Non-steroidal anti-inflammatory agent use may not be associated with mortality of coronavirus disease 19
et al., Scientific Reports, doi:10.1038/s41598-021-84539-5, Mar 2021
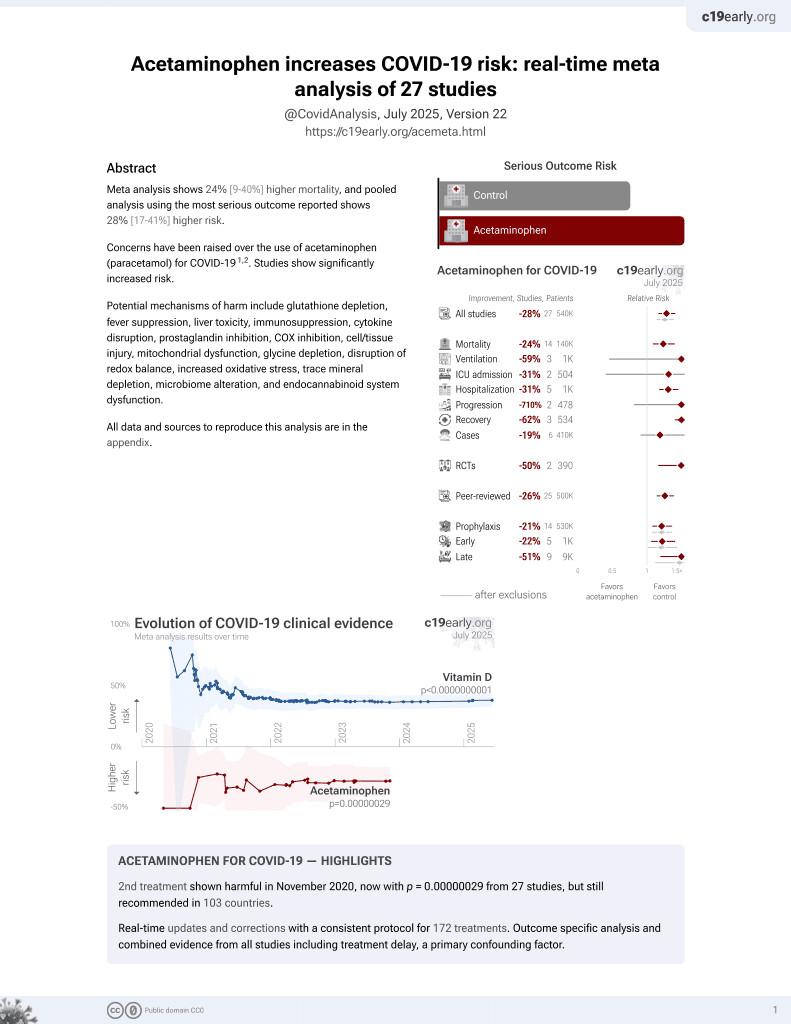
2nd treatment shown to increase risk in
November 2020, now with p = 0.00000029 from 27 studies, but still recommended in 103 countries.
6,600+ studies for
220+ treatments. c19early.org
|
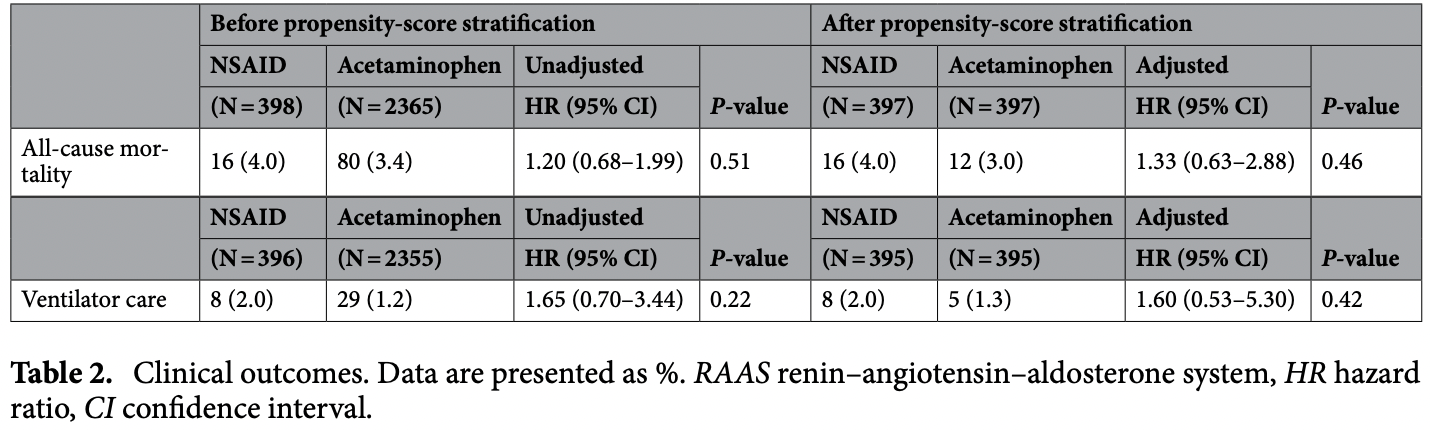
Retrospective 2,365 patients prescribed acetaminophen and 398 prescribed NSAIDs in South Korea, showing no significant differences.
Acetaminophen is also known as paracetamol, Tylenol, Panadol, Calpol, Tempra, Calprofen, Doliprane, Efferalgan, Grippostad C, Dolo, Acamol, Fevadol, Crocin, and Perfalgan.
|
risk of death, 24.8% lower, HR 0.75, p = 0.46, treatment 12 of 397 (3.0%), control 16 of 397 (4.0%), NNT 99, inverted to make HR<1 favor treatment, propensity score matching.
|
|
risk of mechanical ventilation, 37.5% lower, HR 0.62, p = 0.42, treatment 5 of 397 (1.3%), control 8 of 397 (2.0%), NNT 132, inverted to make HR<1 favor treatment, propensity score matching.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Park et al., 3 Mar 2021, retrospective, South Korea, peer-reviewed, 5 authors, this trial compares with another treatment - results may be better when compared to placebo.
Contact: shuaaa.lee@samsung.com, kmhi.yang@samsung.com.
Non-steroidal anti-inflammatory agent use may not be associated with mortality of coronavirus disease 19
Scientific Reports, doi:10.1038/s41598-021-84539-5
Non-steroidal anti-inflammatory drugs (NSAIDs) have been widely used in patients with respiratory infection, but their safety in coronavirus disease 19 (Covid-19) patients has not been fully investigated. We evaluated an association between NSAID use and outcomes of Covid-19. This study was a retrospective observational cohort study based on insurance benefit claims sent to the Health Insurance Review and Assessment Service of Korea by May 15, 2020. These claims comprised all Covid-19-tested cases and history of medical service use for the past 3 years in these patients. The primary outcome was all-cause mortality, and the secondary outcome was need for ventilator care. Among 7590 patients diagnosed with Covid-19, two distinct cohorts were generated based on NSAID or acetaminophen prescription within 2 weeks before Covid-19 diagnosis. A total of 398 patients was prescribed NSAIDs, and 2365 patients were prescribed acetaminophen. After propensity score matching, 397 pairs of data set were generated, and all-cause mortality of the NSAIDs group showed no significant difference compared with the acetaminophen group (4.0% vs. 3.0%; hazard ratio [HR], 1.33; 95% confidence interval [CI], 0.63-2.88; P = 0.46). The rate of ventilator care also did not show significantly different results between the two groups (2.0% vs. 1.3%; HR, 1.60; 95% CI 0.53-5.30; P = 0.42). Use of NSAIDs was not associated with mortality or ventilator care in Covid-19 patients. NSAIDs may be safely used to relieve symptoms in patients with suspicion of Covid-19. In December 2019, a major outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-Cov-2) in Wuhan City, China, was first reported. It was later characterized as coronavirus disease 19 (Covid-19) and has become a global threatening disease with more than 6,000,000 confirmed cases worldwide as of June 2020 1 . By then, more than 11,629 cases of Covid-19 had been diagnosed, and 273 deaths had been reported throughout Korea. Based on this, the government of Korea decided to share the world's first de-identified Covid-19 nationwide patient data collected from the Korean National Health Insurance System for the purpose of investigation. The primary site of infection in Covid-19 is the respiratory system. However, epidemiologic reports indicated that mortality of Covid-19 was much higher in patients with cardiovascular disease 2 , and the most serious complications of Covid-19 are those involving cardiovascular and respiratory systems, as well as sepsis 3, 4 . Non-steroidal anti-inflammatory drugs (NSAIDs) have long been widely used for symptomatic relief of infected patients by controlling pain, fever, and inflammation, although safety concerns remain regarding harmful effects on the cardiovascular system 5 . Moreover, NSAID treatment was associated with pulmonary complication in patients with pneumonia 6 . The association between NSAID use and adverse outcome of Covid-19 has been previously evaluated, but limited..
Author contributions Conceptualization: S.-H.L. Data curation: S.C.Y. and J.K. Formal analysis: J.K. Methodology: J.P., K.Y. Software: J.K. Validation: J.P., K.Y., S.C.Y., and S.-H.L. Investigation: J.P., K.Y., S.-H.L. Writing-original draft: J.P. and S.-H.L. Writing-review and editing: S.C.Y., J.K. and K.Y.
Competing interests The authors declare no competing interests.
References
Basille, Nonsteroidal antiinflammatory drug use and clinical outcomes of community-acquired pneumonia, Am. J. Respir. Crit. Care Med, doi:10.1164/rccm.201802-0229LE
Capuano, Scavone, Racagni, Scaglione, Italian, Society of Pharmacology. NSAIDs in patients with viral infections, including Covid-19: Victims or perpetrators?, Pharmacol. Res, doi:10.1016/j.phrs.2020.104849
Guan, Clinical characteristics of coronavirus disease 2019 in China, N. Engl. J. Med, doi:10.1056/NEJMoa2002032
Huang, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet, doi:10.1016/S0140-6736(20)30183-5
Little, Non-steroidal anti-inflammatory drugs and covid-19, BMJ, doi:10.1136/bmj.m1185
Overhage, Ryan, Reich, Hartzema, Stang, Validation of a common data model for active safety surveillance research, J. Am. Med. Inform. Assoc, doi:10.1136/amiajnl-2011-000376
Rinott, Kozer, Shapira, Bar-Haim, Youngster, Ibuprofen use and clinical outcomes in COVID-19 patients, Clin. Microbiol. Infect, doi:10.1016/j.cmi.2020.06.003
Russell, Moss, Rigg, Van Hemelrijck, COVID-19 and treatment with NSAIDs and corticosteroids: Should we be limiting their use in the clinical setting?, Ecancermedicalscience, doi:10.3332/ecancer.2020.1023
Schjerning, Mcgettigan, Gislason, Cardiovascular effects and safety of (non-aspirin) NSAIDs, Nat. Rev. Cardiol, doi:10.1038/s41569-020-0366-z
Suchard, Simpson, Zorych, Ryan, Madigan, Massive parallelization of serial inference algorithms for a complex generalized linear model, ACM Trans. Model. Comput. Simul, doi:10.1145/2414416.2414791
Szeto, Non-steroidal anti-inflammatory drug (NSAID) therapy in patients with hypertension, cardiovascular, renal or gastrointestinal comorbidities: Joint APAGE/APLAR/APSDE/APSH/APSN/PoA recommendations, Gut, doi:10.1136/gutjnl-2019-319300
Wen, Acute respiratory infection and use of nonsteroidal anti-inflammatory drugs on risk of acute myocardial infarction: A nationwide case-crossover study, J. Infect. Dis, doi:10.1093/infdis/jiw603
You, Precision Healthcare Through Informatics
Zheng, Ma, Zhang, Xie, COVID-19 and the cardiovascular system, Nat. Rev. Cardiol, doi:10.1038/s41569-020-0360-5
Zhou, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study, Lancet, doi:10.1016/S0140-6736(20)30566-3
DOI record:
{
"DOI": "10.1038/s41598-021-84539-5",
"ISSN": [
"2045-2322"
],
"URL": "http://dx.doi.org/10.1038/s41598-021-84539-5",
"abstract": "<jats:title>Abstract</jats:title><jats:p>Non-steroidal anti-inflammatory drugs (NSAIDs) have been widely used in patients with respiratory infection, but their safety in coronavirus disease 19 (Covid-19) patients has not been fully investigated. We evaluated an association between NSAID use and outcomes of Covid-19. This study was a retrospective observational cohort study based on insurance benefit claims sent to the Health Insurance Review and Assessment Service of Korea by May 15, 2020. These claims comprised all Covid-19-tested cases and history of medical service use for the past 3 years in these patients. The primary outcome was all-cause mortality, and the secondary outcome was need for ventilator care. Among 7590 patients diagnosed with Covid-19, two distinct cohorts were generated based on NSAID or acetaminophen prescription within 2 weeks before Covid-19 diagnosis. A total of 398 patients was prescribed NSAIDs, and 2365 patients were prescribed acetaminophen. After propensity score matching, 397 pairs of data set were generated, and all-cause mortality of the NSAIDs group showed no significant difference compared with the acetaminophen group (4.0% vs. 3.0%; hazard ratio [HR], 1.33; 95% confidence interval [CI], 0.63–2.88; <jats:italic>P</jats:italic> = 0.46). The rate of ventilator care also did not show significantly different results between the two groups (2.0% vs. 1.3%; HR, 1.60; 95% CI 0.53–5.30; <jats:italic>P</jats:italic> = 0.42). Use of NSAIDs was not associated with mortality or ventilator care in Covid-19 patients. NSAIDs may be safely used to relieve symptoms in patients with suspicion of Covid-19.</jats:p>",
"alternative-id": [
"84539"
],
"article-number": "5087",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "1 September 2020"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "16 February 2021"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "3 March 2021"
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Park",
"given": "Jungchan",
"sequence": "first"
},
{
"affiliation": [],
"family": "Lee",
"given": "Seung-Hwa",
"sequence": "additional"
},
{
"affiliation": [],
"family": "You",
"given": "Seng Chan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kim",
"given": "Jinseob",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yang",
"given": "Kwangmo",
"sequence": "additional"
}
],
"container-title": "Scientific Reports",
"container-title-short": "Sci Rep",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2021,
3,
3
]
],
"date-time": "2021-03-03T11:02:36Z",
"timestamp": 1614769356000
},
"deposited": {
"date-parts": [
[
2021,
12,
2
]
],
"date-time": "2021-12-02T16:10:27Z",
"timestamp": 1638461427000
},
"funder": [
{
"award": [
"HI19C0811"
],
"name": "Ministry of Health and Welfare, Korea"
}
],
"indexed": {
"date-parts": [
[
2022,
8,
25
]
],
"date-time": "2022-08-25T04:28:48Z",
"timestamp": 1661401728997
},
"is-referenced-by-count": 7,
"issue": "1",
"issued": {
"date-parts": [
[
2021,
3,
3
]
]
},
"journal-issue": {
"issue": "1",
"published-print": {
"date-parts": [
[
2021,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
3,
3
]
],
"date-time": "2021-03-03T00:00:00Z",
"timestamp": 1614729600000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
3,
3
]
],
"date-time": "2021-03-03T00:00:00Z",
"timestamp": 1614729600000
}
}
],
"link": [
{
"URL": "http://www.nature.com/articles/s41598-021-84539-5.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "http://www.nature.com/articles/s41598-021-84539-5",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "http://www.nature.com/articles/s41598-021-84539-5.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1038",
"published": {
"date-parts": [
[
2021,
3,
3
]
]
},
"published-online": {
"date-parts": [
[
2021,
3,
3
]
]
},
"published-print": {
"date-parts": [
[
2021,
12
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"key": "84539_CR1",
"unstructured": "World Health Organization. Rolling updates on coronavirus disease (COVID-19) (2020). https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen. Accessed June 15, 2020."
},
{
"DOI": "10.46234/ccdcw2020.032",
"doi-asserted-by": "crossref",
"key": "84539_CR2",
"unstructured": "The Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. Vital Surveillances: The Epidemiological Characteristics of an Outbreak of 2019 Novel Coronavirus Diseases (COVID-19)—China (2020). http://weekly.chinacdc.cn/en/article/id/e53946e2-c6c4-41e9-9a9b-fea8db1a8f51. Accessed June 15, 2020."
},
{
"DOI": "10.1056/NEJMoa2002032",
"author": "WJ Guan",
"doi-asserted-by": "publisher",
"first-page": "1708",
"journal-title": "N. Engl. J. Med.",
"key": "84539_CR3",
"unstructured": "Guan, W. J. et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 382, 1708–1720. https://doi.org/10.1056/NEJMoa2002032 (2020).",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"author": "F Zhou",
"doi-asserted-by": "publisher",
"first-page": "1054",
"journal-title": "Lancet",
"key": "84539_CR4",
"unstructured": "Zhou, F. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 395, 1054–1062. https://doi.org/10.1016/S0140-6736(20)30566-3 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(13)60900-9",
"author": "Coxib and Traditional NSAID Trialists' Collaboration",
"doi-asserted-by": "publisher",
"first-page": "769",
"journal-title": "Lancet",
"key": "84539_CR5",
"unstructured": "Coxib and Traditional NSAID Trialists’ Collaboration et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet 382, 769–779. https://doi.org/10.1016/S0140-6736(13)60900-9 (2013).",
"volume": "382",
"year": "2013"
},
{
"DOI": "10.1164/rccm.201802-0229LE",
"author": "D Basille",
"doi-asserted-by": "publisher",
"first-page": "128",
"journal-title": "Am. J. Respir. Crit. Care Med.",
"key": "84539_CR6",
"unstructured": "Basille, D. et al. Nonsteroidal antiinflammatory drug use and clinical outcomes of community-acquired pneumonia. Am. J. Respir. Crit. Care Med. 198, 128–131. https://doi.org/10.1164/rccm.201802-0229LE (2018).",
"volume": "198",
"year": "2018"
},
{
"DOI": "10.3332/ecancer.2020.1023",
"author": "B Russell",
"doi-asserted-by": "publisher",
"first-page": "1023",
"journal-title": "Ecancermedicalscience",
"key": "84539_CR7",
"unstructured": "Russell, B., Moss, C., Rigg, A. & Van Hemelrijck, M. COVID-19 and treatment with NSAIDs and corticosteroids: Should we be limiting their use in the clinical setting?. Ecancermedicalscience 14, 1023. https://doi.org/10.3332/ecancer.2020.1023 (2020).",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1136/bmj.m1185",
"author": "P Little",
"doi-asserted-by": "publisher",
"first-page": "m1185",
"journal-title": "BMJ",
"key": "84539_CR8",
"unstructured": "Little, P. Non-steroidal anti-inflammatory drugs and covid-19. BMJ 368, m1185. https://doi.org/10.1136/bmj.m1185 (2020).",
"volume": "368",
"year": "2020"
},
{
"DOI": "10.1016/j.phrs.2020.104849",
"author": "A Capuano",
"doi-asserted-by": "publisher",
"first-page": "104849",
"journal-title": "Pharmacol. Res.",
"key": "84539_CR9",
"unstructured": "Capuano, A., Scavone, C., Racagni, G., Scaglione, F. & Italian Society of Pharmacology. NSAIDs in patients with viral infections, including Covid-19: Victims or perpetrators?. Pharmacol. Res. 157, 104849. https://doi.org/10.1016/j.phrs.2020.104849 (2020).",
"volume": "157",
"year": "2020"
},
{
"DOI": "10.1016/j.cmi.2020.06.003",
"author": "E Rinott",
"doi-asserted-by": "publisher",
"journal-title": "Clin. Microbiol. Infect.",
"key": "84539_CR10",
"unstructured": "Rinott, E., Kozer, E., Shapira, Y., Bar-Haim, A. & Youngster, I. Ibuprofen use and clinical outcomes in COVID-19 patients. Clin. Microbiol. Infect. https://doi.org/10.1016/j.cmi.2020.06.003 (2020).",
"year": "2020"
},
{
"DOI": "10.1136/gutjnl-2019-319300",
"author": "CC Szeto",
"doi-asserted-by": "publisher",
"first-page": "617",
"journal-title": "Gut",
"key": "84539_CR11",
"unstructured": "Szeto, C. C. et al. Non-steroidal anti-inflammatory drug (NSAID) therapy in patients with hypertension, cardiovascular, renal or gastrointestinal comorbidities: Joint APAGE/APLAR/APSDE/APSH/APSN/PoA recommendations. Gut 69, 617–629. https://doi.org/10.1136/gutjnl-2019-319300 (2020).",
"volume": "69",
"year": "2020"
},
{
"DOI": "10.1093/infdis/jiw603",
"author": "YC Wen",
"doi-asserted-by": "publisher",
"first-page": "503",
"journal-title": "J. Infect. Dis.",
"key": "84539_CR12",
"unstructured": "Wen, Y. C. et al. Acute respiratory infection and use of nonsteroidal anti-inflammatory drugs on risk of acute myocardial infarction: A nationwide case-crossover study. J. Infect. Dis. 215, 503–509. https://doi.org/10.1093/infdis/jiw603 (2017).",
"volume": "215",
"year": "2017"
},
{
"DOI": "10.1038/s41569-020-0366-z",
"author": "AM Schjerning",
"doi-asserted-by": "publisher",
"first-page": "574",
"journal-title": "Nat. Rev. Cardiol.",
"key": "84539_CR13",
"unstructured": "Schjerning, A. M., McGettigan, P. & Gislason, G. Cardiovascular effects and safety of (non-aspirin) NSAIDs. Nat. Rev. Cardiol. 17, 574–584. https://doi.org/10.1038/s41569-020-0366-z (2020).",
"volume": "17",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30183-5",
"author": "C Huang",
"doi-asserted-by": "publisher",
"first-page": "497",
"journal-title": "Lancet",
"key": "84539_CR14",
"unstructured": "Huang, C. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395, 497–506. https://doi.org/10.1016/S0140-6736(20)30183-5 (2020).",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1038/s41569-020-0360-5",
"author": "YY Zheng",
"doi-asserted-by": "publisher",
"first-page": "259",
"journal-title": "Nat. Rev. Cardiol.",
"key": "84539_CR15",
"unstructured": "Zheng, Y. Y., Ma, Y. T., Zhang, J. Y. & Xie, X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 17, 259–260. https://doi.org/10.1038/s41569-020-0360-5 (2020).",
"volume": "17",
"year": "2020"
},
{
"author": "SC You",
"first-page": "467",
"key": "84539_CR16",
"unstructured": "You, S. C. et al. In MEDINFO 2017: Precision Healthcare Through Informatics (eds Gundlapalli, A. V. et al.) 467–470 (IOS Press, Amsterdam, 2018).",
"volume-title": "MEDINFO 2017: Precision Healthcare Through Informatics",
"year": "2018"
},
{
"DOI": "10.1136/amiajnl-2011-000376",
"author": "JM Overhage",
"doi-asserted-by": "publisher",
"first-page": "54",
"journal-title": "J. Am. Med. Inform. Assoc.",
"key": "84539_CR17",
"unstructured": "Overhage, J. M., Ryan, P. B., Reich, C. G., Hartzema, A. G. & Stang, P. E. Validation of a common data model for active safety surveillance research. J. Am. Med. Inform. Assoc. 19, 54–60. https://doi.org/10.1136/amiajnl-2011-000376 (2012).",
"volume": "19",
"year": "2012"
},
{
"DOI": "10.1145/2414416.2414791",
"author": "MA Suchard",
"doi-asserted-by": "publisher",
"first-page": "Article 10",
"issue": "1",
"journal-title": "ACM Trans. Model. Comput. Simul.",
"key": "84539_CR18",
"unstructured": "Suchard, M. A., Simpson, S. E., Zorych, I., Ryan, P. & Madigan, D. Massive parallelization of serial inference algorithms for a complex generalized linear model. ACM Trans. Model. Comput. Simul. 23(1), Article 10. https://doi.org/10.1145/2414416.2414791 (2013).",
"volume": "23",
"year": "2013"
}
],
"reference-count": 18,
"references-count": 18,
"relation": {},
"resource": {
"primary": {
"URL": "http://www.nature.com/articles/s41598-021-84539-5"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Multidisciplinary"
],
"subtitle": [],
"title": "Non-steroidal anti-inflammatory agent use may not be associated with mortality of coronavirus disease 19",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1007/springer_crossmark_policy",
"volume": "11"
}