MMP1 Serves as a Common Link and Drug Target Between Gout and Severe COVID-19 Infection
et al., Elsevier BV, doi:10.2139/ssrn.6256797, Feb 2026
Colchicine for COVID-19
5th treatment shown to reduce risk in
September 2020, now with p = 0.0000049 from 54 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
In silico study identifying MMP1 as a shared therapeutic target between gout and severe COVID-19, suggesting colchicine may exert anti-inflammatory effects via direct binding to MMP1.
2 preclinical studies support the efficacy of colchicine for COVID-19:
Ma et al., 23 Feb 2026, preprint, 7 authors.
Contact: wanghui@xxmu.edu.cn, niuyuna@126.com, 913397855@qq.com, gys0801@163.com, liuminghang2025@163.com, fengtairan2025@163.com, 15090126180@163.com.
In silico studies are an important part of preclinical research, however results may be very different in vivo.
Abstract: MMP1 Serves as a Common Link and Drug Target Between Gout and Severe COVID-19 Infection Haowen Ma 1 , Yushuai Geng 1 , Minghang Liu 1 , Tairan Feng 1 , Yang Lv 1 , Hui Wang 1,2 , Yuna Niu 1,2 Affiliations 1 School of Medical Technology, Henan Medical University, Xinxiang, Henan 453003, China. 2 Henan Collaborative Innovation Center of Molecular Diagnosis and Laboratory Medicine, Henan Medical University, Xinxiang, Henan 453003, China. Correspondence: Hui Wang (wanghui@xxmu.edu.cn), Yuna Niu (niuyuna@126.com) Haowen Ma 913397855@qq.com Yushuai Geng gys0801@163.com Minghang Liu liuminghang2025@163.com Tairan Feng fengtairan2025@163.com Yang Lv 15090126180@163.com Hui Wang wanghui@xxmu.edu.cn Yuna Niu niuyuna@126.com ABSTRACT Background Gout is a significant risk factor for severe COVID-19 and associated mortality. Anti-gout therapeutics such as colchicine have shown efficacy against COVID-19, suggesting shared pathological mechanisms. However, the underlying molecular connections between gout and severe COVID-19 remain unclear. Methods We performed an integrated bioinformatic analysis using transcriptomic datasets from gout (GSE160170) and severe COVID-19 (GSE171110) to identify Preprint not peer reviewed
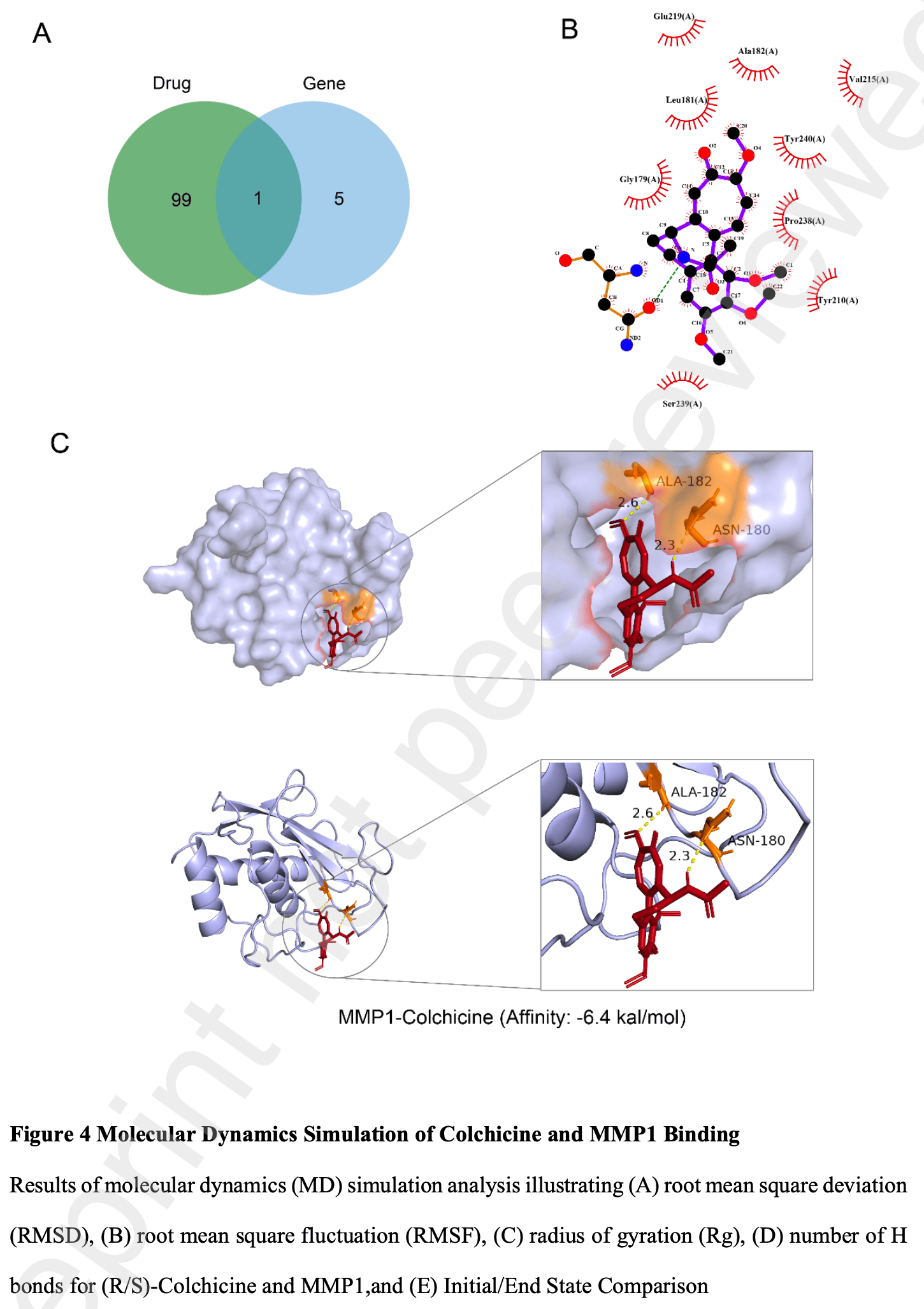
common differentially expressed genes (DEGs). Functional enrichment, proteinprotein interaction (PPI) network analysis, and hub gene identification were conducted. Molecular docking and 100 ns molecular dynamics simulations were used to evaluate the binding between colchicine and the candidate target protein. Additionally, candidate small-molecule drugs and herbal formulas were screened based on hub genes. Results We identified 19 common DEGs enriched in immune-related pathways. Six hub genes (CXCL8, MPO, JUN, MMP1, IL4, IL10) were identified, among which MMP1 was the only gene overlapping with colchicine's predicted targets. Molecular docking revealed stable binding between colchicine and MMP1, with a binding affinity of -6.4 kcal/mol. Molecular dynamics simulations confirmed the stability of the colchicine-MMP1 complex based on RMSD, RMSF, Rg, and hydrogen bonding analyses. Ten candidate small-molecule drugs and six herbal formulas targeting the hub genes were proposed. Single-cell RNA sequencing further validated elevated MMP1 expression in B cells of severe COVID-19 patients. Conclusion Our study identifies MMP1 as a shared therapeutic target for gout and severe COVID-19 and provides novel mechanistic insight into colchicine's antiinflammatory action via direct interaction with MMP1. These findings offer a resource for understanding the comorbidity and may inform the development of new treatment strategies. Keywords: COVID-19, Gout, MMP1, Colchicine, Drug target Introduction The global outbreak of COVID-19, caused by the novel coronavirus SARS-CoV-2, has posed an unprecedented challenge to public health systems worldwide (WangPerlman, 2022; Ye et al., 2020). While a majority of infected individuals experience mild to moderate symptoms, a significant subset progresses to severe disease characterized by excessive inflammation, a dysregulated immune response often referred to as a cytokine storm, which can lead to acute respiratory distress Preprint not peer reviewed
syndrome, multi-organ failure, and death (WangPerlman, 2022). It is well-established that pre-existing comorbidities, such as hypertension, diabetes, and..
DOI record:
{
"DOI": "10.2139/ssrn.6256797",
"URL": "http://dx.doi.org/10.2139/ssrn.6256797",
"abstract": "<jats:p>Background Gout is a significant risk factor for severe COVID-19 and associated mortality. Anti-gout therapeutics such as colchicine have shown efficacy against COVID-19, suggesting shared pathological mechanisms. However, the underlying molecular connections between gout and severe COVID-19 remain unclear.,Methods We performed an integrated bioinformatic analysis using transcriptomic datasets from gout (GSE160170) and severe COVID-19 (GSE171110) to identify common differentially expressed genes (DEGs). Functional enrichment, protein-protein interaction (PPI) network analysis, and hub gene identification were conducted. Molecular docking and 100 ns molecular dynamics simulations were used to evaluate the binding between colchicine and the candidate target protein. Additionally, candidate small-molecule drugs and herbal formulas were screened based on hub genes.,Results We identified 19 common DEGs enriched in immune-related pathways. Six hub genes (CXCL8, MPO, JUN, MMP1, IL4, IL10) were identified, among which MMP1 was the only gene overlapping with colchicine&apos;s predicted targets. Molecular docking revealed stable binding between colchicine and MMP1, with a binding affinity of -6.4 kcal/mol. Molecular dynamics simulations confirmed the stability of the colchicine-MMP1 complex based on RMSD, RMSF, Rg, and hydrogen bonding analyses. Ten candidate small-molecule drugs and six herbal formulas targeting the hub genes were proposed. Single-cell RNA sequencing further validated elevated MMP1 expression in B cells of severe COVID-19 patients.,Conclusion Our study identifies MMP1 as a shared therapeutic target for gout and severe COVID-19 and provides novel mechanistic insight into colchicine&apos;s anti-inflammatory action via direct interaction with MMP1. These findings offer a resource for understanding the comorbidity and may inform the development of new treatment strategies.</jats:p>",
"author": [
{
"affiliation": [],
"family": "Ma",
"given": "Haowen",
"sequence": "first"
},
{
"affiliation": [],
"family": "Geng",
"given": "Yushuai",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Liu",
"given": "Minghang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Feng",
"given": "Tairan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lv",
"given": "Yang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Hui",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-9990-9481",
"affiliation": [],
"authenticated-orcid": true,
"family": "Niu",
"given": "Yuna",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
2,
23
]
],
"date-time": "2026-02-23T16:05:52Z",
"timestamp": 1771862752000
},
"deposited": {
"date-parts": [
[
2026,
2,
23
]
],
"date-time": "2026-02-23T16:05:52Z",
"timestamp": 1771862752000
},
"group-title": "SSRN",
"indexed": {
"date-parts": [
[
2026,
2,
23
]
],
"date-time": "2026-02-23T17:02:48Z",
"timestamp": 1771866168286,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026
]
]
},
"member": "78",
"original-title": [],
"posted": {
"date-parts": [
[
2026
]
]
},
"prefix": "10.2139",
"published": {
"date-parts": [
[
2026
]
]
},
"publisher": "Elsevier BV",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.ssrn.com/abstract=6256797"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"subtype": "preprint",
"title": "MMP1 Serves as a Common Link and Drug Target Between Gout and Severe COVID-19 Infection",
"type": "posted-content"
}
