Frequency and Severity of COVID-19 in Patients with Various Rheumatic Diseases Treated Regularly with Colchicine or Hydroxychloroquine
et al., Journal of Medical Virology, doi:10.1002/jmv.27731, Mar 2022
Colchicine for COVID-19
5th treatment shown to reduce risk in
September 2020, now with p = 0.0000049 from 54 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
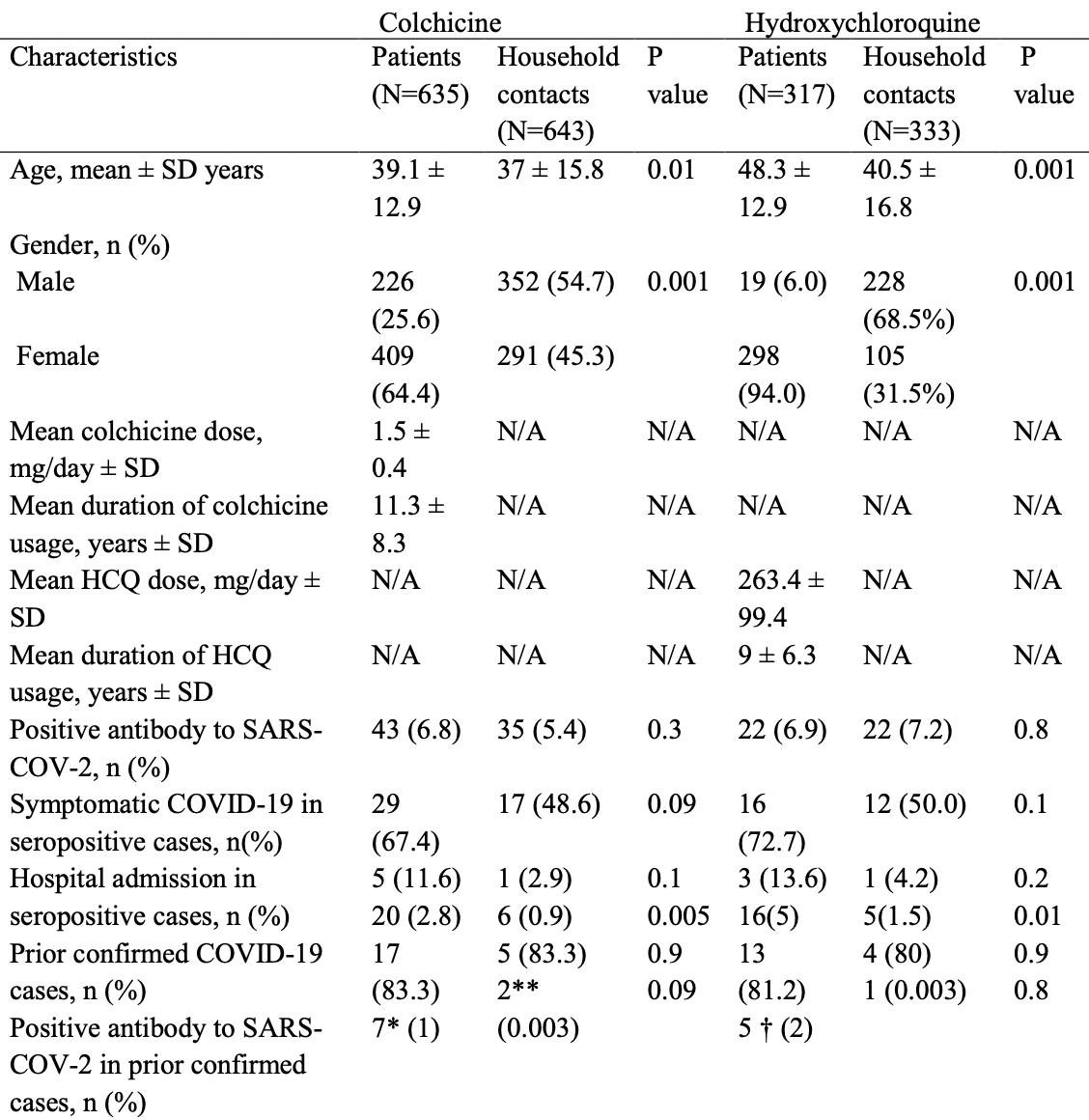
Retrospective 635 HCQ users and 643 household contacts, showing higher risk with colchicine in unadjusted results.
Patients with conditions leading to the use of colchicine may have significantly different baseline risk, e.g.1.
This study is excluded in the after exclusion results of meta-analysis:
excessive unadjusted differences between groups.
Study covers HCQ and colchicine.
|
risk of hospitalization, 406.3% higher, RR 5.06, p = 0.12, treatment 5 of 635 (0.8%), control 1 of 643 (0.2%).
|
|
risk of symptomatic case, 72.7% higher, RR 1.73, p = 0.07, treatment 29 of 635 (4.6%), control 17 of 643 (2.6%).
|
|
risk of case, 24.4% higher, RR 1.24, p = 0.35, treatment 43 of 635 (6.8%), control 35 of 643 (5.4%).
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Oztas et al., 21 Mar 2022, retrospective, Turkey, peer-reviewed, 15 authors.
Frequency and severity of COVID‐19 in patients with various rheumatic diseases treated regularly with colchicine or hydroxychloroquine
Journal of Medical Virology, doi:10.1002/jmv.27731
Introduction. The current study aimed to investigate whether patients regularly using colchicine or hydroxychloroquine (HCQ) have an advantage of protection from COVID-19 or developing less severe disease. Methods. Patients who were taking colchicine or HCQ regularly for a rheumatic disease including Familial Mediterranean Fever, Behçet's syndrome, Systemic Lupus Erythematosus, Rheumatoid Arthritis and Sjogren's syndrome as well as their healthy household contacts as the control group were included into the study. The clinical data regarding COVID-19 were collected using a standard form, and serum samples were analyzed for anti-SARS-COV-2 nucleocapsid IgG. Results. A total of 635 regular colchicine users with their 643 household contacts and 317 regular HCQ users with their 333 household contacts were analyzed. Anti-SARS-Cov2 IgG was positive in 43 (6.8%) regular colchicine users and 35 (5.4%) household contacts (OR=1.3; 95% CI:0.8-2; p=0.3). COVID-19 related symptoms were described by 29 (67.4%) of the patients and 17 (48.6%) household contacts (OR=2.2; 95% CI:0.9-5.5; p=0.09), and hospital admission was observed in five (11.6%) and one (2.9%) of these subjects (OR=4.5; 95% CI:0.5-40.2; p=0.1), respectively. Seropositive subjects were observed in 22 (6.9%) regular HCQ users and 24 (7.2%) household contacts (OR=1.1; CI:0.6-1.9; p=0.8). COVID-19-related symptoms occurred in 16 (72.7%) of the 22 patients and 12 (50%) of 24 household contacts (OR=2.7; 95% CI:0.8-9.1; p=0.1). Three patients (13.6%) were admitted to hospital, while one household contact (4.2%) was hospitalized (OR=3.6; 95% CI:0.3-37.8; p=0.2). Conclusions. Being on a regular treatment of colchicine or HCQ was not resulted in the prevention of COVID-19 or amelioration of its manifestations.
Accepted Article In conclusion, being on the treatment of regular doses of colchicine or HCQ was not effective in the prevention of COVID-19 and ameliorating its manifestations. Nevertheless, these medications did not cause worse outcomes during the course of the COVID-19 neither. However, further studies are needed to assess the preventive role of colchicine or HCQ.
Conflict of Intersest Statement
References
Bourguiba, Delplanque, Vinit, Ackermann, Clinical course of COVID-19 in a cohort of 342 familial Mediterranean fever patients with a long-term treatment by colchicine in a French endemic area, doi:10.1136/annrheumdis-2020-218707
Conway, Grimshaw, Konig, Putman, SARS-CoV-2 Infection and COVID-19 Outcomes in Rheumatic Disease: A Systematic Literature Review And Meta-Analysis, Arthritis & rheumatology, doi:10.1002/art.42030
Fenwick, Croxatto, Coste, Pojer, Changes in SARS-CoV-2 Spike versus Nucleoprotein Antibody Responses Impact the Estimates of Infections in Population-Based Seroprevalence Studies, Journal of virology, doi:10.1128/JVI.01828-20
Hatemi, Christensen, Bang, Bodaghi, 2018 update of the EULAR recommendations for the management of Behçet's syndrome, Annals of the rheumatic diseases, doi:10.1136/annrheumdis-2018-213225
Huang, Wang, Li, Ren, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet, doi:10.1016/S0140-6736
Konig, Kim, Scheetz, Graef, Baseline use of hydroxychloroquine in systemic lupus erythematosus does not preclude SARS-CoV-2 infection and severe COVID-19, Annals of the rheumatic diseases, doi:10.1136/annrheumdis-2020-217690
Misawa, Takahama, Kozaki, Lee, Zou et al., Microtubule-driven spatial arrangement of mitochondria promotes activation of the NLRP3 inflammasome, Nature immunology, doi:10.1038/ni.2550
Muench, Jochum, Wenderoth, Ofenloch-Haehnle, Development and Validation of the Elecsys Anti-SARS-CoV-2 Immunoassay as a Highly Specific Tool for Determining Past Exposure to SARS-CoV-2, Journal of clinical microbiology, doi:10.1128/JCM.01694-20
Ozdogan, Ugurlu, Familial Mediterranean Fever, Presse medicale, doi:10.1016/j.lpm.2018.08.014
Ruan, Yang, Wang, Jiang, Song, Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China, Intensive care medicine, doi:10.1007/s00134-020-05991-x
Schrezenmeier, Dörner, Yao, Zhang, Cui, Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), Infectious Diseases Society of America, doi:10.1038/s41584-020-0372-x
Shi, Nabar, Huang, Kehrl, SARS-Coronavirus Open Reading Frame-8b triggers intracellular stress pathways and activates NLRP3 inflammasomes, Cell death discovery, doi:10.1038/s41420-019-0181-7
Skipper, Pastick, Engen, Bangdiwala, None
Tardif, Bouabdallaoui, L'allier, Gaudet, None
Topless, Gaffo, Stamp, Robinson, Dalbeth et al., Gout and the risk of COVID-19 diagnosis and death in the UK Biobank: a population-based study, The Lancet. Rheumatology, doi:10.1016/S2665-9913(21)00401-X
Zhang, Zhao, Zhang, Wang, The use of antiinflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): The Perspectives of clinical immunologists from China, Clinical immunology, doi:10.1016/j.clim.2020.108393
DOI record:
{
"DOI": "10.1002/jmv.27731",
"ISSN": [
"0146-6615",
"1096-9071"
],
"URL": "http://dx.doi.org/10.1002/jmv.27731",
"alternative-id": [
"10.1002/jmv.27731"
],
"archive": [
"Portico"
],
"assertion": [
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 2,
"value": "2022-03-21"
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-4077-1374",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Oztas",
"given": "Mert",
"sequence": "first"
},
{
"ORCID": "http://orcid.org/0000-0002-1788-3837",
"affiliation": [
{
"name": "Istanbul University, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Bektas",
"given": "Murat",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-3100-0866",
"affiliation": [
{
"name": "Istanbul Medeniyet University,Department of Molecular Biology and GeneticsIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Karacan",
"given": "Ilker",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-2848-4396",
"affiliation": [
{
"name": "Istanbul University, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Aliyeva",
"given": "Numune",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-3157-531X",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Dag",
"given": "Ayten",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7196-0098",
"affiliation": [
{
"name": "Istanbul University, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Aghamuradov",
"given": "Sarvan",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-7099-4827",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Cevirgen",
"given": "Selim Berke",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-6839-8164",
"affiliation": [
{
"name": "Istanbul University, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Sari",
"given": "Selma",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5755-7860",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medical BiochemistryIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Bolayirli",
"given": "Murat",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-5815-6700",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Public HealthIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Can",
"given": "Gunay",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-1952-1135",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Hatemi",
"given": "Gulen",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-4965-2918",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Seyahi",
"given": "Emire",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0003-4632-8258",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Ozdogan",
"given": "Huri",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8219-3720",
"affiliation": [
{
"name": "Istanbul University, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Gul",
"given": "Ahmet",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9561-2282",
"affiliation": [
{
"name": "Istanbul University‐Cerrahpasa, Department of Medicine, Division of RheumatologyIstanbulTurkey"
}
],
"authenticated-orcid": false,
"family": "Ugurlu",
"given": "Serdal",
"sequence": "additional"
}
],
"container-title": [
"Journal of Medical Virology"
],
"content-domain": {
"crossmark-restriction": true,
"domain": [
"onlinelibrary.wiley.com"
]
},
"created": {
"date-parts": [
[
2022,
3,
22
]
],
"date-time": "2022-03-22T04:20:11Z",
"timestamp": 1647922811000
},
"deposited": {
"date-parts": [
[
2022,
3,
22
]
],
"date-time": "2022-03-22T04:20:11Z",
"timestamp": 1647922811000
},
"indexed": {
"date-parts": [
[
2022,
3,
22
]
],
"date-time": "2022-03-22T04:40:54Z",
"timestamp": 1647924054714
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "0146-6615"
},
{
"type": "electronic",
"value": "1096-9071"
}
],
"issued": {
"date-parts": [
[
2022,
3,
21
]
]
},
"language": "en",
"license": [
{
"URL": "http://onlinelibrary.wiley.com/termsAndConditions#vor",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
3,
21
]
],
"date-time": "2022-03-21T00:00:00Z",
"timestamp": 1647820800000
}
},
{
"URL": "http://doi.wiley.com/10.1002/tdm_license_1.1",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
3,
21
]
],
"date-time": "2022-03-21T00:00:00Z",
"timestamp": 1647820800000
}
}
],
"link": [
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1002/jmv.27731",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1002/jmv.27731",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "311",
"original-title": [],
"prefix": "10.1002",
"published": {
"date-parts": [
[
2022,
3,
21
]
]
},
"published-online": {
"date-parts": [
[
2022,
3,
21
]
]
},
"publisher": "Wiley",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://onlinelibrary.wiley.com/doi/10.1002/jmv.27731"
}
},
"score": 1,
"short-container-title": [
"Journal of Medical Virology"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Virology"
],
"subtitle": [],
"title": [
"Frequency and Severity of COVID‐19 in Patients with Various Rheumatic Diseases Treated Regularly with Colchicine or Hydroxychloroquine"
],
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1002/crossmark_policy"
}
oztas
