High-dose intravenous Vitamin C in early stages of severe acute respiratory syndrome coronavirus 2 infection: A double-blind, randomized, controlled clinical trial
et al., Journal of Research in Pharmacy Practice, doi:10.4103/jrpp.jrpp_30_22, IRCT20190917044805N2, Dec 2022
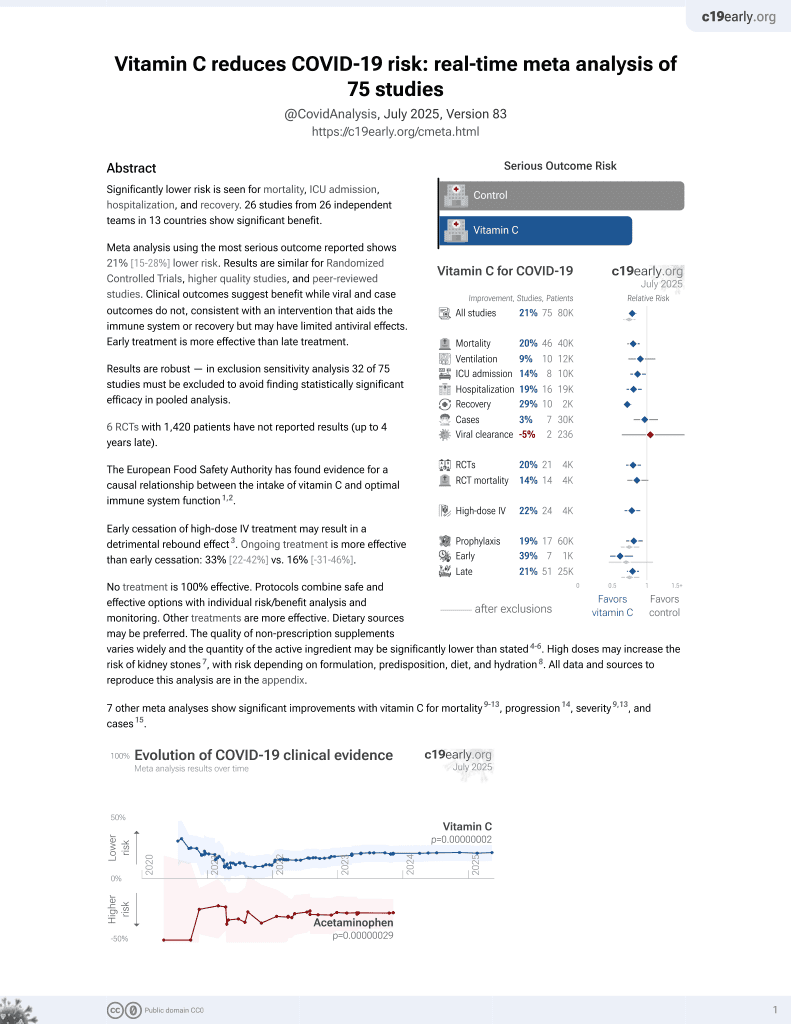
Vitamin C for COVID-19
6th treatment shown to reduce risk in
September 2020, now with p = 0.000000069 from 72 studies, recognized in 22 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT 74 patients in Iran, showing no significant differences in outcomes with high-dose vitamin C treatment. Tables 1b and 2a show conflicting baseline SOFA scores. The percentages of patients receiving antiviral treatments and corticosteroids are switched between the text and Table 1b. Authors indicate ICU admission was an outcome, but the result is not provided. AKI was lower with treatment, though not reaching statistical significance.
This is the 14th of 20 COVID-19 RCTs for vitamin C, which collectively show efficacy with p=0.0016.
This is the 55th of 72 COVID-19 controlled studies for vitamin C, which collectively show efficacy with p=0.000000069.
|
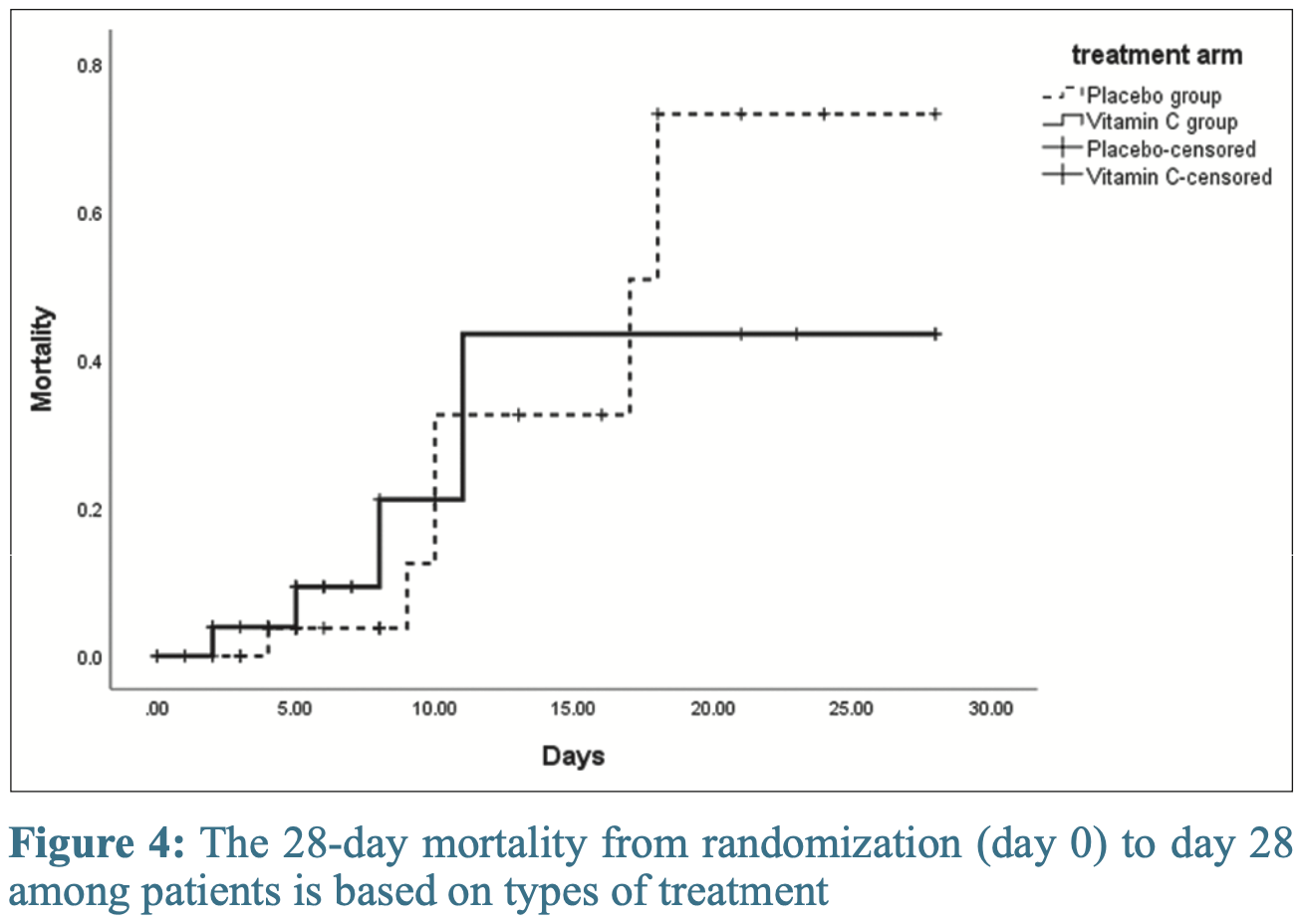
risk of death, 33.3% lower, RR 0.67, p = 0.74, treatment 4 of 37 (10.8%), control 6 of 37 (16.2%), NNT 18, day 28.
|
|
hospitalization time, 12.8% higher, relative time 1.13, p = 0.49, treatment mean 9.24 (±7.5) n=37, control mean 8.19 (±5.34) n=37.
|
|
risk of progression, 15.9% lower, RR 0.84, p = 0.12, treatment 37, control 37, SOFA, day 5.
|
|
risk of progression, 9.3% higher, RR 1.09, p = 0.47, treatment 37, control 37, NEWS, day 5.
|
|
risk of progression, 5.8% higher, RR 1.06, p = 0.38, treatment 37, control 37, WHO, day 5.
|
|
risk of progression, 60.0% lower, RR 0.40, p = 0.14, treatment 4 of 37 (10.8%), control 10 of 37 (27.0%), NNT 6.2, AKI.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Labbani-Motlagh et al., 14 Dec 2022, Double Blind Randomized Controlled Trial, placebo-controlled, Iran, peer-reviewed, 12 authors, study period 5 April, 2020 - 19 November, 2020, dosage 12000mg days 1-4, trial IRCT20190917044805N2.
Contact: sadeghi_k@tums.ac.ir.
High-dose intravenous Vitamin C in early stages of severe acute respiratory syndrome coronavirus 2 infection: A double-blind, randomized, controlled clinical trial
Journal of Research in Pharmacy Practice, doi:10.4103/jrpp.jrpp_30_22
Objective: Based on previous studies in the sepsis population, Vitamin C could prevent injuries when administered in high doses and before the damage is established. This study aimed to evaluate the protective potentials of high-dose Vitamin C in the progression of coronavirus disease 2019 (COVID-19). Methods: A double-blind, placebo-controlled clinical trial was conducted. Patients with moderate-to-severe disease severity based on the World Health Organization definition were enrolled and received 12 g/d Vitamin C (high-dose intravenous Vitamin C [HDIVC]) or placebo for 4 days. Sequential Organ Failure Assessment (SOFA) score as a primary outcome, National Early Warning Score, Ordinal Scale of Clinical Improvement, and cytokine storm biomarkers were recorded on days 0, 3, and 5. Survival was also assessed on day 28 after enrollment. Findings: Seventy-four patients (37 patients in each group) were enrolled from April 5, 2020, to November 19, 2020, and all patients completed follow-up. A lower increase in SOFA score during the first 3 days of treatment (+0.026 vs. +0.204) and a higher decrease in this parameter in the last 2 days (−0.462 vs. −0.036) were observed in the treatment group. However, these differences did not reach a significance level (P = 0.57 and 0.12, respectively). Other indices of clinical and biological improvement, length of hospitalization, and intensive care unit admission days were the same between the two groups. Treatment did not affect the 28-day mortality. Conclusion: Among patients with moderate-to-severe disease of COVID-19, the use of HDIVC plus standard care resulted in no significant difference in SOFA score or 28-day mortality compared to the standard care alone.
Authors' Contribution Z. Labbani-Motlagh, R. Aliannejad, A. Sadeghi, M. Jafary, and M. Talaschian: Acquisition of clinical data and patient's diagnosis and treatment, interpretation of data, drafting the article, and final approval of the article. G. Shafiee and R. Heshmat: Acquisition of data, the conception and design of the study, analysis and interpretation of data, drafting the article, and final approval of the article. S. Amini, M. Akhtari, A. Jamshidi, M. Mahmoudi, and K. Sadeghi: The conception and design of the study, interpretation of data, revising the article critically for important intellectual content, and final approval of the article.
Financial support and sponsorship This study was supported by a grant from the Deputy of Research, Tehran University of Medical Sciences (Grant No. 99-1-96-47169).
Conflicts of interest There are no conflicts of interest.
References
Abobaker, Alzwi, Alraied, Overview of the possible role of vitamin C in management of COVID-19, Pharmacol Rep
Ackermann, Verleden, Kuehnel, Haverich, Welte et al., Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in COVID-19, N Engl J Med
Anderson, Intravenous Ascorbic Acid (IVAA) for COVID-19: Supportive treatment in hospitalized COVID-W19 patients
Barness, Safety considerations with high ascorbic acid dosage, Ann N Y Acad Sci
Brant, Angus, Is high-dose vitamin C beneficial for patients with sepsis?, JAMA
Carr, Wohlrab, Young, Bellomo, Stability of intravenous vitamin C solutions: A technical report, Crit Care Resusc
Chan, Kok, Zhu, Chu, To et al., Genomic characterization of the 2019 novel human-pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan, Emerg Microbes Infect
Cheng, Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)?, Med Drug Discov
De Grooth, Manubulu-Choo, Zandvliet, Spoelstra-De Man, Girbes et al., Vitamin C Pharmacokinetics in Critically Ill Patients: A Randomized Trial of Four IV Regimens, Chest
Ferrando, Suarez-Sipmann, Mellado-Artigas, Hernández, Gea et al., Clinical features, ventilatory management, and outcome of ARDS caused by COVID-19 are similar to other causes of ARDS, Intensive Care Med
Fisher, Kraskauskas, Martin, Farkas, Wegelin et al., Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid, Am J Physiol Lung Cell Mol Physiol
Fisher, Seropian, Kraskauskas, Thakkar, Voelkel et al., Ascorbic acid attenuates lipopolysaccharide-induced acute lung injury, Crit Care Med
Fowler Aa 3 Rd, Syed, Knowlson, Sculthorpe, Farthing et al., Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis, J Transl Med
Fowler Aa 3 Rd, Truwit, Hite, Morris, Dewilde et al., Effect of vitamin c infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: The CITRIS-ALI randomized clinical trial, JAMA
Gao, Xu, Wang, Lv, Ma et al., The efficiency and safety of high-dose vitamin C in patients with COVID-19: A retrospective cohort study, Aging
Gattinoni, Chiumello, Rossi, COVID-19 pneumonia: ARDS or not?, Crit Care
Greenhalgh, Treadwell, Burrow, Roberts, NEWS (or NEWS2) Score When Assessing Possible COVID-19 Patients in Primary Care
Hemilä, Carr, Chalker, Vitamin C may increase the recovery rate of outpatient cases of SARS-CoV-2 infection by 70%: Reanalysis of the COVID A to Z randomized clinical trial, Front Immunol
Hemilä, Louhiala, Vitamin C may affect lung infections, J R Soc Med
Hoffer, Levine, Assouline, Melnychuk, Padayatty et al., Phase I clinical trial of i.v. ascorbic acid in advanced malignancy, Ann Oncol
Jamalimoghadamsiahkali, Zarezade, Koolaji, Seyedalinaghi, Zendehdel et al., Safety and effectiveness of high-dose vitamin C in patients with COVID-19: A randomized open-label clinical trial, Eur J Med Res
Khwaja, KDIGO clinical practice guidelines for acute kidney injury, Nephron Clin Pract
Klok, Boon, Barco, Endres, Geelhoed et al., The Post-COVID-19 Functional Status scale: A tool to measure functional status over time after COVID-19, Eur Respir J
Lo, Mok, High dose vitamin C induced methemoglobinemia and hemolytic anemia in glucose-6-phosphate dehydrogenase deficiency, Am J Emerg Med
Lv, Zhang, Xia, Yu, Zhao, Early use of high-dose vitamin C is beneficial in treatment of sepsis, Ir J Med Sci
Mahmoodpoor, Shadvar, Sanaie, Hadipoor, Pourmoghaddam et al., Effect of Vitamin C on mortality of critically ill patients with severe pneumonia in intensive care unit: A preliminary study, BMC Infect Dis
Milani, Macchi, Guz-Mark, Vitamin C in the treatment of COVID-19, Nutrients
Padayatty, Sun, Wang, Riordan, Hewitt et al., Vitamin C pharmacokinetics: Implications for oral and intravenous use, Ann Intern Med
Patterson, Isales, Fulzele, Low level of Vitamin C and dysregulation of Vitamin C transporter might be involved in the severity of COVID-19 Infection, Aging Dis
Robitaille, Mamer, Miller, Levine, Assouline et al., Oxalic acid excretion after intravenous ascorbic acid administration, Metabolism
Walker, Iazzetta, Law, Kanji, Bolduc et al., Administration of intravenous ascorbic acid-practical considerations for clinicians, Nutrients
Wang, Lin, Lin, Lin, Effects of different ascorbic acid doses on the mortality of critically ill patients: A meta-analysis, Ann Intensive Care
Ye, Wang, Mao, The pathogenesis and treatment of the `Cytokine Storm' in COVID-19, J Infect
Zhang, Rao, Li, Zhu, Liu et al., Pilot trial of high-dose vitamin C in critically ill COVID-19 patients, Ann Intensive Care
DOI record:
{
"DOI": "10.4103/jrpp.jrpp_30_22",
"ISSN": [
"2319-9644"
],
"URL": "http://dx.doi.org/10.4103/jrpp.jrpp_30_22",
"alternative-id": [
"363550"
],
"author": [
{
"affiliation": [],
"family": "Sadeghi",
"given": "Kourosh",
"sequence": "first"
},
{
"affiliation": [],
"family": "Labbani-Motlagh",
"given": "Zohre",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Amini",
"given": "Shahideh",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Aliannejad",
"given": "Rasoul",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sadeghi",
"given": "Anahita",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shafiee",
"given": "Gita",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Heshmat",
"given": "Ramin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jafary",
"given": "Mohamadreza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Talaschian",
"given": "Mona",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Akhtari",
"given": "Maryam",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jamshidi",
"given": "Ahmadreza",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mahmoudi",
"given": "Mahdi",
"sequence": "additional"
}
],
"container-title": "Journal of Research in Pharmacy Practice",
"container-title-short": "J Res Pharm Pract",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
12,
13
]
],
"date-time": "2022-12-13T14:17:26Z",
"timestamp": 1670941046000
},
"deposited": {
"date-parts": [
[
2022,
12,
13
]
],
"date-time": "2022-12-13T14:18:39Z",
"timestamp": 1670941119000
},
"indexed": {
"date-parts": [
[
2022,
12,
14
]
],
"date-time": "2022-12-14T06:38:14Z",
"timestamp": 1670999894061
},
"is-referenced-by-count": 0,
"issue": "2",
"issued": {
"date-parts": [
[
2022
]
]
},
"journal-issue": {
"issue": "2",
"published-print": {
"date-parts": [
[
2022
]
]
}
},
"language": "en",
"member": "2581",
"original-title": [],
"page": "64",
"prefix": "10.4103",
"published": {
"date-parts": [
[
2022
]
]
},
"published-print": {
"date-parts": [
[
2022
]
]
},
"publisher": "Medknow",
"reference": [
{
"DOI": "10.1186/s13613-020-00792-3",
"article-title": "Pilot trial of high-dose vitamin C in critically ill COVID-19 patients",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "5",
"journal-title": "Ann Intensive Care",
"key": "key-10.4103/2319-9644.363550-1",
"year": "2021"
},
{
"DOI": "10.1016/j.jinf.2020.03.037",
"article-title": "The pathogenesis and treatment of the 'Cytokine Storm' in COVID-19",
"author": "Ye",
"doi-asserted-by": "crossref",
"first-page": "607",
"journal-title": "J Infect",
"key": "key-10.4103/2319-9644.363550-2",
"year": "2020"
},
{
"DOI": "10.1007/s00134-020-06192-2",
"article-title": "Clinical features, ventilatory management, and outcome of ARDS caused by COVID-19 are similar to other causes of ARDS",
"author": "Ferrando",
"doi-asserted-by": "crossref",
"first-page": "2200",
"journal-title": "Intensive Care Med",
"key": "key-10.4103/2319-9644.363550-3",
"year": "2020"
},
{
"key": "key-10.4103/2319-9644.363550-4",
"unstructured": "Clinical management of COVID-19: Living guideline, 2022. WHO/2019-nCoV/Clinical/2022.1."
},
{
"DOI": "10.1080/22221751.2020.1719902",
"article-title": "Genomic characterization of the 2019 novel human-pathogenic coronavirus isolated from a patient with atypical pneumonia after visiting Wuhan",
"author": "Chan",
"doi-asserted-by": "crossref",
"first-page": "221",
"journal-title": "Emerg Microbes Infect",
"key": "key-10.4103/2319-9644.363550-5",
"year": "2020"
},
{
"DOI": "10.1007/s43440-020-00176-1",
"article-title": "Overview of the possible role of vitamin C in management of COVID-19",
"author": "Abobaker",
"doi-asserted-by": "crossref",
"first-page": "1517",
"journal-title": "Pharmacol Rep",
"key": "key-10.4103/2319-9644.363550-6",
"year": "2020"
},
{
"DOI": "10.1007/s11845-020-02394-1",
"article-title": "Early use of high-dose vitamin C is beneficial in treatment of sepsis",
"author": "Lv",
"doi-asserted-by": "crossref",
"first-page": "1183",
"journal-title": "Ir J Med Sci",
"key": "key-10.4103/2319-9644.363550-7",
"year": "2021"
},
{
"DOI": "10.14336/AD.2020.0918",
"article-title": "Low level of Vitamin C and dysregulation of Vitamin C transporter might be involved in the severity of COVID-19 Infection",
"author": "Patterson",
"doi-asserted-by": "crossref",
"first-page": "14",
"journal-title": "Aging Dis",
"key": "key-10.4103/2319-9644.363550-8",
"year": "2021"
},
{
"DOI": "10.1186/s13054-020-02880-z",
"article-title": "COVID-19 pneumonia: ARDS or not?",
"author": "Gattinoni",
"doi-asserted-by": "crossref",
"first-page": "154",
"journal-title": "Crit Care",
"key": "key-10.4103/2319-9644.363550-9",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2015432",
"article-title": "Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in COVID-19",
"author": "Ackermann",
"doi-asserted-by": "crossref",
"first-page": "120",
"journal-title": "N Engl J Med",
"key": "key-10.4103/2319-9644.363550-10",
"year": "2020"
},
{
"DOI": "10.1016/j.medidd.2020.100028",
"article-title": "Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)?",
"author": "Cheng",
"doi-asserted-by": "crossref",
"first-page": "100028S",
"journal-title": "Med Drug Discov",
"key": "key-10.4103/2319-9644.363550-11",
"year": "2020"
},
{
"DOI": "10.1097/CCM.0b013e3182120cb8",
"article-title": "Ascorbic acid attenuates lipopolysaccharide-induced acute lung injury",
"author": "Fisher",
"doi-asserted-by": "crossref",
"first-page": "1454",
"journal-title": "Crit Care Med",
"key": "key-10.4103/2319-9644.363550-12",
"year": "2011"
},
{
"DOI": "10.1152/ajplung.00300.2011",
"article-title": "Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid",
"author": "Fisher",
"doi-asserted-by": "crossref",
"first-page": "L20",
"journal-title": "Am J Physiol Lung Cell Mol Physiol",
"key": "key-10.4103/2319-9644.363550-13",
"year": "2012"
},
{
"DOI": "10.1186/1479-5876-12-32",
"article-title": "Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis",
"author": "Fowler",
"doi-asserted-by": "crossref",
"first-page": "32",
"journal-title": "J Transl Med",
"key": "key-10.4103/2319-9644.363550-14",
"year": "2014"
},
{
"DOI": "10.1001/jama.2019.11825",
"article-title": "Effect of vitamin c infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: The CITRIS-ALI randomized clinical trial",
"author": "Fowler",
"doi-asserted-by": "crossref",
"first-page": "1261",
"journal-title": "JAMA",
"key": "key-10.4103/2319-9644.363550-15",
"year": "2019"
},
{
"DOI": "10.1186/s40001-021-00490-1",
"article-title": "Safety and effectiveness of high-dose vitamin C in patients with COVID-19: A randomized open-label clinical trial",
"author": "JamaliMoghadamSiahkali",
"doi-asserted-by": "crossref",
"first-page": "20",
"journal-title": "Eur J Med Res",
"key": "key-10.4103/2319-9644.363550-16",
"year": "2021"
},
{
"DOI": "10.3389/fimmu.2021.674681",
"article-title": "Vitamin C may increase the recovery rate of outpatient cases of SARS-CoV-2 infection by 70%: Reanalysis of the COVID A to Z randomized clinical trial",
"author": "Hemilä",
"doi-asserted-by": "crossref",
"first-page": "674681",
"journal-title": "Front Immunol",
"key": "key-10.4103/2319-9644.363550-17",
"year": "2021"
},
{
"article-title": "Stability of intravenous vitamin C solutions: A technical report",
"author": "Carr",
"first-page": "180",
"journal-title": "Crit Care Resusc",
"key": "key-10.4103/2319-9644.363550-18",
"year": "2018"
},
{
"DOI": "10.3390/nu11091994",
"article-title": "Administration of intravenous ascorbic acid-practical considerations for clinicians",
"author": "Walker",
"doi-asserted-by": "crossref",
"first-page": "E1994",
"journal-title": "Nutrients",
"key": "key-10.4103/2319-9644.363550-19",
"year": "2019"
},
{
"DOI": "10.1111/j.1749-6632.1975.tb29311.x",
"article-title": "Safety considerations with high ascorbic acid dosage",
"author": "Barness",
"doi-asserted-by": "crossref",
"first-page": "523",
"journal-title": "Ann N Y Acad Sci",
"key": "key-10.4103/2319-9644.363550-20",
"year": "1975"
},
{
"DOI": "10.1093/annonc/mdn377",
"doi-asserted-by": "crossref",
"key": "key-10.4103/2319-9644.363550-21",
"unstructured": "Hoffer LJ, Levine M, Assouline S, Melnychuk D, Padayatty SJ, Rosadiuk K, et al. Phase I clinical trial of i.v. ascorbic acid in advanced malignancy. Ann Oncol 2008;19:1969-74."
},
{
"DOI": "10.1016/j.ajem.2020.05.099",
"doi-asserted-by": "crossref",
"key": "key-10.4103/2319-9644.363550-22",
"unstructured": "Lo YH, Mok KL. High dose vitamin C induced methemoglobinemia and hemolytic anemia in glucose-6-phosphate dehydrogenase deficiency. Am J Emerg Med 2020;38:2488.e3- 2488.e5."
},
{
"DOI": "10.1016/j.metabol.2008.09.023",
"article-title": ", Levine M, Assouline S, Melnychuk D, et al.Oxalic acid excretion after intravenous ascorbic acid administration",
"author": "Robitaille",
"doi-asserted-by": "crossref",
"first-page": "263",
"journal-title": "Metabolism",
"key": "key-10.4103/2319-9644.363550-23",
"year": "2009"
},
{
"key": "key-10.4103/2319-9644.363550-24",
"unstructured": "Greenhalgh T, Treadwell J, Burrow R, Roberts N. NEWS (or NEWS2) Score When Assessing Possible COVID-19 Patients in Primary Care. Centre for Evidence-Based Medicine, Nuffield Department of Primary Care Health Sciences, University of Oxford; 2020. p. 20."
},
{
"DOI": "10.1183/13993003.01494-2020",
"article-title": "The Post-COVID-19 Functional Status scale: A tool to measure functional status over time after COVID-19",
"author": "Klok",
"doi-asserted-by": "crossref",
"first-page": "2001494",
"journal-title": "Eur Respir J",
"key": "key-10.4103/2319-9644.363550-25",
"year": "2020"
},
{
"DOI": "10.1159/000339789",
"article-title": "KDIGO clinical practice guidelines for acute kidney injury",
"author": "Khwaja",
"doi-asserted-by": "crossref",
"first-page": "c179",
"journal-title": "Nephron Clin Pract",
"key": "key-10.4103/2319-9644.363550-26",
"year": "2012"
},
{
"DOI": "10.3390/nu13041172",
"article-title": "Vitamin C in the treatment of COVID-19",
"author": "Milani",
"doi-asserted-by": "crossref",
"first-page": "1172",
"journal-title": "Nutrients",
"key": "key-10.4103/2319-9644.363550-27",
"year": "2021"
},
{
"DOI": "10.1001/jama.2019.11643",
"article-title": "Is high-dose vitamin C beneficial for patients with sepsis?",
"author": "Brant",
"doi-asserted-by": "crossref",
"first-page": "1257",
"journal-title": "JAMA",
"key": "key-10.4103/2319-9644.363550-28",
"year": "2019"
},
{
"DOI": "10.1186/s13613-019-0532-9",
"article-title": "Effects of different ascorbic acid doses on the mortality of critically ill patients: A meta-analysis",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "58",
"journal-title": "Ann Intensive Care",
"key": "key-10.4103/2319-9644.363550-29",
"year": "2019"
},
{
"DOI": "10.1186/s12879-021-06288-0",
"article-title": "Effect of Vitamin C on mortality of critically ill patients with severe pneumonia in intensive care unit: A preliminary study",
"author": "Mahmoodpoor",
"doi-asserted-by": "crossref",
"first-page": "616",
"journal-title": "BMC Infect Dis",
"key": "key-10.4103/2319-9644.363550-30",
"year": "2021"
},
{
"DOI": "10.18632/aging.202557",
"article-title": "The efficiency and safety of high-dose vitamin C in patients with COVID-19: A retrospective cohort study",
"author": "Gao",
"doi-asserted-by": "crossref",
"first-page": "7020",
"journal-title": "Aging (Albany NY)",
"key": "key-10.4103/2319-9644.363550-31",
"year": "2021"
},
{
"DOI": "10.7326/0003-4819-140-7-200404060-00010",
"article-title": "Vitamin C pharmacokinetics: Implications for oral and intravenous use",
"author": "Padayatty",
"doi-asserted-by": "crossref",
"first-page": "533",
"journal-title": "Ann Intern Med",
"key": "key-10.4103/2319-9644.363550-32",
"year": "2004"
},
{
"article-title": "Vitamin C Pharmacokinetics in Critically Ill Patients: A Randomized Trial of Four IV Regimens",
"author": "de",
"first-page": "1368",
"journal-title": "Chest",
"key": "key-10.4103/2319-9644.363550-33",
"year": "2018"
},
{
"DOI": "10.1177/014107680710001109",
"article-title": "Vitamin C may affect lung infections",
"author": "Hemilä",
"doi-asserted-by": "crossref",
"first-page": "495",
"journal-title": "J R Soc Med",
"key": "key-10.4103/2319-9644.363550-34",
"year": "2007"
},
{
"key": "key-10.4103/2319-9644.363550-35",
"unstructured": "Anderson PS. Intravenous Ascorbic Acid (IVAA) for COVID-19: Supportive treatment in hospitalized COVID-W19 patients. In: Based on Use in China and US Settings. Pan America Health Organization; 2020."
}
],
"reference-count": 35,
"references-count": 35,
"relation": {},
"resource": {
"primary": {
"URL": "http://www.jrpp.net/text.asp?2022/11/2/64/363550"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Pharmacology (medical)",
"General Pharmacology, Toxicology and Pharmaceutics",
"Pharmacy"
],
"subtitle": [],
"title": "High-dose intravenous Vitamin C in early stages of severe acute respiratory syndrome coronavirus 2 infection: A double-blind, randomized, controlled clinical trial",
"type": "journal-article",
"volume": "11"
}