseline Ct of 39.60. Because the study defines "viral clearance" as a Ct ≥40, this patient was enrolled with a viral load so low they had effectively already cleared the virus before treatment began. This patient contributes to the ACF group's high clearance rate and favorable Ct trajectory despite having little meaningful viral load to reduce.
The main paper defines the third virologic assessment as Day 10-11, whereas the supplement labels Timepoint 3 as "7-10 days after baseline".
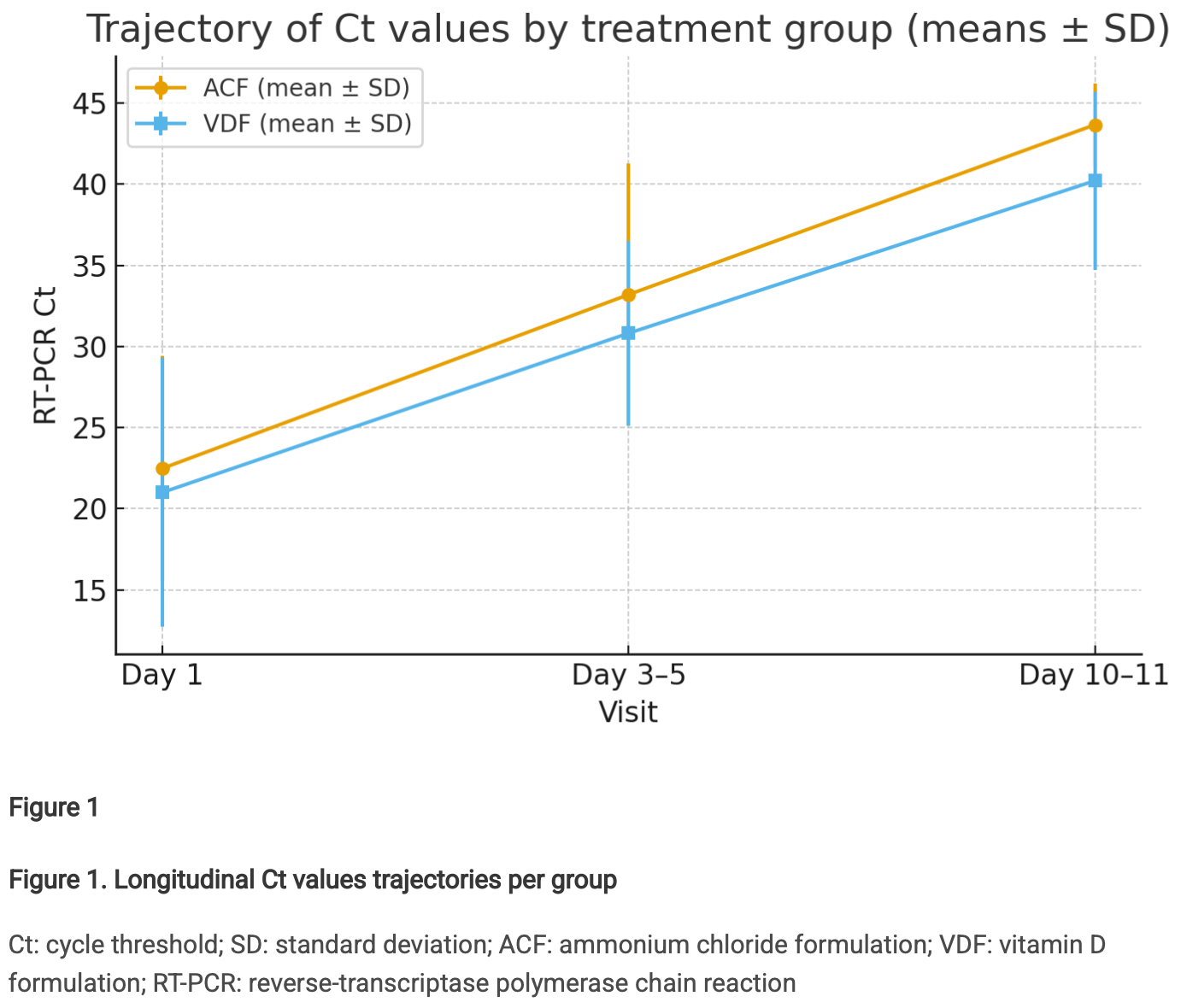
In the ACF arm at day 10-11, 12/16 values are 45.00 and 14/16 are at least 40, so the endpoint is strongly compressed at the upper bound, indicating that the Gaussian-style mean/SD analyses are less comfortable than the paper suggests, because much of the separation is being driven by a ceiling-censored distribution.
Maltezou et al., 8 Mar 2026, Double Blind Randomized Controlled Trial, placebo-controlled, USA, preprint, mean age 58.1, 9 authors, study period 1 September, 2024 - 30 April, 2025, trial NCT07254052 (history).
Abstract: Effectiveness of a sustained-release ammonium
chloride formulation in reducing the viral load of
patients with COVID-19 or influenza: A prospective,
randomized, double-blind, placebo-controlled study
Helena C. Maltezou
National Public Health Organization
Constantine Chalkias
En Ygeia Clinic
Garyfalia Poulakou
National and Kapodistrian University of Athens
Annia Tsolakou
National and Kapodistrian University of Athens
Panagiotis Xintaropoulos
National and Kapodistrian University of Athens
Athanasios Raptis
National School of Public Administration
Nikolaos Syrigos
National and Kapodistrian University of Athens
Nikolaos Tsirikos-Karapanos
Metron Nutraceuticals
Nikolaos Drakoulis
National and Kapodistrian University of Athens
Research Article
Keywords: ammonium chloride, NH4Cl, influenza, SARS-CoV-2, RNA virus infections, virus clearance
Posted Date: March 8th, 2026
DOI: https://doi.org/10.21203/rs.3.rs-9009421/v1
Page 1/21
License: This work is licensed under a Creative Commons Attribution 4.0 International License.
Read Full License
Additional Declarations: The authors declare potential competing interests as follows: Competening
Interests Dr. Drakoulis has an interest in the intellectual property of DIVIRNAM®. Dr. Tsirikos-Karapanos
has an interest in the intellectual property of and direct financial interest in DIVIRNAM®. Dr. Drakoulis’
and Dr. Tsirikos-Karapanos’ contribution in the study was only in the design of the study’s clinical
research protocol. All other authors declare no conflict of interest.
Page 2/21
Abstract
Background
We estimated the effectiveness of a novel sustained-release dietary supplement formulation containing
500 mg ammonium chloride and 2,000 IU vitamin D (ACF;) in reducing the viral load of patients with
COVID-19 or influenza.
Methods
In this prospective, randomized, double-blind, placebo-controlled, study. Eligible patients with COVID-19
or influenza were randomized to receive ACF twice daily or placebo (2,000 IU vitamin D/twice daily; VDF)
for 10 days. Nasopharyngeal swab samples were collected at Day 1, Day 3–5 and Day 10–11 and tested
for SARS-CoV-2 and influenza via RT-PCR. Cycle threshold (Ct) values were measured. The study has
been retrospectively registered in ClinicalTrials.gov (ClinicalTrials.gov identifier: NCT07254052) and the
ISRCTN registry (ISRCTN study registration number: ISRCTN48259966).
Results
Thirty two patients were studied, 28 with COVID-19 and 4 with influenza. No patient developed severe
disease, was hospitalized, or died. Sixteen patients received ACF and 16 VDF (mean age: 58.1 and 60.7
years, respectively; 68.8% and 25% with comorbidities, respectively). On Day 1, the mean Cts were 22.49
in ACF group and 21.01 in VDF group, on Day 3–5, the mean Cts were 33.20 and 30.82, respectively, and
on Day 10–11, the mean Cts were 43.66 and 40.21, respectively. On Day 10–11 the adjusted mean
difference was + 3.12 cycles (95% confidence interval: 0.22–6.02; p-value = 0.036). The Kaplan Meier
analysis indicated faster clearance in the ACF group compared to the VDF group (p-value = 0.016).
Conclusions
Our data indicate that ACF-receiving patients had a statistically significant reduction in viral load
compared to placebo-receiving patients. This is attributed to the pharmacodynamic action of ammonium
chloride and the pharmacokinetic properties of ACF. Larger studies are needed to further investigate the
role of ACF in various RNA-viral infections.
Trial registration:
This study was..
DOI record:
{
"DOI": "10.21203/rs.3.rs-9009421/v1",
"URL": "http://dx.doi.org/10.21203/rs.3.rs-9009421/v1",
"abstract": "<title>Abstract</title>\n <p>Background\n We estimated the effectiveness of a novel sustained-release dietary supplement formulation containing 500 mg ammonium chloride and 2,000 IU vitamin D (ACF;) in reducing the viral load of patients with COVID-19 or influenza.\nMethods\n In this prospective, randomized, double-blind, placebo-controlled, study. Eligible patients with COVID-19 or influenza were randomized to receive ACF twice daily or placebo (2,000 IU vitamin D/twice daily; VDF) for 10 days. Nasopharyngeal swab samples were collected at Day 1, Day 3–5 and Day 10–11 and tested for SARS-CoV-2 and influenza via RT-PCR. Cycle threshold (Ct) values were measured. The study has been retrospectively registered in ClinicalTrials.gov (ClinicalTrials.gov identifier: NCT07254052) and the ISRCTN registry (ISRCTN study registration number: ISRCTN48259966).\nResults\n Thirty two patients were studied, 28 with COVID-19 and 4 with influenza. No patient developed severe disease, was hospitalized, or died. Sixteen patients received ACF and 16 VDF (mean age: 58.1 and 60.7 years, respectively; 68.8% and 25% with comorbidities, respectively). On Day 1, the mean Cts were 22.49 in ACF group and 21.01 in VDF group, on Day 3–5, the mean Cts were 33.20 and 30.82, respectively, and on Day 10–11, the mean Cts were 43.66 and 40.21, respectively. On Day 10–11 the adjusted mean difference was + 3.12 cycles (95% confidence interval: 0.22–6.02; p-value = 0.036). The Kaplan Meier analysis indicated faster clearance in the ACF group compared to the VDF group (p-value = 0.016).\nConclusions\n Our data indicate that ACF-receiving patients had a statistically significant reduction in viral load compared to placebo-receiving patients. This is attributed to the pharmacodynamic action of ammonium chloride and the pharmacokinetic properties of ACF. Larger studies are needed to further investigate the role of ACF in various RNA-viral infections.\nTrial registration:\n This study was retrospectively registered in ClinicalTrials.gov (identifier: NCT07254052; registered on 22 October 2025) and in the ISRCTN registry (registration number: ISRCTN48259966; registered on 27 November 2025).</p>",
"accepted": {
"date-parts": [
[
2026,
3,
2
]
]
},
"author": [
{
"affiliation": [
{
"name": "National Public Health Organization"
}
],
"family": "Maltezou",
"given": "Helena C.",
"sequence": "first"
},
{
"affiliation": [
{
"name": "En Ygeia Clinic"
}
],
"family": "Chalkias",
"given": "Constantine",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National and Kapodistrian University of Athens"
}
],
"family": "Poulakou",
"given": "Garyfalia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National and Kapodistrian University of Athens"
}
],
"family": "Tsolakou",
"given": "Annia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National and Kapodistrian University of Athens"
}
],
"family": "Xintaropoulos",
"given": "Panagiotis",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National School of Public Administration"
}
],
"family": "Raptis",
"given": "Athanasios",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National and Kapodistrian University of Athens"
}
],
"family": "Syrigos",
"given": "Nikolaos",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Metron Nutraceuticals"
}
],
"family": "Tsirikos-Karapanos",
"given": "Nikolaos",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National and Kapodistrian University of Athens"
}
],
"family": "Drakoulis",
"given": "Nikolaos",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
3,
8
]
],
"date-time": "2026-03-08T16:55:57Z",
"timestamp": 1772988957000
},
"deposited": {
"date-parts": [
[
2026,
3,
11
]
],
"date-time": "2026-03-11T12:20:51Z",
"timestamp": 1773231651000
},
"group-title": "In Review",
"indexed": {
"date-parts": [
[
2026,
3,
12
]
],
"date-time": "2026-03-12T04:01:47Z",
"timestamp": 1773288107590,
"version": "3.50.1"
},
"institution": [
{
"name": "Research Square"
}
],
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
8
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "unspecified",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
8
]
],
"date-time": "2026-03-08T00:00:00Z",
"timestamp": 1772928000000
}
}
],
"link": [
{
"URL": "https://www.researchsquare.com/article/rs-9009421/v1",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://www.researchsquare.com/article/rs-9009421/v1.html",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"posted": {
"date-parts": [
[
2026,
3,
8
]
]
},
"prefix": "10.21203",
"published": {
"date-parts": [
[
2026,
3,
8
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "ref1",
"unstructured": "Burden of disease (2026) https://www.who.int/teams/global-influenza-programme/surveillance-and-monitoring/burden-of-disease. Accessed 7 Jan"
}
],
"reference-count": 1,
"references-count": 1,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.researchsquare.com/article/rs-9009421/v1"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"subtype": "preprint",
"title": "Effectiveness of a sustained-release ammonium chloride formulation in reducing the viral load of patients with COVID-19 or influenza: A prospective, randomized, double-blind, placebo-controlled study",
"type": "posted-content"
}