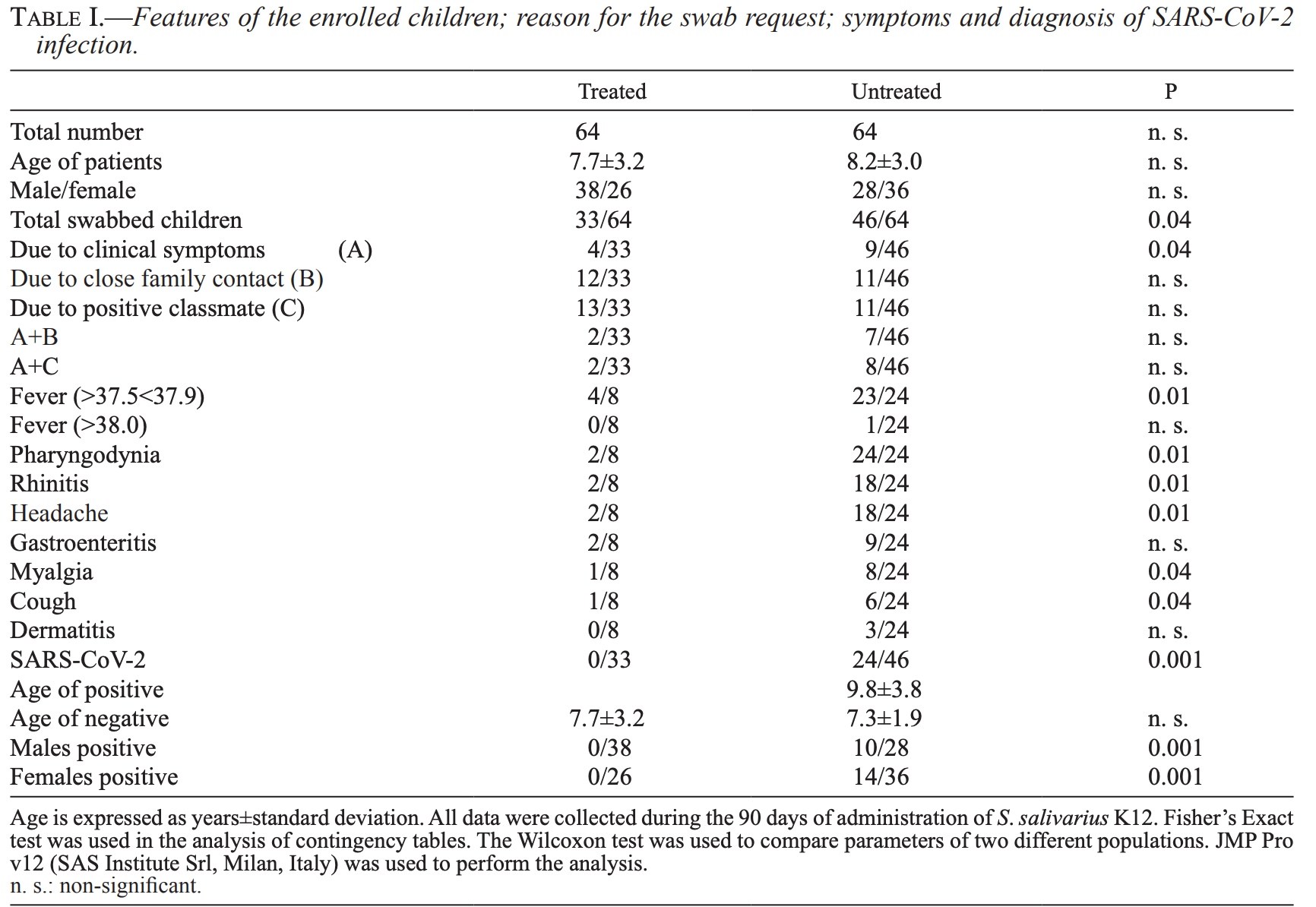
The administration of S. salivarius K12 to children may reduce the rate of SARS-CoV-2 infection
et al., Minerva Medica, doi:10.23736/S0026-4806.21.07487-5, Mar 2021
Probiotics for COVID-19
19th treatment shown to reduce risk in
March 2021, now with p = 0.00000044 from 29 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Interim report on an RCT for prophylactic treatment with S. salivarius K12, showing significantly lower cases with treatment. Only patients with symptoms or known positive contacts were tested. Trial identification/registration details are not provided.
Probiotic efficacy depends on the specific strains used. Specific microbes may decrease or increase COVID-19 risk1.
|
risk of case, 98.0% lower, RR 0.02, p < 0.001, treatment 0 of 64 (0.0%), control 24 of 64 (37.5%), NNT 2.7, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Di Pierro et al., 12 Mar 2021, Randomized Controlled Trial, Italy, peer-reviewed, 2 authors, study period September 2020 - December 2020.
Abstract: ©
This document is protected by international copyright laws. No additional reproduction is authorized. It is permitted for personal use to download and save only one file and print only one copy of this Article. It is not permitted to make additional copies (either sporadically
or systematically, either printed or electronic) of the Article for any purpose. It is not permitted to distribute the electronic copy of the article through online internet and/or intranet file sharing systems, electronic mailing or any other means which may allow access
to the Article. The use of all or any part of the Article for any Commercial Use is not permitted. The creation of derivative works from the Article is not permitted. The production of reprints for personal or commercial use is not permitted. It is not permitted to remove,
cover, overlay, obscure, block, or change any copyright notices or terms of use which the Publisher may post on the Article. It is not permitted to frame or use framing techniques to enclose any trademark, logo, or other proprietary information of the Publisher.
COPYRIGHT 2021 EDIZIONI MINERVA MEDICA
LETTERS TO THE EDITOR
© 2021 EDIZIONI MINERVA MEDICA
Online version at http://www.minervamedica.it
Minerva Medica 2021 August;112(4):514-6
DOI: 10.23736/S0026-4806.21.07487-5
The administration of S. salivarius K12
to children may reduce the rate
of SARS-CoV-2 infection
The Coronavirus disease 2019 (COVID-19) pandemic,
provoked by the worldwide spread of the severe acute
respiratory syndrome coronavirus 2 (SARS-CoV-2),
has caused millions of infections and deaths worldwide
since its emergence at the end of 2019. Unfortunately,
natural immunity does not exist within the human population and no effective drug has been found thus far
specifically for the disease. Incredibly, scientists from
all over the world have developed some effective vaccines at an unprecedented pace.1 Unfortunately, vaccine
manufacturing and distribution are two important bottlenecks. For this reason, vaccination schedules are proceeding more slowly than they should and was perhaps
expected. While waiting for the current vaccination
campaigns to produce the herd effect desired by each
individual country, COVID-19 research has produced
new lines of scientific thought. For instance, recent
evidence showed the possible relationship between the
lung and oral microbiotas. Indeed, analysis of the bronchoalveolar lavage fluid (BALF) of COVID-19 patients
revealed the presence of elevated levels of oral and upper respiratory commensal bacteria.2 Anatomical and
physiological considerations indicate that the oral cavity is the primary source of the lung microbiota community, acquired via aspiration and inhalation.3 Indeed, the
microbiota of the lungs overlaps with that found in the
mouth. In humans, the prominent taxa in BALF samples
include mainly Streptococcus, Prevotella and Veillonella, and these are indeed detected in concurrently collected oral samples. Recent studies have shown that the
microbiota in the lungs contributes to immunological
homeostasis and can potentially alter susceptibility to
viral infection.4 With respect to COVID-19, a particular
abundance of Prevotella and Veillonella spp. in the lung
microbiota composition has been observed in patients
with SARS-CoV-2 pneumonia.5 A report by Iebba et al.6
(not yet peer-reviewed) profiled the oral microbiota of
healthy controls and COVID-19-hospitalized patients,
discovering the existence of four..
DOI record:
{
"DOI": "10.23736/s0026-4806.21.07487-5",
"ISSN": [
"0026-4806",
"1827-1669"
],
"URL": "http://dx.doi.org/10.23736/S0026-4806.21.07487-5",
"author": [
{
"affiliation": [],
"family": "DI PIERRO",
"given": "Francesco",
"sequence": "first"
},
{
"affiliation": [],
"family": "COLOMBO",
"given": "Maria",
"sequence": "additional"
}
],
"container-title": "Minerva Medica",
"container-title-short": "Minerva Med",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
7,
16
]
],
"date-time": "2021-07-16T09:27:49Z",
"timestamp": 1626427669000
},
"deposited": {
"date-parts": [
[
2021,
7,
16
]
],
"date-time": "2021-07-16T09:29:31Z",
"timestamp": 1626427771000
},
"indexed": {
"date-parts": [
[
2024,
2,
7
]
],
"date-time": "2024-02-07T11:25:07Z",
"timestamp": 1707305107765
},
"is-referenced-by-count": 9,
"issue": "4",
"issued": {
"date-parts": [
[
2021,
7
]
]
},
"journal-issue": {
"issue": "4"
},
"link": [
{
"URL": "https://www.minervamedica.it/pdf.php?cod=R10Y2021N04A0514",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "17149",
"original-title": [],
"prefix": "10.23736",
"published": {
"date-parts": [
[
2021,
7
]
]
},
"published-print": {
"date-parts": [
[
2021,
7
]
]
},
"publisher": "Edizioni Minerva Medica",
"reference": [
{
"DOI": "10.1016/S1473-3099(20)30773-8",
"doi-asserted-by": "publisher",
"key": "10.23736/S0026-4806.21.07487-5_ref001"
},
{
"DOI": "10.1093/cid/ciaa203",
"doi-asserted-by": "publisher",
"key": "10.23736/S0026-4806.21.07487-5_ref002"
},
{
"DOI": "10.1007/s11427-017-9151-1",
"doi-asserted-by": "publisher",
"key": "10.23736/S0026-4806.21.07487-5_ref003"
},
{
"DOI": "10.1038/ncomms3106",
"doi-asserted-by": "publisher",
"key": "10.23736/S0026-4806.21.07487-5_ref004"
},
{
"DOI": "10.1093/bioinformatics/btaa285",
"doi-asserted-by": "publisher",
"key": "10.23736/S0026-4806.21.07487-5_ref005"
},
{
"DOI": "10.1101/2020.12.13.422589",
"doi-asserted-by": "publisher",
"key": "10.23736/S0026-4806.21.07487-5_ref006"
},
{
"DOI": "10.23736/S0026-4806.20.06570-2",
"doi-asserted-by": "publisher",
"key": "10.23736/S0026-4806.21.07487-5_ref007"
}
],
"reference-count": 7,
"references-count": 7,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.minervamedica.it/index2.php?show=R10Y2021N04A0514"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"General Medicine"
],
"subtitle": [],
"title": "The administration of S. salivarius K12 to children may reduce the rate of SARS-CoV-2 infection",
"type": "journal-article",
"volume": "112"
}
