Pharmacologic Ascorbic Acid as Early Therapy for Hospitalized Patients with COVID-19: A Randomized Clinical Trial
et al., Life, doi:10.3390/life12030453, Mar 2022
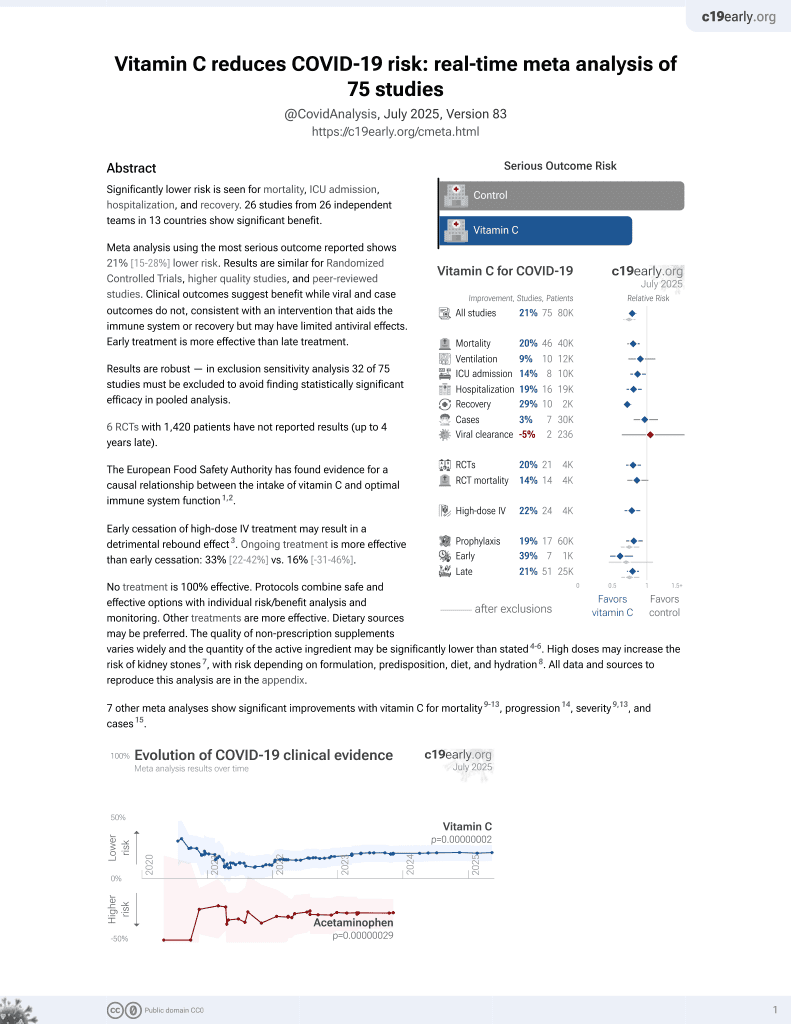
Vitamin C for COVID-19
6th treatment shown to reduce risk in
September 2020, now with p = 0.000000069 from 72 studies, recognized in 22 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT with 66 very late stage (8 days from symptom onset) hospitalized patients, 44 treated with vitamin C and 22 control patients, showing no significant differences with treatment.
This is the 11th of 20 COVID-19 RCTs for vitamin C, which collectively show efficacy with p=0.0016.
This is the 44th of 72 COVID-19 controlled studies for vitamin C, which collectively show efficacy with p=0.000000069.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of progression, 5.0% lower, HR 0.95, p = 0.64, treatment 4 of 44 (9.1%), control 2 of 22 (9.1%), adjusted per study, within 36 hours.
|
|
risk of no improvement, 49.7% better, RR 0.50, p = 0.16, treatment 6 of 44 (13.6%), control 6 of 22 (27.3%), NNT 7.3, adjusted per study, inverted to make RR<1 favor treatment, odds ratio converted to relative risk, within 36 hours.
|
|
risk of no hospital discharge, 22.5% lower, RR 0.78, p = 0.07, treatment 31 of 44 (70.5%), control 20 of 22 (90.9%), NNT 4.9, within 36 hours.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Coppock et al., 19 Mar 2022, Randomized Controlled Trial, USA, peer-reviewed, 14 authors, dosage 300mg/kg day 1, 600mg/kg day 2, 900mg/kg days 3-6.
Pharmacologic Ascorbic Acid as Early Therapy for Hospitalized Patients with COVID-19: A Randomized Clinical Trial
Life, doi:10.3390/life12030453
Despite the widespread availability of effective vaccines, new cases of infection with severe acute respiratory syndrome coronavirus-2, the cause of coronavirus disease 2019 (COVID-19), remain a concern in the settings of vaccine hesitancy and vaccine breakthrough. In this randomized, controlled, phase 2 trial, we hypothesized that high-dose ascorbic acid delivered intravenously to achieve pharmacologic concentrations may target the high viral phase of COVID-19 and thus improve early clinical outcomes. Sixty-six patients admitted with COVID-19 and requiring supplemental oxygen were randomized to receive either escalating doses of intravenous ascorbic acid plus standard of care or standard of care alone. The demographic and clinical characteristics were well-balanced between the two study arms. The primary outcome evaluated in this study was clinical improvement at 72 h after randomization. While the primary outcome was not achieved, point estimates for the composite outcome and its individual components of decreased use of supplemental oxygen, decreased use of bronchodilators, and the time to discharge were all favorable for the treatment arm. Possible favorable effects of ascorbic acid were most apparent during the first 72 h of hospitalization, although these effects disappeared over the course of the entire hospitalization. Future larger trials of intravenous ascorbic acid should be based on our current understanding of COVID-19 with a focus on the potential early benefits of ascorbic in hospitalized patients.
outcomes during the 72-h window after randomization, consistent with our hypotheses. Further consistent with our hypotheses, these trends were nullified as the curves crossed later in the hospitalization. As secondary endpoints, both the times to discharge and a 50% reduction in oxygenation were statistically non-significant when measured over the entire hospitalization. This pattern was not wholly unexpected. As noted above, an early benefit was consistent with our proposed mechanism of action. Regarding safety, the proportion of patients who experienced a clinical decline in the treatment arm did not meet stopping rules and the trial was continued to its full accrual. Though the point estimates were higher for serious adverse events and clinical decline in the first 36 h, the study was not powered to detect these differences, which were not statistically significant. Our trial had limitations. In retrospect, the study was small with optimistic outcome assumptions. Outcomes in this trial were defined in the early part of the pandemic, and do not reflect our current understanding of COVID-19 as a disease process. Furthermore, our patient risk stratification, while focused on age and comorbidities, may be improved in future studies. An emphasis on more severe underlying metabolic disease (i.e., diabetes), as opposed to any pulmonary or cardiovascular disease, may better improve patient selection and risk analyses. Viral load and specific analyses of lymphocyte populations were..
References
Agarwal, Hager, Sevransky, Any Role of High-Dose Vitamin C for Septic Shock in 2021?, Semin. Respir. Crit. Care Med, doi:10.1055/s-0041-1733986
Baj, Novazzi, Pasciuta, Genoni, Ferrante et al., Breakthrough Infections of E484K-Harboring SARS-CoV-2 Delta Variant, Lombardy, Italy, Emerg. Infect. Dis, doi:10.3201/eid2712.211792
Bergwerk, Gonen, Lustig, Amit, Lipsitch et al., COVID-19 Breakthrough Infections in Vaccinated Health Care Workers, N. Engl. J. Med, doi:10.1056/NEJMoa2109072
Biancatelli, Berrill, Marik, The antiviral properties of vitamin C. Expert, Rev. Anti-Infect. Ther, doi:10.1080/14787210.2020.1706483
Chang, Liao, Guan, Guo, Zhao et al., Combined Treatment With Hydrocortisone, Vitamin C, and Thiamine for Sepsis and Septic Shock: A Randomized Controlled Trial, Chest
Chen, Espey, Krishna, Mitchell, Corpe et al., Pharmacologic ascorbic acid concentrations selectively kill cancer cells: Action as a pro-drug to deliver hydrogen peroxide to tissues, Proc. Natl. Acad. Sci, doi:10.1073/pnas.0506390102
Chen, Espey, Sun, Lee, Krishna et al., Ascorbate in pharmacologic concentrations selectively generates ascorbate radical and hydrogen peroxide in extracellular fluid in vivo, Proc. Natl. Acad. Sci, doi:10.1073/pnas.0702854104
Chen, Espey, Sun, Pooput, Kirk et al., Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice, Proc. Natl. Acad. Sci, doi:10.1073/pnas.0804226105
Desai, Neyaz, Szabolcs, Shih, Chen et al., Temporal and spatial heterogeneity of host response to SARS-CoV-2 pulmonary infection, Nat. Commun, doi:10.1038/s41467-020-20139-7
Fowler, None
Fujii, Luethi, Young, Frei, Eastwood et al., Effect of Vitamin C, Hydrocortisone, and Thiamine vs Hydrocortisone Alone on Time Alive and Free of Vasopressor Support Among Patients With Septic Shock, JAMA, doi:10.1001/jama.2019.22176
Fujii, Salanti, Belletti, Bellomo, Carr et al., Effect of adjunctive vitamin C, glucocorticoids, and vitamin B1 on longer-term mortality in adults with sepsis or septic shock: A systematic review and a component network meta-analysis, Intensiv. Care Med, doi:10.1007/s00134-021-06558-0
Hacisuleyman, Hale, Saito, Blachere, Bergh et al., Vaccine Breakthrough Infections with SARS-CoV-2 Variants, N. Engl. J. Med, doi:10.1056/NEJMoa2105000
Harakeh, Jariwalla, Pauling, Suppression of human immunodeficiency virus replication by ascorbate in chronically and acutely infected cells, Proc. Natl. Acad. Sci, doi:10.1073/pnas.87.18.7245
Hoffer, Levine, Assouline, Melnychuk, Padayatty et al., Phase I clinical trial of i.v. ascorbic acid in advanced malignancy, Ann. Oncol, doi:10.1093/annonc/mdn377
Holmes, Veteran, Center, None
Hwang, Ryoo, Park, Jo, Jang et al., Combination therapy of vitamin C and thiamine for septic shock: A multi-centre, double-blinded randomized, controlled study, Intensiv. Care Med, doi:10.1007/s00134-020-06191-3
Iglesias, Vassallo, Patel, Sullivan, Cavanaugh et al., Outcomes of Metabolic Resuscitation Using Ascorbic Acid, Thiamine, and Glucocorticoids in the Early Treatment of Sepsis: The ORANGES Trial, Chest, doi:10.1016/j.chest.2020.02.049
Jeong, Hong, Kim, Jin, Kang et al., Vitamin C-treated murine bone marrow-derived dendritic cells preferentially drive naïve T cells into Th1 cells by increased IL-12 secretions, Cell. Immunol, doi:10.1016/j.cellimm.2010.10.005
Lahore, Role of Mega Dose of Vitamin C in Critical COVID-19 Patients
Levine, Violet, Data Triumph at C, Cancer Cell, doi:10.1016/j.ccell.2017.03.008
Marik, Khangoora, Rivera, Hooper, Catravas, Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock, Chest, doi:10.1016/j.chest.2016.11.036
Mccune, Toepp, Sheehan, Sherani, Petr et al., High dose intravenous vitamin C treatment in Sepsis: Associations with acute kidney injury and mortality, BMC Nephrol, doi:10.1186/s12882-021-02599-1
Menon, Mohamed, Prasannan, Moni, Edathadathil et al., Vitamin C Therapy for Routine Care in Septic Shock (ViCTOR) Trial: Effect of Intravenous Vitamin C, Thiamine, and Hydrocortisone Administration on Inpatient Mortality among Patients with Septic Shock, Indian J. Crit. Care Med, doi:10.5005/jp-journals-10071-23517
Moskowitz, Huang, Hou, Gong, Doshi et al., Effect of Ascorbic Acid, Corticosteroids, and Thiamine on Organ Injury in Septic Shock: The ACTS Randomized Clinical Trial, JAMA, doi:10.1001/jama.2020.11946
Natarajan, Brophy, Effect of Vitamin C Infusion on Organ Failure and Biomarkers of Inflammation and Vascular Injury in Patients with Sepsis and Severe Acute Respiratory Failure, JAMA, doi:10.1001/jama.2019.11825
Nauman, Gray, Parkinson, Levine, Paller, Systematic Review of Intravenous Ascorbate in Cancer Clinical Trials, Antioxidants, doi:10.3390/antiox7070089
Padayatty, Sun, Wang, Riordan, Hewitt et al., Vitamin C Pharmacokinetics: Implications for Oral and Intravenous Use, Ann. Intern. Med, doi:10.7326/0003-4819-140-7-200404060-00010
Research, Administration of Intravenous Vitamin C in Novel Coronavirus Infection (COVID-19) and Decreased Oxygenation
Reth, Hydrogen peroxide as second messenger in lymphocyte activation, Nat. Immunol, doi:10.1038/ni1202-1129
Rosengrave, Spencer, Williman, Mehrtens, Morgan et al., Intravenous vitamin C administration to patients with septic shock: A pilot randomised controlled trial, Crit. Care, doi:10.1186/s13054-022-03900-w
Schoenfeld, Sibenaller, Mapuskar, Wagner, Cramer-Morales et al., O2•− and H2O2-Mediated Disruption of Fe Metabolism Causes the Differential Susceptibility of NSCLC and GBM Cancer Cells to Pharmacological Ascorbate, Cancer Cell, doi:10.1016/j.ccell.2017.07.008
Sevransky, Rothman, Hager, Bernard, Brown et al., Effect of Vitamin C, Thiamine, and Hydrocortisone on Ventilator-and Vasopressor-Free Days in Patients With Sepsis: The VICTAS Randomized Clinical Trial, JAMA, doi:10.1001/jama.2020.24505
Siegel, Morton, Vitamin C and the immune response, Experientia, doi:10.1007/BF02002847
Solís Arce, Warren, Meriggi, Scacco, Mcmurry et al., COVID-19 vaccine acceptance and hesitancy in low-and middle-income countries, Nat. Med
Thomas, Patel, Bittel, Wolski, Wang et al., Effect of High-Dose Zinc and Ascorbic Acid Supplementation vs Usual Care on Symptom Length and Reduction among Ambulatory Patients with SARS-CoV-2 Infection: The COVID A to Z Randomized Clinical Trial, JAMA Netw. Open, doi:10.1001/jamanetworkopen.2021.0369
Truwit, Hite, Morris, Dewilde, Priday et al., None
Van Gorkom, Wolterink, Van Elssen, Wieten, Germeraad et al., Influence of Vitamin C on Lymphocytes: An Overview, Antioxidants, doi:10.3390/antiox7030041
Villamañán, Sobrino, Carpio, Moreno, Arancón et al., Inhaled bronchodilators use and clinical course of adult inpatients with Covid-19 pneumonia in Spain: A retrospective cohort study, Pulm. Pharmacol. Ther, doi:10.1016/j.pupt.2021.102007
Wani, Mufti, Jan, Shah, Qadri et al., Combination of vitamin C, thiamine and hydrocortisone added to standard treatment in the management of sepsis: Results from an open label randomised controlled clinical trial and a review of the literature, Infect. Dis, doi:10.1080/23744235.2020.1718200
DOI record:
{
"DOI": "10.3390/life12030453",
"ISSN": [
"2075-1729"
],
"URL": "http://dx.doi.org/10.3390/life12030453",
"abstract": "<jats:p>Despite the widespread availability of effective vaccines, new cases of infection with severe acute respiratory syndrome coronavirus-2, the cause of coronavirus disease 2019 (COVID-19), remain a concern in the settings of vaccine hesitancy and vaccine breakthrough. In this randomized, controlled, phase 2 trial, we hypothesized that high-dose ascorbic acid delivered intravenously to achieve pharmacologic concentrations may target the high viral phase of COVID-19 and thus improve early clinical outcomes. Sixty-six patients admitted with COVID-19 and requiring supplemental oxygen were randomized to receive either escalating doses of intravenous ascorbic acid plus standard of care or standard of care alone. The demographic and clinical characteristics were well-balanced between the two study arms. The primary outcome evaluated in this study was clinical improvement at 72 h after randomization. While the primary outcome was not achieved, point estimates for the composite outcome and its individual components of decreased use of supplemental oxygen, decreased use of bronchodilators, and the time to discharge were all favorable for the treatment arm. Possible favorable effects of ascorbic acid were most apparent during the first 72 h of hospitalization, although these effects disappeared over the course of the entire hospitalization. Future larger trials of intravenous ascorbic acid should be based on our current understanding of COVID-19 with a focus on the potential early benefits of ascorbic in hospitalized patients.</jats:p>",
"alternative-id": [
"life12030453"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-1212-1470",
"affiliation": [],
"authenticated-orcid": false,
"family": "Coppock",
"given": "Dagan",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0001-9550-3660",
"affiliation": [],
"authenticated-orcid": false,
"family": "Violet",
"given": "Pierre-Christian",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vasquez",
"given": "Gustavo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Belden",
"given": "Katherine",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-3675-8289",
"affiliation": [],
"authenticated-orcid": false,
"family": "Foster",
"given": "Michael",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mullin",
"given": "Bret",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Magee",
"given": "Devon",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5922-7953",
"affiliation": [],
"authenticated-orcid": false,
"family": "Mikell",
"given": "Isabelle",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shah",
"given": "Lokesh",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Powers",
"given": "Victoria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Curcio",
"given": "Brian",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Monti",
"given": "Daniel",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6297-4650",
"affiliation": [],
"authenticated-orcid": false,
"family": "Levine",
"given": "Mark",
"sequence": "additional"
}
],
"container-title": "Life",
"container-title-short": "Life",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
3,
21
]
],
"date-time": "2022-03-21T01:30:14Z",
"timestamp": 1647826214000
},
"deposited": {
"date-parts": [
[
2022,
9,
1
]
],
"date-time": "2022-09-01T06:44:48Z",
"timestamp": 1662014688000
},
"funder": [
{
"DOI": "10.13039/100019408",
"award": [
"NA"
],
"doi-asserted-by": "publisher",
"name": "Marcus Foundation"
},
{
"DOI": "10.13039/100000062",
"award": [
"DK053212-16"
],
"doi-asserted-by": "publisher",
"name": "National Institute of Diabetes and Digestive and Kidney Diseases"
}
],
"indexed": {
"date-parts": [
[
2024,
3,
25
]
],
"date-time": "2024-03-25T00:48:59Z",
"timestamp": 1711327739272
},
"is-referenced-by-count": 12,
"issue": "3",
"issued": {
"date-parts": [
[
2022,
3,
19
]
]
},
"journal-issue": {
"issue": "3",
"published-online": {
"date-parts": [
[
2022,
3
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
3,
19
]
],
"date-time": "2022-03-19T00:00:00Z",
"timestamp": 1647648000000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/2075-1729/12/3/453/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "453",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2022,
3,
19
]
]
},
"published-online": {
"date-parts": [
[
2022,
3,
19
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"article-title": "Estimated COVID-19 Burden",
"author": "Centers for Disease Control and Prevention",
"key": "ref1"
},
{
"DOI": "10.1038/s41591-021-01454-y",
"doi-asserted-by": "publisher",
"key": "ref2"
},
{
"DOI": "10.1056/NEJMoa2109072",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.1056/NEJMoa2105000",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.3201/eid2712.211792",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1038/s41467-020-20139-7",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1007/BF02002847",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.1016/j.cellimm.2010.10.005",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.1073/pnas.87.18.7245",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.3390/antiox7030041",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.1080/14787210.2020.1706483",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.1038/ni1202-1129",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.7326/0003-4819-140-7-200404060-00010",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.1073/pnas.0702854104",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.1073/pnas.0804226105",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1016/j.ccell.2017.03.008",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"article-title": "Adverse Events",
"author": "Centers for Disease Control and Prevention",
"key": "ref17"
},
{
"article-title": "High-Dose Vitamin C Treatment in Critically Ill COVID-19 Patients",
"author": "Sisli Hamidiye Etfal Training and Research Hospital",
"key": "ref18"
},
{
"article-title": "Role of Mega Dose of Vitamin C in Critical COVID-19 Patients",
"key": "ref19"
},
{
"article-title": "Hunter Holmes Mcguire Veteran Affairs Medical Center; Mcguire Research Institute, Administration of Intravenous Vitamin C in Novel Coronavirus Infection (COVID-19) and Decreased Oxygenation",
"key": "ref20"
},
{
"DOI": "10.1001/jamanetworkopen.2021.0369",
"doi-asserted-by": "publisher",
"key": "ref21"
},
{
"DOI": "10.1073/pnas.0506390102",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.1093/annonc/mdn377",
"doi-asserted-by": "publisher",
"key": "ref23"
},
{
"DOI": "10.3390/antiox7070089",
"doi-asserted-by": "publisher",
"key": "ref24"
},
{
"DOI": "10.1016/j.ccell.2017.07.008",
"doi-asserted-by": "publisher",
"key": "ref25"
},
{
"DOI": "10.1001/jama.2019.22176",
"doi-asserted-by": "publisher",
"key": "ref26"
},
{
"DOI": "10.1016/j.chest.2020.02.049",
"doi-asserted-by": "publisher",
"key": "ref27"
},
{
"DOI": "10.1080/23744235.2020.1718200",
"doi-asserted-by": "publisher",
"key": "ref28"
},
{
"DOI": "10.1016/j.chest.2020.02.065",
"doi-asserted-by": "publisher",
"key": "ref29"
},
{
"DOI": "10.1001/jama.2020.11946",
"doi-asserted-by": "publisher",
"key": "ref30"
},
{
"DOI": "10.1007/s00134-020-06191-3",
"doi-asserted-by": "publisher",
"key": "ref31"
},
{
"DOI": "10.5005/jp-journals-10071-23517",
"doi-asserted-by": "publisher",
"key": "ref32"
},
{
"DOI": "10.1001/jama.2020.24505",
"doi-asserted-by": "publisher",
"key": "ref33"
},
{
"DOI": "10.1001/jama.2019.11825",
"doi-asserted-by": "publisher",
"key": "ref34"
},
{
"DOI": "10.1007/s00134-021-06558-0",
"doi-asserted-by": "publisher",
"key": "ref35"
},
{
"DOI": "10.1055/s-0041-1733986",
"doi-asserted-by": "publisher",
"key": "ref36"
},
{
"DOI": "10.1186/s13054-022-03900-w",
"doi-asserted-by": "publisher",
"key": "ref37"
},
{
"DOI": "10.1016/j.chest.2016.11.036",
"doi-asserted-by": "publisher",
"key": "ref38"
},
{
"DOI": "10.1186/s12882-021-02599-1",
"doi-asserted-by": "publisher",
"key": "ref39"
},
{
"article-title": "National Hospital Care Survey",
"author": "Centers for Disease Control and Prevention",
"key": "ref40"
},
{
"article-title": "COVID-19: Developing Drugs and Biological Products for Treatment or Prevention Guidance for Industry",
"author": "United States Food and Drug Administration",
"key": "ref41"
},
{
"DOI": "10.1016/j.pupt.2021.102007",
"doi-asserted-by": "publisher",
"key": "ref42"
}
],
"reference-count": 42,
"references-count": 42,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/2075-1729/12/3/453"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Paleontology",
"Space and Planetary Science",
"General Biochemistry, Genetics and Molecular Biology",
"Ecology, Evolution, Behavior and Systematics"
],
"subtitle": [],
"title": "Pharmacologic Ascorbic Acid as Early Therapy for Hospitalized Patients with COVID-19: A Randomized Clinical Trial",
"type": "journal-article",
"volume": "12"
}