Inosine pranobex was associated with a reduction in mortality in COVID-19 by over 22%, as reported in a retrospective study utilizing complete health data from the Czech national registry
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2026.108519, Mar 2026
Retrospective 67,841 inosine pranobex recipients matched 1:2 with 135,682 controls from the Czech national registry, showing significantly reduced COVID-19 mortality with treatment.
Negative control outcome analyses (non-COVID mortality, cardiovascular mortality, external-cause mortality) showed no benefit, suggesting the effect is not purely a healthy-user bias. Authors note that treatment was prescribed preferentially to higher-risk patients, yet recipients still showed lower mortality, which strengthens the apparent benefit but cannot fully exclude unmeasured confounders.
|
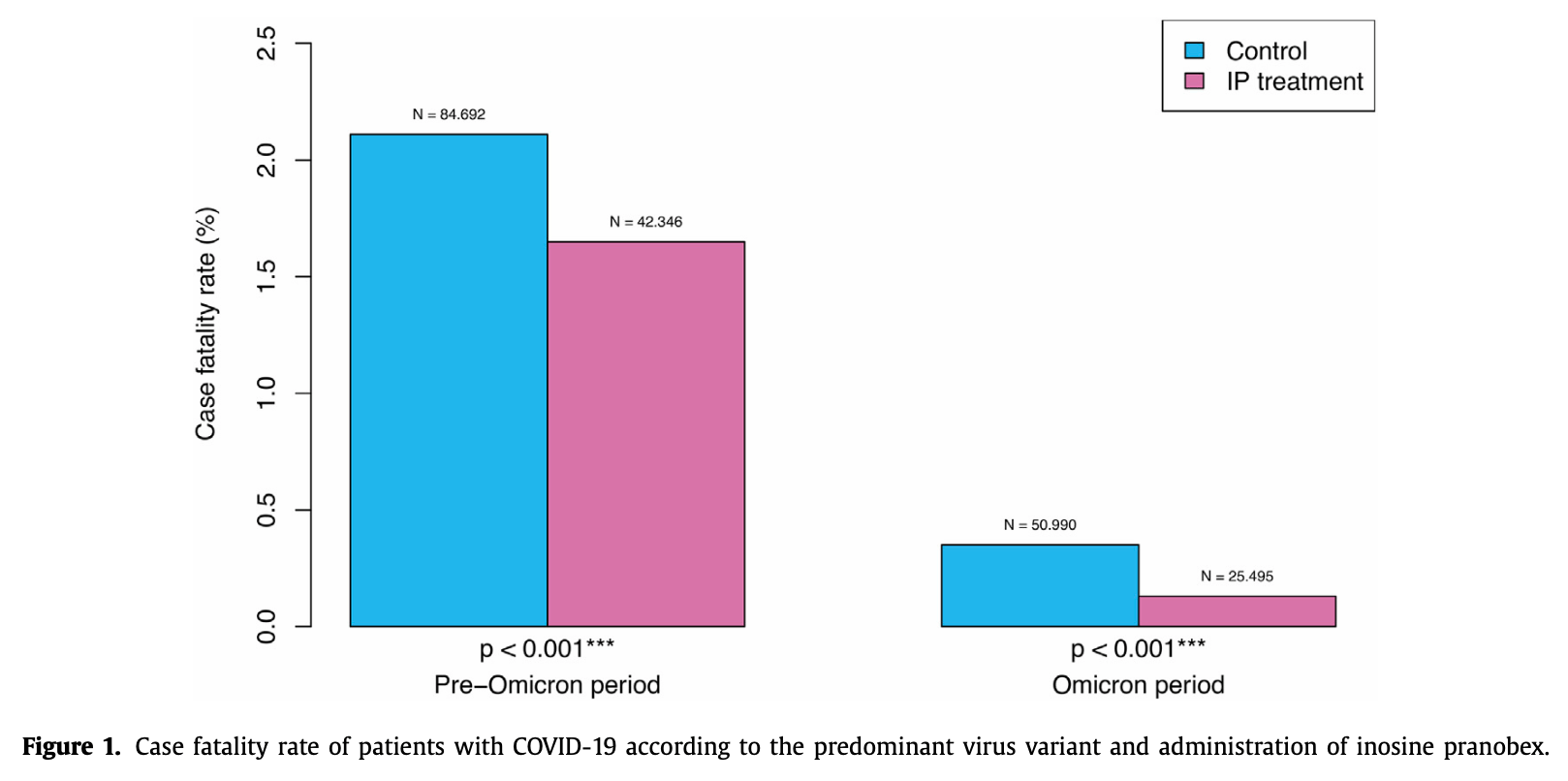
risk of death, 46.3% lower, RR 0.54, p = 0.11, treatment 42,346, control 84,692.
|
|
risk of death, 22.0% lower, RR 0.78, p < 0.001, treatment 697 of 42,346 (1.6%), control 1,788 of 84,692 (2.1%), NNT 215, pre-omicon.
|
|
risk of death, 65.0% lower, RR 0.35, p < 0.001, treatment 32 of 25,495 (0.1%), control 181 of 50,990 (0.4%), NNT 436, omicron.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Veselý et al., 31 Mar 2026, retrospective, Czech Republic, peer-reviewed, 5 authors, study period 1 March, 2020 - 31 December, 2021.
Contact: dalibor.vesely@lf3.cuni.cz, veselydalibor@seznam.cz.
Inosine pranobex is an orally administered small-molecule immunomodulator with broad antiviral activity. It enhances natural killer cell cytotoxicity and T-lymphocyte function and promotes Th1-type cytokine responses, thereby augmenting host antiviral defenses.
Abstract:
ARTICLE IN PRESS
[International Journal of Infectious Diseases xxx (xxxx) xxx](https://doi.org/10.1016/j.ijid.2026.108519)
Contents lists available at ScienceDirect
International Journal of Infectious Diseases
journal homepage:
[www.elsevier.com/locate/ijid](http://www.elsevier.com/locate/ijid)
Inosine pranobex was associated with a reduction in mortality in COVID-19 by over 22%, as reported in a retrospective study utilizing complete health data from the Czech national registry
Dalibor Veselý 1 , ∗ , Václav ˇ Capek 2 , Ondˇ rej Šanca 3 , Jiˇ rí Beran 4 , Jiˇ rí Slíva 1
- 1 Department of Pharmacology, Third Faculty of Medicine, Charles University in Prague, Prague, Czech Republic
- 2 Independent researcher
- 3 Institute of Health Information and Statistics of the Czech Republic, Prague, Czech Republic
- 4 Department for Tropical, Travel Medicine and Immunization, Institute for Postgraduate Medical Education, Prague, Czech Republic
a r t i c l e i n f o
Article history: Received 24 August 2025 Revised 17 February 2026 Accepted 28 February 2026
Available online xxx pranobex
Keywords: COVID-19 Pandemic Inosine Isoprinosine Mortality
Risk factor
DOI record:
{
"DOI": "10.1016/j.ijid.2026.108519",
"ISSN": [
"1201-9712"
],
"URL": "http://dx.doi.org/10.1016/j.ijid.2026.108519",
"alternative-id": [
"S1201971226001542"
],
"article-number": "108519",
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Inosine pranobex was associated with a reduction in mortality in COVID-19 by over 22%, as reported in a retrospective study utilizing complete health data from the Czech national registry"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "International Journal of Infectious Diseases"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.ijid.2026.108519"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2026 The Authors. Published by Elsevier Ltd on behalf of International Society for Infectious Diseases."
}
],
"author": [
{
"ORCID": "https://orcid.org/0009-0003-8069-2368",
"affiliation": [],
"authenticated-orcid": false,
"family": "Veselý",
"given": "Dalibor",
"sequence": "first"
},
{
"affiliation": [],
"family": "Čapek",
"given": "Václav",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Šanca",
"given": "Ondřej",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Beran",
"given": "Jiří",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-2690-1889",
"affiliation": [],
"authenticated-orcid": false,
"family": "Slíva",
"given": "Jiří",
"sequence": "additional"
}
],
"container-title": "International Journal of Infectious Diseases",
"container-title-short": "International Journal of Infectious Diseases",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.com",
"clinicalkey.com.au",
"clinicalkey.es",
"clinicalkey.fr",
"clinicalkey.jp",
"ijidonline.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
31
]
],
"date-time": "2026-03-31T12:06:47Z",
"timestamp": 1774958807000
},
"deposited": {
"date-parts": [
[
2026,
3,
31
]
],
"date-time": "2026-03-31T12:06:53Z",
"timestamp": 1774958813000
},
"funder": [
{
"DOI": "10.13039/100007397",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/100007397",
"id-type": "DOI"
}
],
"name": "Univerzita Karlova v Praze"
}
],
"indexed": {
"date-parts": [
[
2026,
4,
1
]
],
"date-time": "2026-04-01T05:30:34Z",
"timestamp": 1775021434016,
"version": "3.50.1"
},
"is-referenced-by-count": 1,
"issued": {
"date-parts": [
[
2026,
3
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
1
]
],
"date-time": "2026-03-01T00:00:00Z",
"timestamp": 1772323200000
}
},
{
"URL": "https://www.elsevier.com/legal/tdmrep-license",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
1
]
],
"date-time": "2026-03-01T00:00:00Z",
"timestamp": 1772323200000
}
},
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
28
]
],
"date-time": "2026-02-28T00:00:00Z",
"timestamp": 1772236800000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971226001542?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971226001542?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "108519",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2026,
3
]
]
},
"published-print": {
"date-parts": [
[
2026,
3
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.2165/00003495-198632050-00001",
"article-title": "Inosine pranobex",
"author": "Campoli-Richards",
"doi-asserted-by": "crossref",
"first-page": "383",
"journal-title": "Drugs",
"key": "10.1016/j.ijid.2026.108519_bib0001",
"volume": "32",
"year": "1986"
},
{
"DOI": "10.1007/s12325-019-00995-6",
"article-title": "Inosine pranobex: a key player in the game against a wide range of viral infections and non-infectious diseases",
"author": "Sliva",
"doi-asserted-by": "crossref",
"first-page": "1878",
"journal-title": "Adv Ther",
"key": "10.1016/j.ijid.2026.108519_bib0002",
"volume": "36",
"year": "2019"
},
{
"DOI": "10.3390/v13112246",
"article-title": "Inosine pranobex deserves attention as a potential immunomodulator to achieve early alteration of the COVID-19 disease course",
"author": "Beran",
"doi-asserted-by": "crossref",
"first-page": "2246",
"journal-title": "Viruses",
"key": "10.1016/j.ijid.2026.108519_bib0003",
"volume": "13",
"year": "2021"
},
{
"DOI": "10.1146/annurev-immunol-032712-100001",
"article-title": "Control of human viral infections by natural killer cells",
"author": "Jost",
"doi-asserted-by": "crossref",
"first-page": "163",
"journal-title": "Annu Rev Immunol",
"key": "10.1016/j.ijid.2026.108519_bib0004",
"volume": "31",
"year": "2013"
},
{
"DOI": "10.1016/j.intimp.2016.11.023",
"article-title": "Inosine acedoben dimepranol promotes an early and sustained increase in the natural killer cell component of circulating lymphocytes: a clinical trial supporting anti-viral indications",
"author": "Rumel Ahmed",
"doi-asserted-by": "crossref",
"first-page": "108",
"journal-title": "Int Immunopharmacol",
"key": "10.1016/j.ijid.2026.108519_bib0005",
"volume": "42",
"year": "2017"
},
{
"DOI": "10.1002/eji.201847948",
"article-title": "Inosine pranobex enhances human NK cell cytotoxicity by inducing metabolic activation and NKG2D ligand expression",
"author": "McCarthy",
"doi-asserted-by": "crossref",
"first-page": "130",
"journal-title": "Eur J Immunol",
"key": "10.1016/j.ijid.2026.108519_bib0006",
"volume": "50",
"year": "2020"
},
{
"DOI": "10.3390/pathogens9121055",
"article-title": "Inosine pranobex significantly decreased the case-fatality rate among PCR positive elderly with SARS-CoV-2 at three nursing homes in the Czech Republic",
"author": "Beran",
"doi-asserted-by": "crossref",
"first-page": "1055",
"journal-title": "Pathogens",
"key": "10.1016/j.ijid.2026.108519_bib0007",
"volume": "9",
"year": "2020"
},
{
"article-title": "Efficacy and safety of inosine pranobex in COVID-19 patients: a multicenter phase 3 randomized double-blind, placebo-controlled trial",
"author": "C",
"journal-title": "Adv Ther",
"key": "10.1016/j.ijid.2026.108519_bib0008",
"volume": "5",
"year": "2022"
},
{
"DOI": "10.1016/j.ijid.2024.01.017",
"article-title": "Case fatality rates of COVID-19 during epidemic periods of variants of concern: a meta-analysis by continents",
"author": "Xia",
"doi-asserted-by": "crossref",
"journal-title": "Int J Infect Dis",
"key": "10.1016/j.ijid.2026.108519_bib0009",
"volume": "141",
"year": "2024"
},
{
"DOI": "10.3389/fpubh.2022.884121",
"article-title": "Risk of SARS-CoV-2 reinfection 18 months after primary infection: population-level observational study",
"author": "Flacco",
"doi-asserted-by": "crossref",
"journal-title": "Front Public Health",
"key": "10.1016/j.ijid.2026.108519_bib0010",
"volume": "10",
"year": "2022"
},
{
"DOI": "10.1038/s41586-020-2918-0",
"article-title": "Age-specific mortality and immunity patterns of SARS-CoV-2",
"author": "O’Driscoll",
"doi-asserted-by": "crossref",
"first-page": "140",
"journal-title": "Nature",
"key": "10.1016/j.ijid.2026.108519_bib0011",
"volume": "590",
"year": "2021"
},
{
"DOI": "10.1136/bmj.m4509",
"article-title": "Infection fatality risk for SARS-CoV-2 in community dwelling population of Spain: nationwide seroepidemiological study",
"author": "Pastor-Barriuso",
"doi-asserted-by": "crossref",
"first-page": "m4509",
"journal-title": "BMJ",
"key": "10.1016/j.ijid.2026.108519_bib0012",
"volume": "371",
"year": "2020"
},
{
"DOI": "10.1016/j.ijid.2020.07.029",
"article-title": "Comorbidities and the risk of severe or fatal outcomes associated with coronavirus disease 2019: a systematic review and meta-analysis",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "47",
"journal-title": "Int J Infect Dis",
"key": "10.1016/j.ijid.2026.108519_bib0013",
"volume": "99",
"year": "2020"
},
{
"DOI": "10.3389/fmed.2021.588013",
"article-title": "Chronic diseases as a predictor for severity and mortality of COVID-19: a systematic review with cumulative meta-analysis",
"author": "Geng",
"doi-asserted-by": "crossref",
"journal-title": "Front Med",
"key": "10.1016/j.ijid.2026.108519_bib0014",
"volume": "8",
"year": "2021"
},
{
"key": "10.1016/j.ijid.2026.108519_bib0015",
"unstructured": "Šanca O, Jarkovský J, Klika P, Davidová H, Klimeš D, Mužík J, et al. Přehled vykázaných tříznakových diagnóz dle MKN-10. 2024. https://www.nzip.cz/clanek/1760-prehled-vykazane-triznakove-diagnozy-mkn-10-nrhzs-datovy-souhrn [accessed 30 April 2025]."
},
{
"DOI": "10.1093/eurheartj/ehaa433",
"article-title": "Association of hypertension and antihypertensive treatment with COVID-19 mortality: a retrospective observational study",
"author": "Gao",
"doi-asserted-by": "crossref",
"first-page": "2058",
"journal-title": "Eur Heart J",
"key": "10.1016/j.ijid.2026.108519_bib0016",
"volume": "41",
"year": "2020"
},
{
"DOI": "10.2337/dci21-0001",
"article-title": "Diabetes and COVID-19: population impact 18 months into the pandemic",
"author": "Gregg",
"doi-asserted-by": "crossref",
"first-page": "1916",
"journal-title": "Diabetes Care",
"key": "10.1016/j.ijid.2026.108519_bib0017",
"volume": "44",
"year": "2021"
},
{
"DOI": "10.1016/j.medcli.2020.12.017",
"article-title": "Coronary heart disease and COVID-19: a meta-analysis",
"author": "Liang",
"doi-asserted-by": "crossref",
"first-page": "547",
"journal-title": "Med Clín",
"key": "10.1016/j.ijid.2026.108519_bib0018",
"volume": "156",
"year": "2021"
},
{
"DOI": "10.1016/j.ajem.2020.07.009",
"article-title": "Effect of heart failure on the outcome of COVID-19 — a meta analysis and systematic review",
"author": "Yonas",
"doi-asserted-by": "crossref",
"first-page": "204",
"journal-title": "Am J Emerg Med",
"key": "10.1016/j.ijid.2026.108519_bib0019",
"volume": "46",
"year": "2021"
},
{
"DOI": "10.20892/j.issn.2095-3941.2020.0559",
"article-title": "Effects of cancer on patients with COVID-19: a systematic review and meta-analysis of 63,019 participants",
"author": "Yang",
"doi-asserted-by": "crossref",
"first-page": "298",
"journal-title": "Cancer Biol Med",
"key": "10.1016/j.ijid.2026.108519_bib0020",
"volume": "18",
"year": "2021"
},
{
"DOI": "10.1111/jce.14770",
"article-title": "Outcomes and mortality associated with atrial arrhythmias among patients hospitalized with COVID-19",
"author": "Peltzer",
"doi-asserted-by": "crossref",
"first-page": "3077",
"journal-title": "J Cardiovasc Electrophysiol",
"key": "10.1016/j.ijid.2026.108519_bib0021",
"volume": "31",
"year": "2020"
},
{
"DOI": "10.1016/j.jstrokecerebrovasdis.2020.104949",
"article-title": "Impact of cerebrovascular and cardiovascular diseases on mortality and severity of COVID-19–systematic review, meta-analysis, and meta-regression",
"author": "Pranata",
"doi-asserted-by": "crossref",
"journal-title": "J Stroke Cerebrovasc Dis",
"key": "10.1016/j.ijid.2026.108519_bib0022",
"volume": "29",
"year": "2020"
},
{
"DOI": "10.1038/s41598-023-36208-y",
"article-title": "Anemia as a risk factor for disease progression in patients admitted for COVID-19: data from a large, multicenter cohort study",
"author": "Veronese",
"doi-asserted-by": "crossref",
"first-page": "9035",
"journal-title": "Sci Rep",
"key": "10.1016/j.ijid.2026.108519_bib0023",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1097/MCP.0000000000000834",
"article-title": "Chronic obstructive pulmonary disease and COVID-19: interrelationships",
"author": "Singh",
"doi-asserted-by": "crossref",
"first-page": "76",
"journal-title": "Curr Opin Pulm Med",
"key": "10.1016/j.ijid.2026.108519_bib0024",
"volume": "28",
"year": "2022"
},
{
"DOI": "10.1016/j.cpcardiol.2021.101007",
"article-title": "Peripheral artery disease and COVID-19 outcomes: insights from the Yale DOM-CovX registry",
"author": "Smolderen",
"doi-asserted-by": "crossref",
"journal-title": "Curr Probl Cardiol",
"key": "10.1016/j.ijid.2026.108519_bib0025",
"volume": "47",
"year": "2022"
},
{
"DOI": "10.1038/s41598-022-06276-7",
"article-title": "Mortality and readmission rates among hospitalized COVID-19 patients with varying stages of chronic kidney disease: a multicenter retrospective cohort",
"author": "Appelman",
"doi-asserted-by": "crossref",
"first-page": "2258",
"journal-title": "Sci Rep",
"key": "10.1016/j.ijid.2026.108519_bib0026",
"volume": "12",
"year": "2022"
},
{
"DOI": "10.1007/s11684-020-0822-5",
"article-title": "Durability of neutralizing antibodies and T-cell response post SARS-CoV-2 infection",
"author": "Tan",
"doi-asserted-by": "crossref",
"first-page": "746",
"journal-title": "Front Med",
"key": "10.1016/j.ijid.2026.108519_bib0027",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1016/j.isci.2022.103743",
"article-title": "Immunity to SARS-CoV-2 up to 15 months after infection",
"author": "Marcotte",
"doi-asserted-by": "crossref",
"journal-title": "iScience",
"key": "10.1016/j.ijid.2026.108519_bib0028",
"volume": "25",
"year": "2022"
},
{
"article-title": "Longitudinal analysis shows durable and broad immune memory after SARS-CoV-2 infection with persisting antibody responses and memory B and T cells",
"author": "Cohen",
"journal-title": "Cell Rep Med",
"key": "10.1016/j.ijid.2026.108519_bib0029",
"volume": "2",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2118691",
"article-title": "Protection against SARS-CoV-2 after Covid-19 vaccination and previous infection",
"author": "Hall",
"doi-asserted-by": "crossref",
"first-page": "1207",
"journal-title": "N Engl J Med",
"key": "10.1016/j.ijid.2026.108519_bib0030",
"volume": "386",
"year": "2022"
},
{
"DOI": "10.1016/j.envres.2022.112911",
"article-title": "SARS-CoV-2 reinfections: overview of efficacy and duration of natural and hybrid immunity",
"author": "Pilz",
"doi-asserted-by": "crossref",
"journal-title": "Env Res",
"key": "10.1016/j.ijid.2026.108519_bib0031",
"volume": "209",
"year": "2022"
},
{
"DOI": "10.1186/s12879-022-07418-y",
"article-title": "SARS-CoV-2 vaccine effectiveness against infection, symptomatic and severe COVID-19: a systematic review and meta-analysis",
"author": "Ssentongo",
"doi-asserted-by": "crossref",
"first-page": "439",
"journal-title": "BMC Infect Dis",
"key": "10.1016/j.ijid.2026.108519_bib0032",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.1016/S0140-6736(22)00152-0",
"article-title": "Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: results of a systematic review and meta-regression",
"author": "Feikin",
"doi-asserted-by": "crossref",
"first-page": "924",
"journal-title": "Lancet",
"key": "10.1016/j.ijid.2026.108519_bib0033",
"volume": "399",
"year": "2022"
},
{
"DOI": "10.1001/jama.2021.19499",
"article-title": "Association between mRNA vaccination and COVID-19 hospitalization and disease severity",
"author": "Tenforde",
"doi-asserted-by": "crossref",
"first-page": "2043",
"journal-title": "JAMA",
"key": "10.1016/j.ijid.2026.108519_bib0034",
"volume": "326",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)32661-1",
"article-title": "AZ - safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomized controlled trials in Brazil, South Africa, and the UK",
"author": "Voysey",
"doi-asserted-by": "crossref",
"first-page": "99",
"journal-title": "Lancet",
"key": "10.1016/j.ijid.2026.108519_bib0035",
"volume": "397",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2101544",
"article-title": "Jansen safety and efficacy of single-dose Ad26.COV2.S vaccine against Covid-19",
"author": "Sadoff",
"doi-asserted-by": "crossref",
"first-page": "2187",
"journal-title": "N Engl J Med",
"key": "10.1016/j.ijid.2026.108519_bib0036",
"volume": "384",
"year": "2021"
},
{
"key": "10.1016/j.ijid.2026.108519_bib0037",
"unstructured": "Ewopharma International, s.r.o. Isoprinosine 500 mg tablety SmPC. State Institute for Drug Control (SÚKL); 2019. https://prehledy.sukl.cz/prehledy/v1/dokumenty/12916 [accessed 10 August 2025]."
},
{
"key": "10.1016/j.ijid.2026.108519_bib0038",
"unstructured": "Themis Medicare Limited. Viralex SmPC. Rwanda Food and Drug Authority; 2024. https://rwandafda.gov.rw/wp-content/uploads/2025/04//VIRALEX-InosinePranobex500mgTablets-SmPC.pdf [accessed 10 August 2025]."
},
{
"DOI": "10.1016/S1473-3099(22)00507-2",
"article-title": "Real-world effectiveness of early molnupiravir or nirmatrelvir–ritonavir in hospitalised patients with COVID-19 without supplemental oxygen requirement on admission during Hong Kong’s omicron BA.2 wave: a retrospective cohort study",
"author": "Wong",
"doi-asserted-by": "crossref",
"first-page": "1681",
"journal-title": "Lancet Infect Dis",
"key": "10.1016/j.ijid.2026.108519_bib0039",
"volume": "22",
"year": "2022"
}
],
"reference-count": 39,
"references-count": 39,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S1201971226001542"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Inosine pranobex was associated with a reduction in mortality in COVID-19 by over 22%, as reported in a retrospective study utilizing complete health data from the Czech national registry",
"type": "journal-article",
"update-policy": "https://doi.org/10.1016/elsevier_cm_policy"
}