Assessment Of The Efficacy Of Spironolactone For COVID-19 ARDS Patients
et al., Aydin Sağlik Dergi̇si̇, doi:10.17932/IAU.ASD.2015.007/asd_v07i3002, Oct 2021
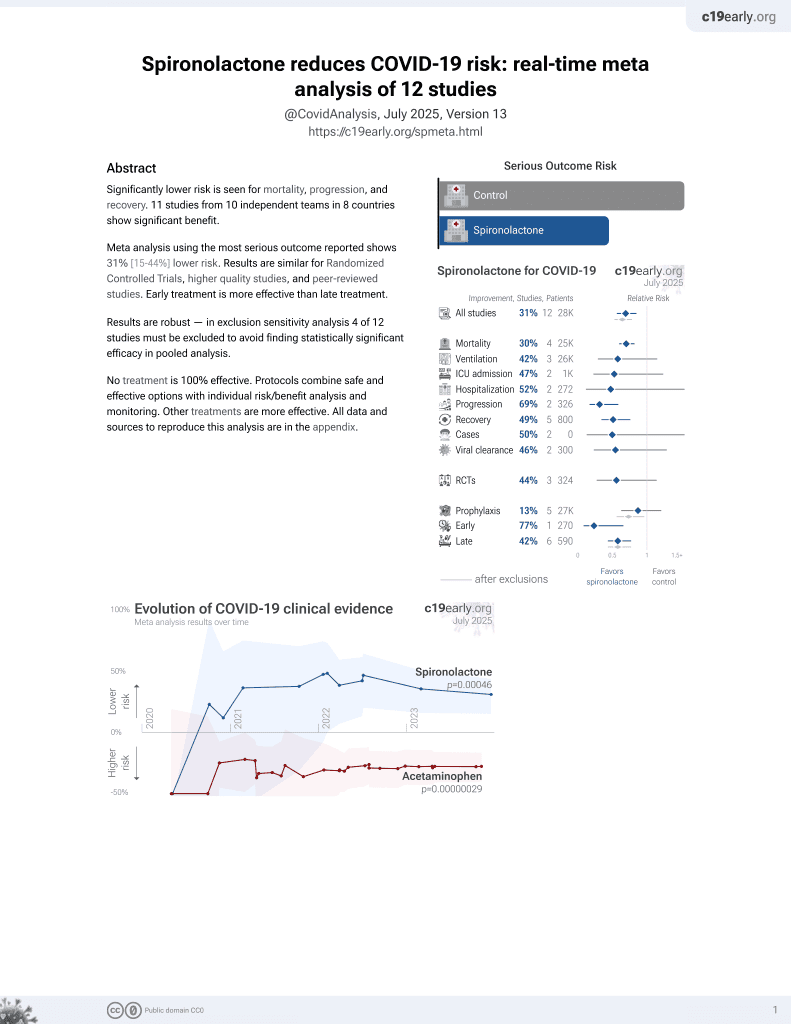
37th treatment shown to reduce risk in
February 2022, now with p = 0.00046 from 12 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Retrospective 30 COVID-19 ARDS ICU patients and 30 control patients, showing lower mortality with treatment.
|
risk of death, 46.2% lower, RR 0.54, p = 0.002, treatment 14 of 30 (46.7%), control 26 of 30 (86.7%), NNT 2.5.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Ersoy et al., 13 Oct 2021, retrospective, Turkey, peer-reviewed, 7 authors.
Covid19 ARDS olgularında spironolaktonun etkinliğinin değerlendirilmesi Assessment Of The Efficacy Of Spironolactone For COVID-19 ARDS Patients
AYDIN SAĞLIK DERGİSİ, doi:10.17932/iau.asd.2015.007/asd_v07i3002
The aim of our study is to compare patients monitored for ARDS diagnosis linked to COVID-19 treated with spironolactone with patients who did not use spironolactone and to retrospectively identify whether there was any positive effect on mortality using clinical and laboratory data from patients.
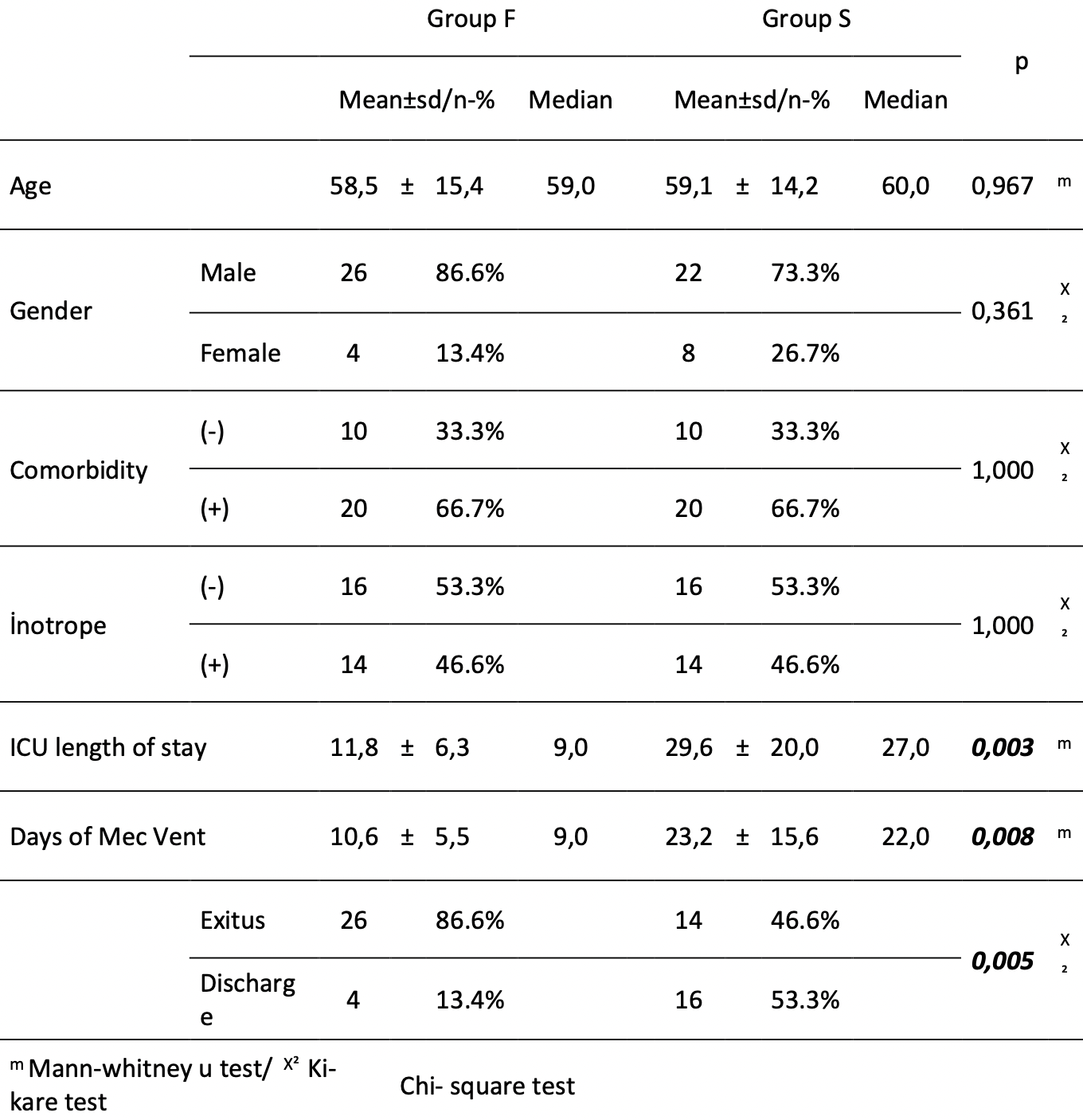
MATERIAL AND METHOD Two groups were created of patients treated due to severe ARDS diagnosis linked to COVID-19. One group administered spironolactone (Group S, n=30) and the other group was not (Group F, n=30). Groups were compared in terms of demographic characteristics, presence of comorbidity, inotropic agent requirements, (intensive care unit) ICU length of stay', days of mechanical ventilation and mortality. Additionally, fever, PO2/FIO2 (Horowitz ratio), lymphocyte count, CRP value, SOFA scorer, and d-dimer levels on the ICU length of stay, 1st day, 3rd day, 5th day, 8th day, 12th day, day of discharge or day of exitus were compared between the groups and statistically analyzed.
RESULTS Group S was found to have a higher ICU length of stay and days of mechanical ventilation compared to Group F. (p<0.05). Group F had significantly a higher mortality rate compared to Group S. (p<0.05). The final Horowitz value in Group F was found to be significantly lower compared to Group S. (p<0.05). The lymphocyte values in Group F were significantly lower than Group S on the 1st, 3rd, 5th, 8th and 12th day and at the final measurement. (p<0.05). The CRP values in Group F were significantly higher than Group S on the 3rd, 5th, 8th and 12th day and at the final measurement. (p<0.05). The SOFA scores on the 8th and 12th days and at the final measurement in Group F were found to be significantly higher than GroupS. (p<0.05).
CONCLUSION In our study, parameters for disease severity regressed, patients survived for longer and mortality was identified to be lower in the group using spironolactone.
References
Alhazzani, Møller, Arabi, Loeb, Gong et al., Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19), Intensive Care Med
Aronson, Ferner, Drugs and the renin-angiotensin system in covid-19, BMJ, doi:10.1136/bmj.m1313
Busse, Chow, Mccurdy, Khanna, COVID-19 and the RAAS-a potential role for angiotensin II?, Crit Care
Butler, Anstrom, Felker, Givertz, Kalogeropoulos et al., Efficacy and Safety of Spironolactone in Acute Heart Failure: The ATHENA-HF Randomized Clinical Trial, JAMA Cardiol
Chung, Bernheim, Mei, CT imaging features of 2019 novel coronavirus (2019-nCoV), Radiology
Ferrario, Jessup, Chappell, Effect of angiotensinconverting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2, Circulation, doi:10.1161/CIRCULATIONAHA.104.5104611589734
Gattinoni, Chiumello, Caironi, Busana, Romitti et al., COVID-19 pneumonia: different respiratory treatment for different phenotypes?, Intensive Care Med
Guan, Ni, Hu, Liang, Ou et al., China Medical Treatment Expert Group for Covid-19. Clinical characteristics of coronavirus disease 2019 in China, N Engl J Med, doi:10.1056/NEJMoa2002032
He, Deng, Li, Coronavirus disease 2019 (COVID-19): what we know?, J Med Virol
Heneghan, Brassey, NSAIDs in acute respiratory infection
Hoffmann, Kleine-Weber, &schroeder, SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor, Cell
Liaduet, Szabo, Blocking mineralocorticoid receptor with spironolactone may have a wide range of therapeutic actions in severe COVID-19 disease, Critical Care, doi:10.1186/s13054-020-03055-6
Lu, Drug treatment options for the 2019-new coronavirus (2019-nCoV), Biosci Trends
Murthy, Gomersall, Fowler, Care for critically ill patients with COVID-19, JAMA
Sodhi, Wohlford-Lenane, Yamaguchi, Attenuation of pulmonary ACE2 activity impairs inactivation of des-Arg bradykinin/BKB1R axis and facilitates LPS-induced neutrophil infiltration, Am J Physiol Lung Cell Mol Physiol
South, Tomlinson, Edmonston, Hiremath, Sparks, Controversies of renin-angiotensin system inhibition during the COVID-19 pandemic, Nat Rev Nephrol
Stockman, Bellamy, Garner, SARS: systematic review of treatment effects, PLoSMed, doi:.10.1371/journal.pmed.003034316968120
Thille, Esteban, Fernández-Segoviano, Comparison of the Berlin definition for acute respiratory distress syndrome with autopsy, Am J Respir Crit Care Med
Wang, Dhindsa, Povysil, Zoghbi, Motelow et al., TMPRSS2 Transcriptional Inhibition as a Therapeutic Strategy for COVID-19, doi:10.20944/preprints202003.0360.v2
Xu, Xiaochun, Acute respiratory failure in COVID-19: is it "typical, ARDS? Critical Care, doi:10.1186/s13054-020-02911-9
Zhang, Penninger, Li, Zhong, Slutsky, Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target, Intensive Care Med, doi:10.1007/s00134-020-05985-9
Zheng, Hu, Li, Respiratory treatment procedures in patients with severe novel coronavirus infected pneumonia: an expert opinion, Chin J CritCare Intensive Care Med, doi:10.3877/cma.j.issn.2096-1537.2020.0004
Zhou, Yu, Du, a retrospective cohort study, doi:10.1016/S0140-6736(20)30566-3
DOI record:
{
"DOI": "10.17932/iau.asd.2015.007/asd_v07i3002",
"ISSN": [
"2149-5769",
"2717-6800"
],
"URL": "http://dx.doi.org/10.17932/IAU.ASD.2015.007/asd_v07i3002",
"author": [
{
"affiliation": [],
"family": "ERSOY",
"given": "Aysin",
"sequence": "first"
},
{
"affiliation": [],
"family": "GÜVEN",
"given": "Bülent Barış",
"sequence": "additional"
},
{
"affiliation": [],
"family": "ERTÜRK",
"given": "Tuna",
"sequence": "additional"
},
{
"affiliation": [],
"family": "YURTSEVEN",
"given": "Fulya",
"sequence": "additional"
},
{
"affiliation": [],
"family": "KARAMAN",
"given": "Zöhre",
"sequence": "additional"
},
{
"affiliation": [],
"family": "GÜNER",
"given": "Temel",
"sequence": "additional"
},
{
"affiliation": [],
"family": "KÖMPE",
"given": "Özge",
"sequence": "additional"
}
],
"container-title": "AYDIN SAĞLIK DERGİSİ",
"container-title-short": "Aydın sağlık derg.",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
11,
1
]
],
"date-time": "2021-11-01T10:40:10Z",
"timestamp": 1635763210000
},
"deposited": {
"date-parts": [
[
2021,
11,
1
]
],
"date-time": "2021-11-01T10:40:12Z",
"timestamp": 1635763212000
},
"indexed": {
"date-parts": [
[
2022,
4,
5
]
],
"date-time": "2022-04-05T14:23:38Z",
"timestamp": 1649168618974
},
"is-referenced-by-count": 2,
"issue": "3",
"issued": {
"date-parts": [
[
2015
]
]
},
"journal-issue": {
"issue": "3",
"published-online": {
"date-parts": [
[
2015
]
]
},
"published-print": {
"date-parts": [
[
2015
]
]
}
},
"member": "7633",
"original-title": [],
"page": "191-209",
"prefix": "10.17932",
"published": {
"date-parts": [
[
2015
]
]
},
"published-online": {
"date-parts": [
[
2015
]
]
},
"published-print": {
"date-parts": [
[
2015
]
]
},
"publisher": "Istanbul Aydin University",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://aydinsaglikdergisi.aydin.edu.tr/wp-content/uploads/2021/10/asd_v07i3002.pdf"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subtitle": [],
"title": "Covid19 ARDS olgularında spironolaktonun etkinliğinin değerlendirilmesi Assessment Of The Efficacy Of Spironolactone For COVID-19 ARDS Patients",
"type": "journal-article",
"volume": "7"
}
ersoy