Baseline Drug Treatments as Indicators of Increased Risk of COVID-19 Mortality in Spain and Italy
et al., International Journal of Environmental Research and Public Health, doi:10.3390/ijerph182211786, Nov 2021
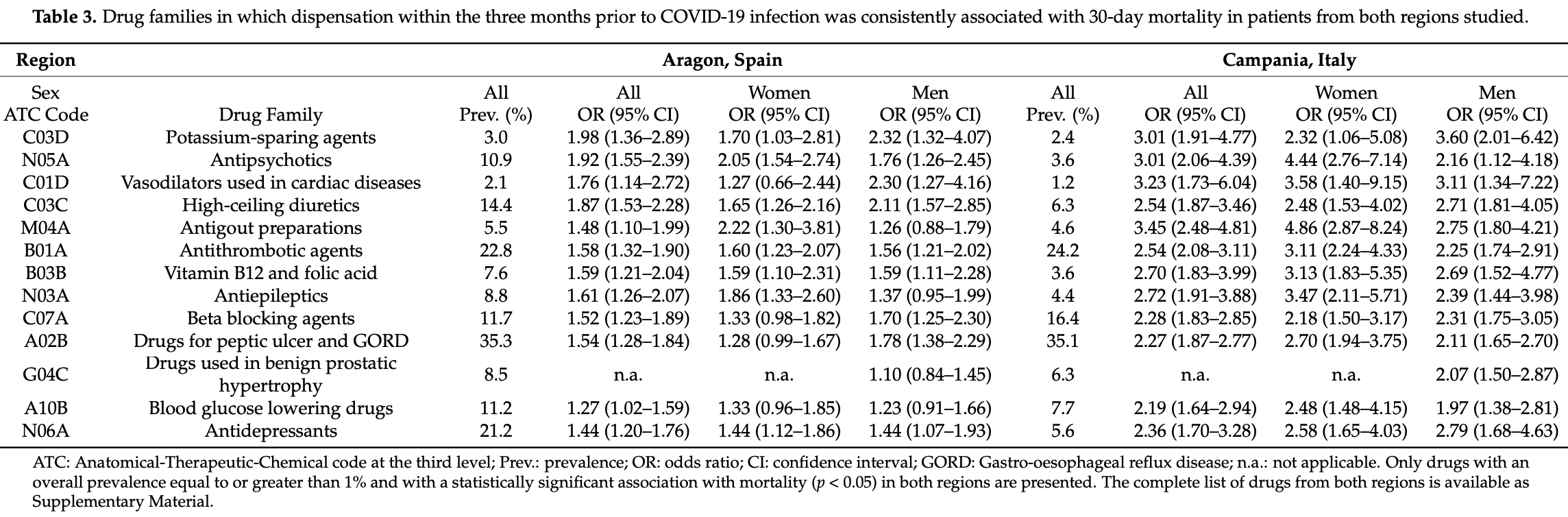
Retrospective 8,570 individuals in Spain and Italy, showing higher mortality with combined vitamin B9 and B12 supplementation. Adjustments only considered age.
|
risk of death, 87.4% higher, OR 1.87, p < 0.001, combined, RR approximated with OR.
|
|
risk of death, 170.0% higher, OR 2.70, p < 0.001, Campania, RR approximated with OR.
|
|
risk of death, 59.0% higher, OR 1.59, p < 0.001, Aragon, RR approximated with OR.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Bliek-Bueno et al., 10 Nov 2021, retrospective, multiple countries, peer-reviewed, mean age 67.7, 15 authors, study period 4 March, 2020 - 17 April, 2020, this trial uses multiple treatments in the treatment arm (combined with Vitamin B12) - results of individual treatments may vary.
Contact: sara.mucherino@unina.it (corresponding author), kbliek@salud.aragon.es, bpoblador.iacs@aragon.es, franciscagonzalezrubio@gmail.com, maza@salud.aragon.es, mclerencia@salud.aragon.es, ignatios.ioakeimskoufa@fhi.no, jcarmona@iisaragon.es, sprados.iacs@aragon.es, valentina.orlando@unina.it, enrica.menditto@unina.it, enrico.coscioni@regione.campania.it, alessandro.perrella@aocardarelli.it, ugo.trama@regione.campania.it, agimenomi.iacs@aragon.es.
Baseline Drug Treatments as Indicators of Increased Risk of COVID-19 Mortality in Spain and Italy
International Journal of Environmental Research and Public Health, doi:10.3390/ijerph182211786
This study aims to identify baseline medications that, as a proxy for the diseases they are dispensed for, are associated with increased risk of mortality in COVID-19 patients from two regions in Spain and Italy using real-world data. We conducted a cross-country, retrospective, observational study including 8570 individuals from both regions with confirmed SARS-CoV-2 infection between 4 March and 17 April 2020, and followed them for a minimum of 30 days to allow sufficient time for the studied event, in this case death, to occur. Baseline demographic variables and all drugs dispensed in community pharmacies three months prior to infection were extracted from the PRECOVID Study cohort (Aragon, Spain) and the Campania Region Database (Campania, Italy) and analyzed using logistic regression models. Results show that the presence at baseline of potassium-sparing agents, antipsychotics, vasodilators, high-ceiling diuretics, antithrombotic agents, vitamin B12, folic acid, and antiepileptics were systematically associated with mortality in COVID-19 patients from both countries. Treatments for chronic cardiovascular and metabolic diseases, systemic inflammation, and processes with increased risk of thrombosis as proxies for the conditions they are intended for can serve as timely indicators of an increased likelihood of mortality after the infection, and the assessment of pharmacological profiles can be an additional approach to the identification of at-risk individuals in clinical practice.
Supplementary Materials: The following are available online at https://www.mdpi.com/article/10 .3390/ijerph182211786/s1. Table S1 : Dispensation rates of the complete list of drugs studied and adjusted odds ratios of 30-day mortality in each region. Funding: This research was funded by the Government of Aragon through the Recognized Groups Grant reference B01_20R and Decree-Law 3/2020, published on 3 June.
Author Contributions:
Institutional Review Board Statement: The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Clinical Research Ethics Committee of Aragón (CEICA; Protocol number PI20/226). CEICA waived the requirement to obtain informed consent from Aragon participants due to the epidemiological nature of the project and the use of anonymized data that was presented at an aggregated level. The governance board of Unità del Farmaco della Regione Campania granted permission to the researchers of the CIRFF to use anonymized data for this study.
Conflicts of Interest: The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data, in the writing of the manuscript or in the decision to publish the results.
References
Bhatt, Jering, Vaduganathan, Claggett, Cunningham et al., Clinical Outcomes in Patients with Heart Failure Hospitalized with COVID-19, JACC Hear. Fail
Chen, Klein, Garibaldi, Li, Wu et al., Aging in COVID-19: Vulnerability, immunity and intervention, Ageing Res. Rev, doi:10.1016/j.arr.2020.101205
Choudhary, Sharma, Silakari, The interplay between inflammatory pathways and COVID-19: A critical review on pathogenesis and therapeutic options, Microb. Pathog, doi:10.1016/j.micpath.2020.104673
Corman, Landt, Kaiser, Molenkamp, Meijer et al., Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR, Eurosurveillance, doi:10.2807/1560-7917.ES.2020.25.3.2000045
De Alba, Orlando, Monetti, Mucherino, Gimeno-Miguel et al., Comorbidity in an Older Population with Type-2 Diabetes Mellitus: Identification of the Characteristics and Healthcare Utilization of High-Cost Patients, Front. Pharmacol, doi:10.3389/fphar.2020.586187
Du, Li, Wang, Shen, Ma et al., A Systematic Review and Meta-Analysis of Risk Factors Associated with Severity and Death in COVID-19 Patients, Can. J. Infect. Dis. Med Microbiol, doi:10.1155/2021/6660930
Ganguly, Alam, Role of homocysteine in the development of cardiovascular disease, Nutr. J, doi:10.1186/1475-2891-14-6
Gao, Ding, Dong, Zhang, Azkur et al., Risk factors for severe and critically ill COVID-19 patients: A review, Allergy, doi:10.1111/all.14657
Gülcü, Aksakal, Aydemir, Do Gan, Saraç et al., Association between previous anticoagulant use and mortality among hospitalized patients with COVID-19, J. Thromb. Thrombolysis
Iloanusi, Mgbere, Essien, Polypharmacy among COVID-19 patients: A systematic review, J. Am. Pharm. Assoc, doi:10.1016/j.japh.2021.05.006
Juste, Menditto, Orlando, Monetti, Miguel et al., Treatment Patterns of Diabetes in Italy: A Population-Based Study, Front. Pharmacol, doi:10.3389/fphar.2019.00870
Li, Zhong, Wang, Zeng, Luo et al., Clinical determinants of the severity of COVID-19: A systematic review and meta-analysis, PLoS ONE, doi:10.1371/journal.pone.0250602
Mcqueenie, Foster, Jani, Katikireddi, Sattar et al., Correction: Multimorbidity, polypharmacy, and COVID-19 infection within the UK Biobank cohort, PLoS ONE, doi:10.1371/journal.pone.0251613
Orlando, Coscioni, Guarino, Mucherino, Perrella et al., Drug-utilisation profiles and COVID-19, Sci. Rep, doi:10.1038/s41598-021-88398-y
Orlando, Monetti, Juste, Russo, Mucherino et al., Drug utilization pattern of antibiotics: The role of age, sex and municipalities in determining variation, Risk Manag. Healthc. Policy, doi:10.2147/RMHP.S223042
Orlando, Mucherino, Guarino, Guerriero, Trama et al., Gender Differences in Medication Use: A Drug Utilization Study Based on Real World Data, Int. J. Environ. Res. Public Health, doi:10.3390/ijerph17113926
Orlando, Rea, Savaré, Guarino, Mucherino et al., Development and validation of a clinical risk score to predict the risk of SARS-CoV-2 infection from administrative data: A population-based cohort study from Italy, PLoS ONE, doi:10.1371/journal.pone.0237202
Pijls, Jolani, Atherley, Derckx, Dijkstra et al., Demographic risk factors for COVID-19 infection, severity, ICU admission and death: A meta-analysis of 59 studies, BMJ Open, doi:10.1136/bmjopen-2020-044640
Poblador-Plou, Carmona-Pírez, Ioakeim-Skoufa, Poncel-Falcó, Bliek-Bueno et al., Baseline Chronic Comorbidity and Mortality in Laboratory-Confirmed COVID-19 Cases: Results from the PRECOVID Study in Spain, Int. J. Environ. Res. Public Health, doi:10.3390/ijerph17145171
Pratt, Kerr, Barratt, Kemp-Casey, Ellett et al., The validity of the Rx-Risk Comorbidity Index using medicines mapped to the Anatomical Therapeutic Chemical (ATC) Classification System, BMJ Open
Ren, Yu, Xu, Overton, Chiamvimonvat et al., Lack of association of antihypertensive drugs with the risk and severity of COVID-19: A meta-analysis, J. Cardiol, doi:10.1016/j.jjcc.2020.10.015
Yuan, Chen, Li, Chen, Wang et al., Mortality and pre-hospitalization use of low-dose aspirin in COVID-19 patients with coronary artery disease, J. Cell. Mol. Med, doi:10.1111/jcmm.16198
DOI record:
{
"DOI": "10.3390/ijerph182211786",
"ISSN": [
"1660-4601"
],
"URL": "http://dx.doi.org/10.3390/ijerph182211786",
"abstract": "<jats:p>This study aims to identify baseline medications that, as a proxy for the diseases they are dispensed for, are associated with increased risk of mortality in COVID-19 patients from two regions in Spain and Italy using real-world data. We conducted a cross-country, retrospective, observational study including 8570 individuals from both regions with confirmed SARS-CoV-2 infection between 4 March and 17 April 2020, and followed them for a minimum of 30 days to allow sufficient time for the studied event, in this case death, to occur. Baseline demographic variables and all drugs dispensed in community pharmacies three months prior to infection were extracted from the PRECOVID Study cohort (Aragon, Spain) and the Campania Region Database (Campania, Italy) and analyzed using logistic regression models. Results show that the presence at baseline of potassium-sparing agents, antipsychotics, vasodilators, high-ceiling diuretics, antithrombotic agents, vitamin B12, folic acid, and antiepileptics were systematically associated with mortality in COVID-19 patients from both countries. Treatments for chronic cardiovascular and metabolic diseases, systemic inflammation, and processes with increased risk of thrombosis as proxies for the conditions they are intended for can serve as timely indicators of an increased likelihood of mortality after the infection, and the assessment of pharmacological profiles can be an additional approach to the identification of at-risk individuals in clinical practice.</jats:p>",
"alternative-id": [
"ijerph182211786"
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-4812-4657",
"affiliation": [],
"authenticated-orcid": false,
"family": "Bliek-Bueno",
"given": "Kevin",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0003-3357-5655",
"affiliation": [],
"authenticated-orcid": false,
"family": "Mucherino",
"given": "Sara",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-5119-5093",
"affiliation": [],
"authenticated-orcid": false,
"family": "Poblador-Plou",
"given": "Beatriz",
"sequence": "additional"
},
{
"affiliation": [],
"family": "González-Rubio",
"given": "Francisca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aza-Pascual-Salcedo",
"given": "Mercedes",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-8209-8878",
"affiliation": [],
"authenticated-orcid": false,
"family": "Orlando",
"given": "Valentina",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Clerencia-Sierra",
"given": "Mercedes",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-6518-749X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Ioakeim-Skoufa",
"given": "Ignatios",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Coscioni",
"given": "Enrico",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carmona-Pírez",
"given": "Jonás",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-2812-8500",
"affiliation": [],
"authenticated-orcid": false,
"family": "Perrella",
"given": "Alessandro",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Trama",
"given": "Ugo",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-5704-6056",
"affiliation": [],
"authenticated-orcid": false,
"family": "Prados-Torres",
"given": "Alexandra",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Menditto",
"given": "Enrica",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-5440-1710",
"affiliation": [],
"authenticated-orcid": false,
"family": "Gimeno-Miguel",
"given": "Antonio",
"sequence": "additional"
}
],
"container-title": [
"International Journal of Environmental Research and Public Health"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2021,
11,
10
]
],
"date-time": "2021-11-10T14:19:21Z",
"timestamp": 1636553961000
},
"deposited": {
"date-parts": [
[
2021,
11,
10
]
],
"date-time": "2021-11-10T15:11:37Z",
"timestamp": 1636557097000
},
"funder": [
{
"DOI": "10.13039/501100010067",
"award": [
"Recognized Groups Grant reference B01_20R and Decree-Law 3/2020, published on 3 June"
],
"doi-asserted-by": "publisher",
"name": "Government of Aragon"
}
],
"indexed": {
"date-parts": [
[
2021,
12,
9
]
],
"date-time": "2021-12-09T15:57:11Z",
"timestamp": 1639065431319
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "electronic",
"value": "1660-4601"
}
],
"issue": "22",
"issued": {
"date-parts": [
[
2021,
11,
10
]
]
},
"journal-issue": {
"issue": "22",
"published-online": {
"date-parts": [
[
2021,
11
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
11,
10
]
],
"date-time": "2021-11-10T00:00:00Z",
"timestamp": 1636502400000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1660-4601/18/22/11786/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "11786",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2021,
11,
10
]
]
},
"published-online": {
"date-parts": [
[
2021,
11,
10
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"key": "ref1",
"unstructured": "Coronavirus Disease (COVID-19) Weekly Epidemiological Update and Weekly Operational Update\nhttps://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports"
},
{
"DOI": "10.3390/ijerph17145171",
"doi-asserted-by": "publisher",
"key": "ref2"
},
{
"DOI": "10.1371/journal.pone.0250602",
"doi-asserted-by": "publisher",
"key": "ref3"
},
{
"DOI": "10.1155/2021/6660930",
"doi-asserted-by": "publisher",
"key": "ref4"
},
{
"DOI": "10.1016/j.micpath.2020.104673",
"doi-asserted-by": "publisher",
"key": "ref5"
},
{
"DOI": "10.1016/j.japh.2021.05.006",
"doi-asserted-by": "publisher",
"key": "ref6"
},
{
"DOI": "10.1038/s41598-021-88398-y",
"doi-asserted-by": "publisher",
"key": "ref7"
},
{
"DOI": "10.1371/journal.pone.0237202",
"doi-asserted-by": "publisher",
"key": "ref8"
},
{
"DOI": "10.3389/fphar.2020.586187",
"doi-asserted-by": "publisher",
"key": "ref9"
},
{
"DOI": "10.1136/bmjopen-2017-021122",
"doi-asserted-by": "publisher",
"key": "ref10"
},
{
"DOI": "10.3389/fphar.2019.00870",
"doi-asserted-by": "publisher",
"key": "ref11"
},
{
"DOI": "10.3390/ijerph17113926",
"doi-asserted-by": "publisher",
"key": "ref12"
},
{
"DOI": "10.2147/RMHP.S223042",
"doi-asserted-by": "publisher",
"key": "ref13"
},
{
"DOI": "10.2807/1560-7917.ES.2020.25.3.2000045",
"doi-asserted-by": "publisher",
"key": "ref14"
},
{
"DOI": "10.1136/bmjopen-2020-044640",
"doi-asserted-by": "publisher",
"key": "ref15"
},
{
"DOI": "10.1016/j.arr.2020.101205",
"doi-asserted-by": "publisher",
"key": "ref16"
},
{
"DOI": "10.1111/all.14657",
"doi-asserted-by": "publisher",
"key": "ref17"
},
{
"DOI": "10.1016/j.jjcc.2020.10.015",
"doi-asserted-by": "publisher",
"key": "ref18"
},
{
"DOI": "10.1007/s11239-021-02489-1",
"doi-asserted-by": "publisher",
"key": "ref19"
},
{
"DOI": "10.1111/jcmm.16198",
"doi-asserted-by": "publisher",
"key": "ref20"
},
{
"DOI": "10.1371/journal.pone.0251613",
"doi-asserted-by": "publisher",
"key": "ref21"
},
{
"DOI": "10.1016/j.jchf.2020.11.003",
"doi-asserted-by": "publisher",
"key": "ref22"
},
{
"DOI": "10.1186/1475-2891-14-6",
"doi-asserted-by": "publisher",
"key": "ref23"
}
],
"reference-count": 23,
"references-count": 23,
"relation": {},
"score": 1,
"short-container-title": [
"IJERPH"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Health, Toxicology and Mutagenesis",
"Public Health, Environmental and Occupational Health"
],
"subtitle": [],
"title": [
"Baseline Drug Treatments as Indicators of Increased Risk of COVID-19 Mortality in Spain and Italy"
],
"type": "journal-article",
"volume": "18"
}
bliekbueno