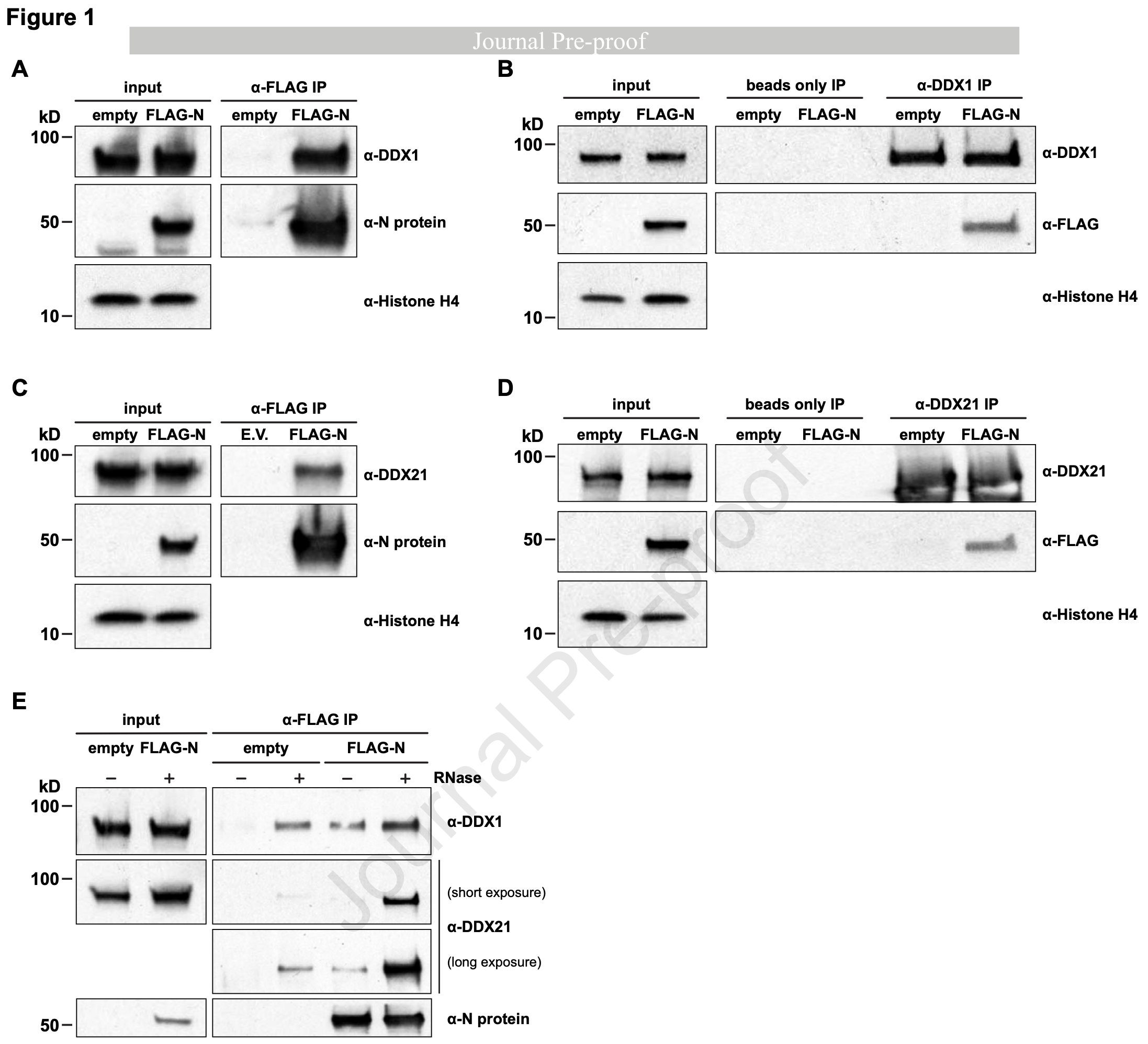
Direct interaction between human DDX1 and SARS-CoV-2 Nucleocapsid protein is regulated by phosphorylation
et al., Journal of Biological Chemistry, doi:10.1016/j.jbc.2026.111408, Mar 2026
In vitro study showing that the SARS-CoV-2 nucleocapsid (N) protein interacts with human RNA helicases DDX1 and DDX21 through mechanistically distinct pathways, with DDX1 binding being phosphorylation-dependent and DDX21 binding being phosphorylation-independent.
Wang et al., 26 Mar 2026, USA, peer-reviewed, 5 authors.
Contact: azu3jn@virginia.edu.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Direct interaction between human DDX1 and SARS-CoV-2 Nucleocapsid protein is regulated by phosphorylation
Journal of Biological Chemistry, doi:10.1016/j.jbc.2026.111408
The nucleocapsid (N) protein of SARS-CoV-2 is essential for viral replication and transcription, in part through interactions with host proteins. Here, we delineate distinct mechanisms underlying N protein association with human RNA helicases DDX1 and DDX21. Co-immunoprecipitation assays in HEK293 cells modified to express N protein revealed that DDX1 binding requires the N protein serine-arginine (SR) region, as SR deletion markedly reduced interaction. Inhibition of glycogen synthase kinase-3 (GSK-3), which targets the SR region, serine-to-alanine substitutions within the SR region, and alkaline phosphatase treatment of extract, respectively, demonstrated that phosphorylation of the SR region is critical for DDX1 binding. Furthermore, phosphorylated or phospho-mimetic SR peptides both prevented N protein-DDX1 complex formation and disrupted preformed complexes in vitro, whereas unphosphorylated peptides had no effect, confirming a phosphorylation-dependent binding mechanism. In contrast, interaction with DDX21 was unaffected by SR deletion or phosphorylation status and required both the N-and C-terminal domains of the N protein. RNase treatment enhanced N-DDX21 association without altering N-DDX1 interactions, indicating distinct regulation by RNA. Domain mapping of the two helicases identified the DDX1 N-terminal and the DDX21 C-terminal domains as interfaces that bind the N protein. Together, these findings support phosphorylation-dependent recruitment of DDX1 versus phosphorylation-independent engagement of DDX21, highlighting mechanistically distinct strategies by which SARS-CoV-2 N co-opts host helicases.
SUPPORTING INFORMATION This article contains supporting information.
AUTHOR CONTRIBUTIONS
Conceptualization
References
Abbasian, Mahmanzar, Rahimian, Mahdavi, Tokhanbigli et al., Global landscape of SARS-CoV-2 mutations and conserved regions, J Transl Med
Adly, Bi, Carlson, Syed, Ciling et al., Assembly of SARS-CoV-2 ribonucleosomes by truncated, N J Biol Chem
Alexopoulou, Couchman, Whiteford, The CMV early enhancer/chicken beta actin (CAG) promoter can be used to drive transgene expression during the differentiation of murine embryonic stem cells into vascular progenitors, BMC Cell Biol
Ariumi, Host Cellular RNA Helicases Regulate SARS-CoV-2 Infection, J Virol
Bilbrough, Piemontese, Seitz, Dissecting the role of protein phosphorylation: a chemical biology toolbox, Chem Soc Rev
Botova, Camacho-Zarco, Tognetti, Bessa, Guseva et al., A specific phosphorylation-dependent conformational switch in SARS-CoV-2 nucleocapsid protein inhibits RNA binding, Sci Adv
Bouhaddou, Memon, Meyer, White, Rezelj et al., The Global Phosphorylation Landscape of SARS-CoV-2, Infection Cell
Cao, Wang, Huang, Tan, The Role of SARS-CoV-2 Nucleocapsid Protein in Host Inflammation, Viruses
Carlson, Adly, Bi, Howard, Frost et al., Reconstitution of the SARS-CoV-2 ribonucleosome provides insights into genomic RNA packaging and regulation by phosphorylation, J Biol Chem
Carlson, Asfaha, Ghent, Howard, Hartooni et al., Phosphoregulation of Phase Separation by the SARS-CoV-2 N Protein Suggests a Biophysical Basis for its Dual Functions, Mol Cell
Chang, Hou, Chang, Hsiao, Huang, The SARS coronavirus nucleocapsid protein--forms and functions, Antiviral Res
Cubuk, Alston, Incicco, Singh, Stuchell-Brereton et al., The SARS-CoV-2 nucleocapsid protein is dynamic, disordered, and phase separates with RNA, Nat Commun
Dou, Lin, Wang, Wang, Jia et al., The CAG promoter maintains high-level transgene expression in HEK293 cells, FEBS Open Bio
Emmott, Munday, Bickerton, Britton, Rodgers et al., The cellular interactome of the coronavirus infectious bronchitis virus nucleocapsid protein and functional implications for virus biology, J Virol
Favetta, Wang, Cubuk, Singh, Barai et al., Phosphorylation toggles the SARS-CoV-2 nucleocapsid protein between two membraneassociated condensate states, Nat Commun
Gordon, Jang, Bouhaddou, Xu, Obernier et al., A SARS-CoV-2 protein interaction map reveals targets for drug repurposing, Nature
Heidersbach, Dorighi, Gomez, Jacobi, Haley, A versatile, high-efficiency platform for CRISPR-based gene activation, Nat Commun
Jack, Ferro, Trnka, Wehri, Nadgir et al., SARS-CoV-2 nucleocapsid protein forms condensates with viral genomic RNA, PLoS Biol
Johnson, Zhou, Lokugamage, Vu, Bopp et al., Nucleocapsid mutations in SARS-CoV-2 augment replication and pathogenesis, PLoS Pathog
Kang, Yang, Hong, Zhang, Huang et al., Crystal structure of SARS-CoV-2 nucleocapsid protein RNA binding domain reveals potential unique drug targeting sites, Acta Pharm Sin B
Katiyar, Arduini, Li, Liang, SARS-CoV-2 Assembly: Gaining Infectivity and Beyond, Viruses
Kozeleková, Náplavová, Brom, Gašparik, Šimek et al., Phosphorylated and Phosphomimicking Variants May Differ-A Case Study of 14-3-3, Protein Front Chem
Lichty, Malecki, Agnew, Michelson-Horowitz, Tan, Comparison of affinity tags for protein purification, Protein Expr Purif
Liu, Bai, Zhang, Gao, Liu et al., SARS-CoV-2 N Protein Antagonizes Stress Granule Assembly and IFN Production by Interacting with G3BPs to Facilitate Viral Replication, J Virol
Lodola, Secchi, Sinigiani, De Palma, Rossi et al., Interaction of SARS-CoV-2 Nucleocapsid Protein and Human RNA Helicases DDX1 and DDX3X Modulates Their Activities on Double-Stranded RNA, Int J Mol Sci
Lokugamage, Zhou, Alvarado, Plante, Ahearn et al., Convergent evolution in nucleocapsid facilitated SARS-CoV-2 adaptation for human infection, J Virol
Loonen, Van Steenis, Bauer, Šoštarić, Phosphorylation Changes SARS-CoV-2 Nucleocapsid Protein's Structural Dynamics and Its Interaction With RNA, Proteins
Lu, Ye, Singh, Cao, Diedrich et al., The SARS-CoV-2 nucleocapsid phosphoprotein forms mutually exclusive condensates with RNA and the membrane-associated M protein, Nat Commun
Min, Huang, Feng, Jia, Sun et al., A New Cellular Interactome of SARS-CoV-2 Nucleocapsid Protein and Its Biological Implications, Mol Cell Proteomics
Ni, Han, Zhou, Zhou, Lei, Structural insights into ribonucleoprotein dissociation by nucleocapsid protein interacting with non-structural protein 3 in SARS-CoV-2, Commun Biol
Nikolakaki, Giannakouros, SR/RS Motifs as Critical Determinants of Coronavirus Life Cycle Front, Mol Biosci
Nishi, Hashimoto, Panchenko, Phosphorylation in protein-protein binding: effect on stability and function, Structure
Oh, Shin, SARS-CoV-2 Nucleocapsid Protein Targets RIG-I-Like Receptor Pathways to Inhibit the Induction of Interferon Response, Cells
Pan, Shen, Yu, Ge, Chen et al., SARS-CoV-2 N protein promotes NLRP3 inflammasome activation to induce hyperinflammation, Nat Commun
Peng, Lee, Tarn, Phosphorylation of the arginine/serine dipeptide-rich motif of the severe acute respiratory syndrome coronavirus nucleocapsid protein modulates its multimerization, translation inhibitory activity and cellular localization, FEBS J
Perdikari, Murthy, Ryan, Watters, Naik et al., SARS-CoV-2 nucleocapsid protein phase-separates with RNA and with human hnRNPs, EMBO J
Reardon, Stuwe, Shah, Yu, Hughes et al., Modifications to the SR-Rich Region of the SARS-CoV-2 Nucleocapsid Regulate Self-Association and Attenuate RNA Interactions bioRxiv
Ribeiro-Filho, Jara, Batista, Schleder, Costa Tonoli et al., Structural dynamics of SARS-CoV-2 nucleocapsid protein induced by RNA binding, PLoS Comput Biol
Roden, Dai, Giannetti, Seim, Lee et al., Doublestranded RNA drives SARS-CoV-2 nucleocapsid protein to undergo phase separation at specific temperatures, Nucleic Acids Res
Savastano, Ibáñez De Opakua, Rankovic, Zweckstetter, Nucleocapsid protein of SARS-CoV-2 phase separates into RNA-rich polymerasecontaining condensates, Nat Commun
Shapira, Vimalanathan, Rens, Pichler, Peña-Díaz et al., Inhibition of glycogen synthase kinase-3-beta (GSK3β) blocks nucleocapsid phosphorylation and SARS-CoV-2 replication, Mol Biomed
Squeglia, Romano, Ruggiero, Maga, Berisio, Host DDX Helicases as Possible SARS-CoV-2 Proviral Factors: A Structural Overview of Their Hijacking Through Multiple Viral, Proteins Front Chem
Stuwe, Reardon, Yu, Shah, Hughes et al., Phosphorylation in the Ser/Arg-rich region of the nucleocapsid of SARS-CoV-2 regulates phase separation by inhibiting self-association of a distant helix, J Biol Chem
Sullivan, Morse, Grabarkewitz, Bayachou, Rouzina et al., Mechanism of SARS-CoV-2 Nucleocapsid Protein Phosphorylation-induced Functional Switch bioRxiv
Surjit, Lal, The SARS-CoV nucleocapsid protein: a protein with multifarious activities, Infect Genet Evol
Syed, Ciling, Chen, Carlson, Adly et al., The cellular RNA helicase DDX1 interacts with coronavirus nonstructural protein 14 and enhances viral replication, J Virol
Tapescu, Cherry, DDX RNA helicases: key players in cellular homeostasis and innate antiviral immunity, J Virol
Terpe, Overview of tag protein fusions: from molecular and biochemical fundamentals to commercial systems, Appl Microbiol Biotechnol
Tugaeva, Hawkins, Smith, Bayfield, Ker et al., The Mechanism of SARS-CoV-2 Nucleocapsid Protein Recognition by the Human 14-3-3, Proteins J Mol Biol
Wang, Joshi, Miller, Higgins, Slattery et al., A Role for Monomethylation of Histone H3-K27 in Gene Activity in, Genetics
Wang, Valadez-Ingersoll, Gilmore, Control of nuclear localization of the nucleocapsid protein of SARS-CoV-2, Virology
Wang, Yang, Song, Li, Zhou et al., Current understanding of the role of DDX21 in orchestrating gene expression in health and diseases, Life Sci
Wu, Chen, Yeh, Nucleocapsid phosphorylation and RNA helicase DDX1 recruitment enables coronavirus transition from discontinuous to continuous transcription, Cell Host Microbe
Wu, Cheng, Zhou, Sun, Zhang, The SARS-CoV-2 nucleocapsid protein: its role in the viral life cycle, structure and functions, and use as a potential target in the development of vaccines and diagnostics, Virol J
Wu, Qavi, Hachim, Kavian, Cole et al., Characterization of SARS-CoV-2 nucleocapsid protein reveals multiple functional consequences of the C-terminal domain iScience
Wu, Xing, Meng, Fu, Xue et al., Nucleocapsid mutations R203K/G204R increase the infectivity, fitness, and virulence of SARS-CoV-2, Cell Host Microbe
Xia, Wang, Ying, Huang, Zhang et al., RAGE Is a Receptor for SARS-CoV-2 N Protein and Mediates N Protein-induced Acute Lung Injury, Am J Respir Cell Mol Biol
Xiao, Fan, Li, Hou, DDX21 at the Nexus of RNA Metabolism, Cancer Oncogenesis, and Host-Virus Crosstalk: Decoding Its Biomarker Potential and Therapeutic Implications, Int J Mol Sci
Xiao, Song, Zhou, Lu, Fang et al., Coronavirus nucleocapsid proteins: a multifaceted modulator in the innate immune evasion, Front Microbiol
Yaron, Heaton, Levy, Johnson, Jordan et al., Host protein kinases required for SARS-CoV-2 nucleocapsid phosphorylation and viral replication, Sci Signal
Ye, West, Silletti, Corbett, Architecture and self-assembly of the SARS-CoV-2 nucleocapsid protein, Protein Sci
Zhu, Zhang, Wang, Li, Yang et al., A Novel Coronavirus from Patients with Pneumonia in China, N Engl J Med
DOI record:
{
"DOI": "10.1016/j.jbc.2026.111408",
"ISSN": [
"0021-9258"
],
"URL": "http://dx.doi.org/10.1016/j.jbc.2026.111408",
"alternative-id": [
"S0021925826002784"
],
"article-number": "111408",
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Direct interaction between human DDX1 and SARS-CoV-2 Nucleocapsid protein is regulated by phosphorylation"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Journal of Biological Chemistry"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.jbc.2026.111408"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2026 THE AUTHORS. Published by Elsevier Inc on behalf of American Society for Biochemistry and Molecular Biology."
}
],
"author": [
{
"affiliation": [],
"family": "Wang",
"given": "Liangjun",
"sequence": "first"
},
{
"affiliation": [],
"family": "Baxley",
"given": "Ryan M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Largaespada",
"given": "David A.",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-7508-6230",
"affiliation": [],
"authenticated-orcid": false,
"family": "Aihara",
"given": "Hideki",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-1783-619X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Bielinsky",
"given": "Anja Katrin",
"sequence": "additional"
}
],
"container-title": "Journal of Biological Chemistry",
"container-title-short": "Journal of Biological Chemistry",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"jbc.org",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
26
]
],
"date-time": "2026-03-26T16:34:18Z",
"timestamp": 1774542858000
},
"deposited": {
"date-parts": [
[
2026,
3,
27
]
],
"date-time": "2026-03-27T03:41:57Z",
"timestamp": 1774582917000
},
"funder": [
{
"DOI": "10.13039/100000057",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/100000057",
"id-type": "DOI"
}
],
"name": "National Institute of General Medical Sciences"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
27
]
],
"date-time": "2026-03-27T05:34:52Z",
"timestamp": 1774589692035,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
1
]
],
"date-time": "2026-03-01T00:00:00Z",
"timestamp": 1772323200000
}
},
{
"URL": "https://www.elsevier.com/legal/tdmrep-license",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
1
]
],
"date-time": "2026-03-01T00:00:00Z",
"timestamp": 1772323200000
}
},
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 23,
"start": {
"date-parts": [
[
2026,
3,
24
]
],
"date-time": "2026-03-24T00:00:00Z",
"timestamp": 1774310400000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0021925826002784?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0021925826002784?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "111408",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2026,
3
]
]
},
"published-print": {
"date-parts": [
[
2026,
3
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1056/NEJMoa2001017",
"article-title": "A Novel Coronavirus from Patients with Pneumonia in China, 2019",
"author": "Zhu",
"doi-asserted-by": "crossref",
"first-page": "727",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jbc.2026.111408_bib1",
"volume": "382",
"year": "2020"
},
{
"article-title": "The SARS-CoV-2 nucleocapsid protein: its role in the viral life cycle, structure and functions, and use as a potential target in the development of vaccines and diagnostics",
"author": "Wu",
"issue": "6",
"journal-title": "Virol J",
"key": "10.1016/j.jbc.2026.111408_bib2",
"volume": "20",
"year": "2023"
},
{
"DOI": "10.1016/j.apsb.2020.04.009",
"article-title": "Crystal structure of SARS-CoV-2 nucleocapsid protein RNA binding domain reveals potential unique drug targeting sites",
"author": "Kang",
"doi-asserted-by": "crossref",
"first-page": "1228",
"journal-title": "Acta Pharm Sin B",
"key": "10.1016/j.jbc.2026.111408_bib3",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1016/j.antiviral.2013.12.009",
"article-title": "The SARS coronavirus nucleocapsid protein--forms and functions",
"author": "Chang",
"doi-asserted-by": "crossref",
"first-page": "39",
"journal-title": "Antiviral Res",
"key": "10.1016/j.jbc.2026.111408_bib4",
"volume": "103",
"year": "2014"
},
{
"DOI": "10.1016/j.meegid.2007.07.004",
"article-title": "The SARS-CoV nucleocapsid protein: a protein with multifarious activities",
"author": "Surjit",
"doi-asserted-by": "crossref",
"first-page": "397",
"journal-title": "Infect Genet Evol",
"key": "10.1016/j.jbc.2026.111408_bib5",
"volume": "8",
"year": "2008"
},
{
"DOI": "10.1002/pro.3909",
"article-title": "Architecture and self-assembly of the SARS-CoV-2 nucleocapsid protein",
"author": "Ye",
"doi-asserted-by": "crossref",
"first-page": "1890",
"journal-title": "Protein Sci",
"key": "10.1016/j.jbc.2026.111408_bib6",
"volume": "29",
"year": "2020"
},
{
"DOI": "10.1038/s41467-021-21953-3",
"doi-asserted-by": "crossref",
"key": "10.1016/j.jbc.2026.111408_bib7",
"unstructured": "Cubuk, J., Alston, J. J., Incicco, J. J., Singh, S., Stuchell-Brereton, M. D., Ward, M. D. et al. (2021) The SARS-CoV-2 nucleocapsid protein is dynamic, disordered, and phase separates with RNA Nat Commun 12, 1936"
},
{
"DOI": "10.1016/j.isci.2021.102681",
"article-title": "Characterization of SARS-CoV-2 nucleocapsid protein reveals multiple functional consequences of the C-terminal domain",
"author": "Wu",
"doi-asserted-by": "crossref",
"journal-title": "iScience",
"key": "10.1016/j.jbc.2026.111408_bib8",
"volume": "24",
"year": "2021"
},
{
"DOI": "10.1111/j.1742-4658.2008.06564.x",
"article-title": "Phosphorylation of the arginine/serine dipeptide-rich motif of the severe acute respiratory syndrome coronavirus nucleocapsid protein modulates its multimerization, translation inhibitory activity and cellular localization",
"author": "Peng",
"doi-asserted-by": "crossref",
"first-page": "4152",
"journal-title": "FEBS J",
"key": "10.1016/j.jbc.2026.111408_bib9",
"volume": "275",
"year": "2008"
},
{
"DOI": "10.1016/j.molcel.2020.11.025",
"article-title": "Phosphoregulation of Phase Separation by the SARS-CoV-2 N Protein Suggests a Biophysical Basis for its Dual Functions",
"author": "Carlson",
"doi-asserted-by": "crossref",
"first-page": "1092",
"journal-title": "Mol Cell",
"key": "10.1016/j.jbc.2026.111408_bib10",
"volume": "80",
"year": "2020"
},
{
"DOI": "10.1126/scisignal.abm0808",
"article-title": "Host protein kinases required for SARS-CoV-2 nucleocapsid phosphorylation and viral replication",
"author": "Yaron",
"doi-asserted-by": "crossref",
"first-page": "eabm0808",
"journal-title": "Sci Signal",
"key": "10.1016/j.jbc.2026.111408_bib11",
"volume": "15",
"year": "2022"
},
{
"DOI": "10.1002/prot.26842",
"article-title": "Phosphorylation Changes SARS-CoV-2 Nucleocapsid Protein's Structural Dynamics and Its Interaction With RNA",
"author": "Loonen",
"doi-asserted-by": "crossref",
"first-page": "1701",
"journal-title": "Proteins",
"key": "10.1016/j.jbc.2026.111408_bib12",
"volume": "93",
"year": "2025"
},
{
"DOI": "10.1371/journal.pbio.3001425",
"article-title": "SARS-CoV-2 nucleocapsid protein forms condensates with viral genomic RNA",
"author": "Jack",
"doi-asserted-by": "crossref",
"journal-title": "PLoS Biol",
"key": "10.1016/j.jbc.2026.111408_bib13",
"volume": "19",
"year": "2021"
},
{
"DOI": "10.3390/v16111648",
"article-title": "SARS-CoV-2 Assembly: Gaining Infectivity and Beyond",
"author": "Katiyar",
"doi-asserted-by": "crossref",
"journal-title": "Viruses",
"key": "10.1016/j.jbc.2026.111408_bib14",
"volume": "16",
"year": "2024"
},
{
"DOI": "10.1038/s41467-020-19843-1",
"article-title": "Nucleocapsid protein of SARS-CoV-2 phase separates into RNA-rich polymerase-containing condensates",
"author": "Savastano",
"doi-asserted-by": "crossref",
"first-page": "6041",
"journal-title": "Nat Commun",
"key": "10.1016/j.jbc.2026.111408_bib15",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.15252/embj.2020106478",
"article-title": "SARS-CoV-2 nucleocapsid protein phase-separates with RNA and with human hnRNPs",
"author": "Perdikari",
"doi-asserted-by": "crossref",
"journal-title": "EMBO J",
"key": "10.1016/j.jbc.2026.111408_bib16",
"volume": "39",
"year": "2020"
},
{
"DOI": "10.1016/j.jbc.2022.102560",
"article-title": "Reconstitution of the SARS-CoV-2 ribonucleosome provides insights into genomic RNA packaging and regulation by phosphorylation",
"author": "Carlson",
"doi-asserted-by": "crossref",
"journal-title": "J Biol Chem",
"key": "10.1016/j.jbc.2026.111408_bib17",
"volume": "298",
"year": "2022"
},
{
"DOI": "10.1016/j.jbc.2024.107354",
"article-title": "Phosphorylation in the Ser/Arg-rich region of the nucleocapsid of SARS-CoV-2 regulates phase separation by inhibiting self-association of a distant helix",
"author": "Stuwe",
"doi-asserted-by": "crossref",
"journal-title": "J Biol Chem",
"key": "10.1016/j.jbc.2026.111408_bib18",
"volume": "300",
"year": "2024"
},
{
"DOI": "10.3389/fmolb.2020.00219",
"article-title": "SR/RS Motifs as Critical Determinants of Coronavirus Life Cycle",
"author": "Nikolakaki",
"doi-asserted-by": "crossref",
"first-page": "219",
"journal-title": "Front Mol Biosci",
"key": "10.1016/j.jbc.2026.111408_bib19",
"volume": "7",
"year": "2020"
},
{
"DOI": "10.1016/j.jmb.2021.166875",
"article-title": "The Mechanism of SARS-CoV-2 Nucleocapsid Protein Recognition by the Human 14-3-3 Proteins",
"author": "Tugaeva",
"doi-asserted-by": "crossref",
"journal-title": "J Mol Biol",
"key": "10.1016/j.jbc.2026.111408_bib20",
"volume": "433",
"year": "2021"
},
{
"DOI": "10.1038/s41467-025-62922-4",
"article-title": "Phosphorylation toggles the SARS-CoV-2 nucleocapsid protein between two membrane-associated condensate states",
"author": "Favetta",
"doi-asserted-by": "crossref",
"first-page": "7970",
"journal-title": "Nat Commun",
"key": "10.1016/j.jbc.2026.111408_bib21",
"volume": "16",
"year": "2025"
},
{
"DOI": "10.1128/jvi.02091-24",
"article-title": "Convergent evolution in nucleocapsid facilitated SARS-CoV-2 adaptation for human infection",
"author": "Lokugamage",
"doi-asserted-by": "crossref",
"journal-title": "J Virol",
"key": "10.1016/j.jbc.2026.111408_bib22",
"volume": "99",
"year": "2025"
},
{
"DOI": "10.1371/journal.ppat.1012741",
"article-title": "SARS-CoV-2 evolution balances conflicting roles of N protein phosphorylation",
"author": "Syed",
"doi-asserted-by": "crossref",
"journal-title": "PLoS Pathog",
"key": "10.1016/j.jbc.2026.111408_bib23",
"volume": "20",
"year": "2024"
},
{
"DOI": "10.1128/JVI.00392-10",
"article-title": "The cellular RNA helicase DDX1 interacts with coronavirus nonstructural protein 14 and enhances viral replication",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "8571",
"journal-title": "J Virol",
"key": "10.1016/j.jbc.2026.111408_bib24",
"volume": "84",
"year": "2010"
},
{
"DOI": "10.3390/ijms24065784",
"article-title": "Interaction of SARS-CoV-2 Nucleocapsid Protein and Human RNA Helicases DDX1 and DDX3X Modulates Their Activities on Double-Stranded RNA",
"author": "Lodola",
"doi-asserted-by": "crossref",
"journal-title": "Int J Mol Sci",
"key": "10.1016/j.jbc.2026.111408_bib25",
"volume": "24",
"year": "2023"
},
{
"DOI": "10.1128/jvi.00002-22",
"article-title": "Host Cellular RNA Helicases Regulate SARS-CoV-2 Infection",
"author": "Ariumi",
"doi-asserted-by": "crossref",
"journal-title": "J Virol",
"key": "10.1016/j.jbc.2026.111408_bib26",
"volume": "96",
"year": "2022"
},
{
"DOI": "10.1128/jvi.00040-24",
"article-title": "DDX RNA helicases: key players in cellular homeostasis and innate antiviral immunity",
"author": "Tapescu",
"doi-asserted-by": "crossref",
"journal-title": "J Virol",
"key": "10.1016/j.jbc.2026.111408_bib27",
"volume": "98",
"year": "2024"
},
{
"DOI": "10.3389/fchem.2020.602162",
"article-title": "Host DDX Helicases as Possible SARS-CoV-2 Proviral Factors: A Structural Overview of Their Hijacking Through Multiple Viral Proteins",
"author": "Squeglia",
"doi-asserted-by": "crossref",
"journal-title": "Front Chem",
"key": "10.1016/j.jbc.2026.111408_bib28",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1128/JVI.00321-13",
"article-title": "The cellular interactome of the coronavirus infectious bronchitis virus nucleocapsid protein and functional implications for virus biology",
"author": "Emmott",
"doi-asserted-by": "crossref",
"first-page": "9486",
"journal-title": "J Virol",
"key": "10.1016/j.jbc.2026.111408_bib29",
"volume": "87",
"year": "2013"
},
{
"DOI": "10.1038/s41586-020-2286-9",
"article-title": "A SARS-CoV-2 protein interaction map reveals targets for drug repurposing",
"author": "Gordon",
"doi-asserted-by": "crossref",
"first-page": "459",
"journal-title": "Nature",
"key": "10.1016/j.jbc.2026.111408_bib30",
"volume": "583",
"year": "2020"
},
{
"DOI": "10.1016/j.mcpro.2023.100579",
"article-title": "A New Cellular Interactome of SARS-CoV-2 Nucleocapsid Protein and Its Biological Implications",
"author": "Min",
"doi-asserted-by": "crossref",
"journal-title": "Mol Cell Proteomics",
"key": "10.1016/j.jbc.2026.111408_bib31",
"volume": "22",
"year": "2023"
},
{
"DOI": "10.1016/j.chom.2014.09.009",
"article-title": "Nucleocapsid phosphorylation and RNA helicase DDX1 recruitment enables coronavirus transition from discontinuous to continuous transcription",
"author": "Wu",
"doi-asserted-by": "crossref",
"first-page": "462",
"journal-title": "Cell Host Microbe",
"key": "10.1016/j.jbc.2026.111408_bib32",
"volume": "16",
"year": "2014"
},
{
"article-title": "Modifications to the SR-Rich Region of the SARS-CoV-2 Nucleocapsid Regulate Self-Association and Attenuate RNA Interactions",
"author": "Reardon",
"journal-title": "bioRxiv",
"key": "10.1016/j.jbc.2026.111408_bib33",
"year": "2023"
},
{
"DOI": "10.1016/j.jbc.2023.105362",
"article-title": "Assembly of SARS-CoV-2 ribonucleosomes by truncated N",
"author": "Adly",
"doi-asserted-by": "crossref",
"journal-title": "J Biol Chem",
"key": "10.1016/j.jbc.2026.111408_bib34",
"volume": "299",
"year": "2023"
},
{
"DOI": "10.3389/fchem.2022.835733",
"article-title": "Phosphorylated and Phosphomimicking Variants May Differ-A Case Study of 14-3-3 Protein",
"author": "Kozeleková",
"doi-asserted-by": "crossref",
"journal-title": "Front Chem",
"key": "10.1016/j.jbc.2026.111408_bib35",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1126/sciadv.aax2323",
"article-title": "A specific phosphorylation-dependent conformational switch in SARS-CoV-2 nucleocapsid protein inhibits RNA binding",
"author": "Botova",
"doi-asserted-by": "crossref",
"journal-title": "Sci Adv",
"key": "10.1016/j.jbc.2026.111408_bib36",
"volume": "10",
"year": "2024"
},
{
"DOI": "10.1039/D1CS00991E",
"article-title": "Dissecting the role of protein phosphorylation: a chemical biology toolbox",
"author": "Bilbrough",
"doi-asserted-by": "crossref",
"first-page": "5691",
"journal-title": "Chem Soc Rev",
"key": "10.1016/j.jbc.2026.111408_bib37",
"volume": "51",
"year": "2022"
},
{
"DOI": "10.1016/j.str.2011.09.021",
"article-title": "Phosphorylation in protein-protein binding: effect on stability and function",
"author": "Nishi",
"doi-asserted-by": "crossref",
"first-page": "1807",
"journal-title": "Structure",
"key": "10.1016/j.jbc.2026.111408_bib38",
"volume": "19",
"year": "2011"
},
{
"article-title": "Mechanism of SARS-CoV-2 Nucleocapsid Protein Phosphorylation-induced Functional Switch",
"author": "Sullivan",
"journal-title": "bioRxiv",
"key": "10.1016/j.jbc.2026.111408_bib39",
"year": "2025"
},
{
"DOI": "10.1016/j.pep.2005.01.019",
"article-title": "Comparison of affinity tags for protein purification",
"author": "Lichty",
"doi-asserted-by": "crossref",
"first-page": "98",
"journal-title": "Protein Expr Purif",
"key": "10.1016/j.jbc.2026.111408_bib40",
"volume": "41",
"year": "2005"
},
{
"DOI": "10.1007/s00253-002-1158-6",
"article-title": "Overview of tag protein fusions: from molecular and biochemical fundamentals to commercial systems",
"author": "Terpe",
"doi-asserted-by": "crossref",
"first-page": "523",
"journal-title": "Appl Microbiol Biotechnol",
"key": "10.1016/j.jbc.2026.111408_bib41",
"volume": "60",
"year": "2003"
},
{
"article-title": "The CMV early enhancer/chicken beta actin (CAG) promoter can be used to drive transgene expression during the differentiation of murine embryonic stem cells into vascular progenitors",
"author": "Alexopoulou",
"issue": "2",
"journal-title": "BMC Cell Biol",
"key": "10.1016/j.jbc.2026.111408_bib42",
"volume": "9",
"year": "2008"
},
{
"DOI": "10.1038/s41467-023-36452-w",
"article-title": "A versatile, high-efficiency platform for CRISPR-based gene activation",
"author": "Heidersbach",
"doi-asserted-by": "crossref",
"first-page": "902",
"journal-title": "Nat Commun",
"key": "10.1016/j.jbc.2026.111408_bib43",
"volume": "14",
"year": "2023"
},
{
"DOI": "10.1002/2211-5463.13029",
"article-title": "The CAG promoter maintains high-level transgene expression in HEK293 cells",
"author": "Dou",
"doi-asserted-by": "crossref",
"first-page": "95",
"journal-title": "FEBS Open Bio",
"key": "10.1016/j.jbc.2026.111408_bib44",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1016/j.cell.2020.06.034",
"article-title": "The Global Phosphorylation Landscape of SARS-CoV-2 Infection",
"author": "Bouhaddou",
"doi-asserted-by": "crossref",
"first-page": "685",
"journal-title": "Cell",
"key": "10.1016/j.jbc.2026.111408_bib45",
"volume": "182",
"year": "2020"
},
{
"DOI": "10.1186/s12967-023-03996-w",
"article-title": "Global landscape of SARS-CoV-2 mutations and conserved regions",
"author": "Abbasian",
"doi-asserted-by": "crossref",
"first-page": "152",
"journal-title": "J Transl Med",
"key": "10.1016/j.jbc.2026.111408_bib46",
"volume": "21",
"year": "2023"
},
{
"DOI": "10.1016/j.chom.2021.11.005",
"article-title": "Nucleocapsid mutations R203K/G204R increase the infectivity, fitness, and virulence of SARS-CoV-2",
"author": "Wu",
"doi-asserted-by": "crossref",
"first-page": "1788",
"journal-title": "Cell Host Microbe",
"key": "10.1016/j.jbc.2026.111408_bib47",
"volume": "29",
"year": "2021"
},
{
"DOI": "10.1371/journal.ppat.1010627",
"article-title": "Nucleocapsid mutations in SARS-CoV-2 augment replication and pathogenesis",
"author": "Johnson",
"doi-asserted-by": "crossref",
"journal-title": "PLoS Pathog",
"key": "10.1016/j.jbc.2026.111408_bib48",
"volume": "18",
"year": "2022"
},
{
"DOI": "10.1016/j.lfs.2024.122716",
"article-title": "Current understanding of the role of DDX21 in orchestrating gene expression in health and diseases",
"author": "Wang",
"doi-asserted-by": "crossref",
"journal-title": "Life Sci",
"key": "10.1016/j.jbc.2026.111408_bib49",
"volume": "349",
"year": "2024"
},
{
"DOI": "10.3390/ijms252413581",
"article-title": "DDX21 at the Nexus of RNA Metabolism, Cancer Oncogenesis, and Host-Virus Crosstalk: Decoding Its Biomarker Potential and Therapeutic Implications",
"author": "Xiao",
"doi-asserted-by": "crossref",
"journal-title": "Int J Mol Sci",
"key": "10.1016/j.jbc.2026.111408_bib50",
"volume": "25",
"year": "2024"
},
{
"DOI": "10.1371/journal.pcbi.1010121",
"article-title": "Structural dynamics of SARS-CoV-2 nucleocapsid protein induced by RNA binding",
"author": "Ribeiro-Filho",
"doi-asserted-by": "crossref",
"journal-title": "PLoS Comput Biol",
"key": "10.1016/j.jbc.2026.111408_bib51",
"volume": "18",
"year": "2022"
},
{
"DOI": "10.1093/nar/gkac596",
"article-title": "Double-stranded RNA drives SARS-CoV-2 nucleocapsid protein to undergo phase separation at specific temperatures",
"author": "Roden",
"doi-asserted-by": "crossref",
"first-page": "8168",
"journal-title": "Nucleic Acids Res",
"key": "10.1016/j.jbc.2026.111408_bib52",
"volume": "50",
"year": "2022"
},
{
"DOI": "10.1038/s42003-023-04570-2",
"article-title": "Structural insights into ribonucleoprotein dissociation by nucleocapsid protein interacting with non-structural protein 3 in SARS-CoV-2",
"author": "Ni",
"doi-asserted-by": "crossref",
"first-page": "193",
"journal-title": "Commun Biol",
"key": "10.1016/j.jbc.2026.111408_bib53",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.1128/jvi.00412-22",
"article-title": "SARS-CoV-2 N Protein Antagonizes Stress Granule Assembly and IFN Production by Interacting with G3BPs to Facilitate Viral Replication",
"author": "Liu",
"doi-asserted-by": "crossref",
"journal-title": "J Virol",
"key": "10.1016/j.jbc.2026.111408_bib54",
"volume": "96",
"year": "2022"
},
{
"DOI": "10.1016/j.virol.2024.110232",
"article-title": "Control of nuclear localization of the nucleocapsid protein of SARS-CoV-2",
"author": "Wang",
"doi-asserted-by": "crossref",
"journal-title": "Virology",
"key": "10.1016/j.jbc.2026.111408_bib55",
"volume": "600",
"year": "2024"
},
{
"DOI": "10.1038/s41467-020-20768-y",
"article-title": "The SARS-CoV-2 nucleocapsid phosphoprotein forms mutually exclusive condensates with RNA and the membrane-associated M protein",
"author": "Lu",
"doi-asserted-by": "crossref",
"first-page": "502",
"journal-title": "Nat Commun",
"key": "10.1016/j.jbc.2026.111408_bib56",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.3390/v17081046",
"article-title": "The Role of SARS-CoV-2 Nucleocapsid Protein in Host Inflammation",
"author": "Cao",
"doi-asserted-by": "crossref",
"journal-title": "Viruses",
"key": "10.1016/j.jbc.2026.111408_bib57",
"volume": "17",
"year": "2025"
},
{
"DOI": "10.3390/cells10030530",
"article-title": "SARS-CoV-2 Nucleocapsid Protein Targets RIG-I-Like Receptor Pathways to Inhibit the Induction of Interferon Response",
"author": "Oh",
"doi-asserted-by": "crossref",
"journal-title": "Cells",
"key": "10.1016/j.jbc.2026.111408_bib58",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.3389/fmicb.2025.1658339",
"article-title": "Coronavirus nucleocapsid proteins: a multifaceted modulator in the innate immune evasion",
"author": "Xiao",
"doi-asserted-by": "crossref",
"journal-title": "Front Microbiol",
"key": "10.1016/j.jbc.2026.111408_bib59",
"volume": "16",
"year": "2025"
},
{
"DOI": "10.1038/s41467-021-25015-6",
"article-title": "SARS-CoV-2 N protein promotes NLRP3 inflammasome activation to induce hyperinflammation",
"author": "Pan",
"doi-asserted-by": "crossref",
"first-page": "4664",
"journal-title": "Nat Commun",
"key": "10.1016/j.jbc.2026.111408_bib60",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1165/rcmb.2022-0351OC",
"article-title": "RAGE Is a Receptor for SARS-CoV-2 N Protein and Mediates N Protein-induced Acute Lung Injury",
"author": "Xia",
"doi-asserted-by": "crossref",
"first-page": "508",
"journal-title": "Am J Respir Cell Mol Biol",
"key": "10.1016/j.jbc.2026.111408_bib61",
"volume": "69",
"year": "2023"
},
{
"DOI": "10.1186/s43556-022-00111-1",
"article-title": "Inhibition of glycogen synthase kinase-3-beta (GSK3β) blocks nucleocapsid phosphorylation and SARS-CoV-2 replication",
"author": "Shapira",
"doi-asserted-by": "crossref",
"first-page": "43",
"journal-title": "Mol Biomed",
"key": "10.1016/j.jbc.2026.111408_bib62",
"volume": "3",
"year": "2022"
},
{
"DOI": "10.1534/genetics.117.300585",
"article-title": "A Role for Monomethylation of Histone H3-K27 in Gene Activity in",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "1023",
"journal-title": "Genetics",
"key": "10.1016/j.jbc.2026.111408_bib63",
"volume": "208",
"year": "2018"
}
],
"reference-count": 63,
"references-count": 63,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S0021925826002784"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Direct interaction between human DDX1 and SARS-CoV-2 Nucleocapsid protein is regulated by phosphorylation",
"type": "journal-article",
"update-policy": "https://doi.org/10.1016/elsevier_cm_policy"
}