Individualizing Risk Prediction for Positive Coronavirus Disease 2019 Testing
et al., Chest, doi:10.1016/j.chest.2020.05.580, Jun 2020
Melatonin for COVID-19
25th treatment shown to reduce risk in
May 2021, now with p = 0.000000015 from 18 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
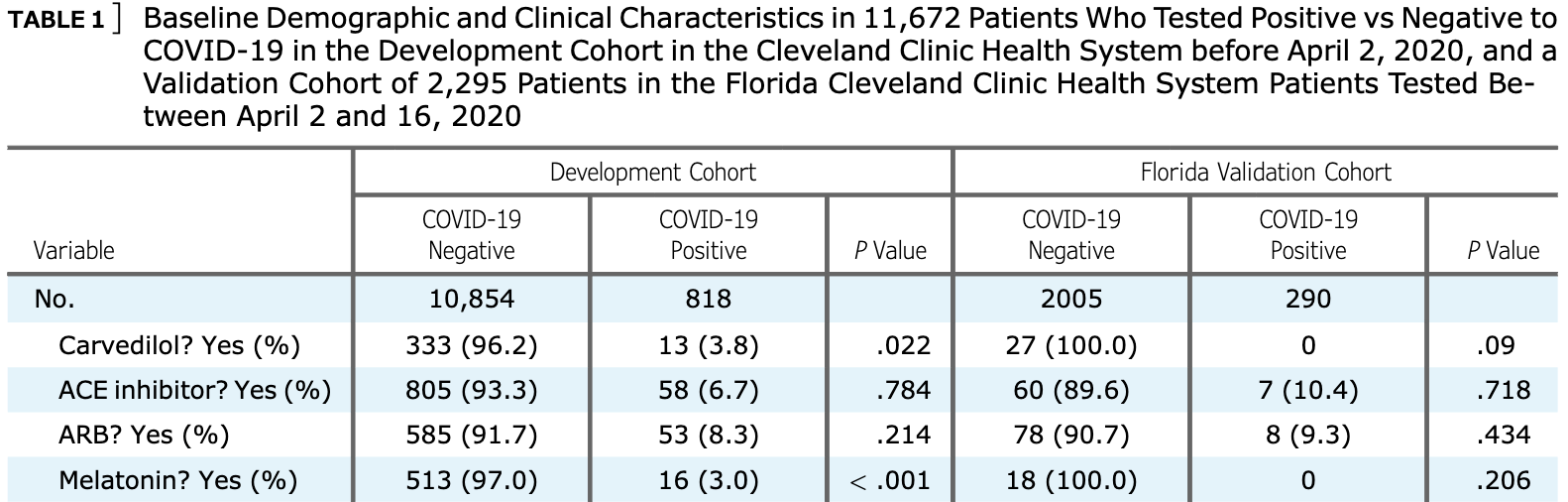
Retrospective 11,672 patients tested for COVID-19 with 818 testing positive, showing significantly lower risk with melatonin use.
Standard of Care (SOC) for COVID-19 in the study country,
the USA, is very poor with very low average efficacy for approved treatments1.
Only expensive, high-profit treatments were approved for early treatment. Low-cost treatments were excluded, reducing the probability of early treatment due to access and cost barriers, and eliminating complementary and synergistic benefits seen with many low-cost treatments.
|
risk of case, 58.0% lower, RR 0.42, p < 0.001, treatment 16 of 529 (3.0%), control 802 of 11,143 (7.2%), NNT 24, development cohort.
|
|
risk of case, 99.7% lower, RR 0.003, p = 0.09, treatment 0 of 18 (0.0%), control 290 of 2,005 (14.5%), NNT 6.9, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm), Florida validation cohort.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Jehi et al., 10 Jun 2020, retrospective, USA, peer-reviewed, 8 authors.
Individualizing Risk Prediction for Positive Coronavirus Disease 2019 Testing
Chest, doi:10.1016/j.chest.2020.05.580
BACKGROUND: Coronavirus disease 2019 (COVID-19) is sweeping the globe. Despite multiple case-series, actionable knowledge to tailor decision-making proactively is missing. RESEARCH QUESTION: Can a statistical model accurately predict infection with COVID-19? STUDY DESIGN AND METHODS: We developed a prospective registry of all patients tested for COVID-19 in Cleveland Clinic to create individualized risk prediction models. We focus here on the likelihood of a positive nasal or oropharyngeal COVID-19 test. A least absolute shrinkage and selection operator logistic regression algorithm was constructed that removed variables that were not contributing to the model's cross-validated concordance index. After external validation in a temporally and geographically distinct cohort, the statistical prediction model was illustrated as a nomogram and deployed in an online risk calculator. RESULTS: In the development cohort, 11,672 patients fulfilled study criteria, including 818 patients (7.0%) who tested positive for COVID-19; in the validation cohort, 2295 patients fulfilled criteria, including 290 patients who tested positive for COVID-19. Male, African American, older patients, and those with known COVID-19 exposure were at higher risk of being positive for COVID-19. Risk was reduced in those who had pneumococcal polysaccharide or influenza vaccine or who were on melatonin, paroxetine, or carvedilol. Our model had favorable discrimination (c-statistic ¼ 0.863 in the development cohort and 0.840 in the validation cohort) and calibration. We present sensitivity, specificity, negative predictive value, and positive predictive value at different prediction cutoff points. The calculator is freely available at https://riskcalc.org/COVID19. INTERPRETATION: Prediction of a COVID-19 positive test is possible and could help direct health-care resources. We demonstrate relevance of age, race, sex, and socioeconomic characteristics in COVID-19 susceptibility and suggest a potential modifying role of certain common vaccinations and drugs that have been identified in drug-repurposing studies.
Author contributions: L. J. is the guarantor of submission and participated in literature search, figures, study design, data collection, data interpretation, and writing; X. J. participated in data analysis and figures; A. M. participated in data collection and data analysis; S. E. participated in data interpretation, study design, and writing; B. P. R., S. G., and J. B. Y. participated in data interpretation and writing; and M. W. K. participated in literature search, study design, data interpretation, data analysis, and writing. Financial/nonfinancial disclosures: The authors have reported to CHEST the following: A. M. reports grants from Novo Nordisk, Boehringer Ingelheim, Merck, Novartis, and National Institutes of Health (NIH), outside the submitted work. M. W. K. reports grants from Novo Nordisk, Boehringer Ingelheim, Merck, Novartis, and NIH, consulting for Stratify Genomics and RenatlyxAI, outside the submitted work. None declared (L. J., X. J., S. E., B. P. R., S. G., J. B. Y.).
Role of sponsors: The sponsor had no role in the design of the study, the collection and analysis of the data, or the preparation of the manuscript. Additional information: The e-Appendix and e-Figure can be found in the Supplemental Materials section of the online article.
References
Chen, Zhou, Dong, Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study, Lancet
Goff, Hayashi, Martínez-Gil, Synthetic toll-like receptor 4 (TLR4) and TLR7 ligands as influenza virus vaccine adjuvants induce rapid, sustained, and broadly protective responses, J Virol
Harrell, Califf, Pryor, Lee, Rosati, Evaluating the yield of medical tests, JAMA
Harris, Taylor, Minor, The REDCap consortium: building an international community of software partners, J Biomed Inform
Harris, Taylor, Thielke, Payne, Gonzalez et al., Research electronic data capture (REDCap): a metadata-driven methodology and workflow process for providing translational research informatics support, J Biomed Inform
Iqr ¼ Interquartile Range ; Callaway, Cyranoski, Mallapaty, Stoye, Tollefson, The coronavirus pandemic in five powerful charts, Nature
Jin, Lian, Hu, Epidemiological, clinical and virological characteristics of 74 cases of coronavirusinfected disease 2019 (COVID-19) with gastrointestinal symptoms, Gut
Kattan, Gerds, The index of prediction accuracy: an intuitive measure useful for evaluating risk prediction models, Diagn Progn Res
Kattan, Nomograms: introduction, Semin Urol Oncol
Lipsitch, Swerdlow, Finelli, Defining the epidemiology of Covid-19 -studies needed, N Engl J Med
Milinovich, Kattan, Extracting and utilizing electronic health data from Epic for research, Ann Transl Med
Ogbadu, Singh, Gupta, Mehra, Sen, Ageing reduces angiotensin II type 1 receptor antagonism mediated pre-conditioning effects in ischemic kidneys by inducing oxidative and inflammatory stress, Exp Gerontol
Pretesting, dL Missing
Sharfstein, Becker, Mello, Diagnostic testing for the novel coronavirus, JAMA
Shi, Yu, Zhao, Wang, Zhao et al., Host susceptibility to severe COVID-19 and establishment of a host risk score: findings of 487 cases outside Wuhan, Crit Care
Steyerberg, Vickers, Cook, Assessing the performance of prediction models: a framework for traditional and novel measures, Epidemiology
Sun, Koh, Marimuthu, Epidemiological and clinical predictors of COVID-19, Clin Infect Dis
Wang, Xu, Gao, SARS-CoV-2 in different types of clinical specimens, JAMA
Wang, Yang, Li, Wen, Zhang, Clinical features of 69 cases with coronavirus disease 2019 in Wuhan, China, Clin Infect Dis
Weiss, Murdoch, Clinical course and mortality risk of severe COVID-19, Lancet
Wu, Chen, Cai, Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China, JAMA Intern Med
Wu, Li, Wang, Xiu, Zhang, Carvedilol inhibits angiotensin ii-induced proliferation and contraction in hepatic stellate cells through the RhoA/Rho-Kinase pathway, Biomed Res Int
Yang, Yu, Xu, Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study [published correction appears, Lancet Respir Med
Yoon, Kim, Kim, Ageassociated changes in the vascular reninangiotensin system in mice, Oxide Med Cell Longev
Zhang, Dong, Cao, Clinical characteristics of 140 patients infected with SARS-CoV-2 in
Zhou, Hou, Shen, Networkbased drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2, Cell Discov
Zhou, Yu, Du, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study [published correction appears, Lancet
DOI record:
{
"DOI": "10.1016/j.chest.2020.05.580",
"ISSN": [
"0012-3692"
],
"URL": "http://dx.doi.org/10.1016/j.chest.2020.05.580",
"alternative-id": [
"S0012369220316548"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Individualizing Risk Prediction for Positive Coronavirus Disease 2019 Testing"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Chest"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.chest.2020.05.580"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2020 American College of Chest Physicians. Published by Elsevier Inc. All rights reserved."
}
],
"author": [
{
"affiliation": [],
"family": "Jehi",
"given": "Lara",
"sequence": "first"
},
{
"affiliation": [],
"family": "Ji",
"given": "Xinge",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Milinovich",
"given": "Alex",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Erzurum",
"given": "Serpil",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rubin",
"given": "Brian P.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gordon",
"given": "Steve",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Young",
"given": "James B.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kattan",
"given": "Michael W.",
"sequence": "additional"
}
],
"container-title": "Chest",
"container-title-short": "Chest",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.fr",
"clinicalkey.jp",
"clinicalkey.com.au",
"clinicalkey.es",
"clinicalkey.com",
"journal.chestnet.org",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2020,
6,
10
]
],
"date-time": "2020-06-10T16:39:33Z",
"timestamp": 1591807173000
},
"deposited": {
"date-parts": [
[
2022,
11,
16
]
],
"date-time": "2022-11-16T04:23:43Z",
"timestamp": 1668572623000
},
"funder": [
{
"DOI": "10.13039/100006108",
"award": [
"UL1TR002548"
],
"doi-asserted-by": "publisher",
"name": "National Center for Advancing Translational Sciences"
},
{
"DOI": "10.13039/100001003",
"doi-asserted-by": "publisher",
"name": "Boehringer Ingelheim"
},
{
"DOI": "10.13039/100007311",
"doi-asserted-by": "publisher",
"name": "Cleveland Clinic"
},
{
"DOI": "10.13039/100004334",
"doi-asserted-by": "publisher",
"name": "Merck"
},
{
"DOI": "10.13039/501100004191",
"doi-asserted-by": "publisher",
"name": "Novo Nordisk"
},
{
"DOI": "10.13039/100000002",
"doi-asserted-by": "publisher",
"name": "National Institutes of Health"
},
{
"DOI": "10.13039/100004336",
"doi-asserted-by": "publisher",
"name": "Novartis"
}
],
"indexed": {
"date-parts": [
[
2023,
12,
21
]
],
"date-time": "2023-12-21T17:05:47Z",
"timestamp": 1703178347971
},
"is-referenced-by-count": 134,
"issue": "4",
"issued": {
"date-parts": [
[
2020,
10
]
]
},
"journal-issue": {
"issue": "4",
"published-print": {
"date-parts": [
[
2020,
10
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2020,
10,
1
]
],
"date-time": "2020-10-01T00:00:00Z",
"timestamp": 1601510400000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0012369220316548?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0012369220316548?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "1364-1375",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2020,
10
]
]
},
"published-print": {
"date-parts": [
[
2020,
10
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"author": "Callaway",
"key": "10.1016/j.chest.2020.05.580_bib1"
},
{
"DOI": "10.1001/jama.2020.3864",
"article-title": "Diagnostic testing for the novel coronavirus",
"author": "Sharfstein",
"doi-asserted-by": "crossref",
"first-page": "1437",
"issue": "15",
"journal-title": "JAMA",
"key": "10.1016/j.chest.2020.05.580_bib2",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1056/NEJMp2002125",
"article-title": "Defining the epidemiology of Covid-19 - studies needed.",
"author": "Lipsitch",
"doi-asserted-by": "crossref",
"first-page": "1194",
"issue": "13",
"journal-title": "N Engl J Med",
"key": "10.1016/j.chest.2020.05.580_bib3",
"volume": "382",
"year": "2020"
},
{
"article-title": "SARS-CoV-2 in different types of clinical specimens",
"author": "Wang",
"first-page": "1843",
"issue": "18",
"journal-title": "JAMA",
"key": "10.1016/j.chest.2020.05.580_bib4",
"volume": "323",
"year": "2020"
},
{
"article-title": "Nomograms: introduction",
"author": "Kattan",
"first-page": "79",
"issue": "2",
"journal-title": "Semin Urol Oncol",
"key": "10.1016/j.chest.2020.05.580_bib5",
"volume": "20",
"year": "2002"
},
{
"DOI": "10.1038/s41421-020-0153-3",
"article-title": "Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "14",
"journal-title": "Cell Discov",
"key": "10.1016/j.chest.2020.05.580_bib6",
"volume": "6",
"year": "2020"
},
{
"DOI": "10.21037/atm.2018.01.13",
"article-title": "Extracting and utilizing electronic health data from Epic for research",
"author": "Milinovich",
"doi-asserted-by": "crossref",
"first-page": "42",
"issue": "3",
"journal-title": "Ann Transl Med",
"key": "10.1016/j.chest.2020.05.580_bib7",
"volume": "6",
"year": "2018"
},
{
"DOI": "10.1016/j.jbi.2008.08.010",
"article-title": "Research electronic data capture (REDCap): a metadata-driven methodology and workflow process for providing translational research informatics support",
"author": "Harris",
"doi-asserted-by": "crossref",
"first-page": "377",
"issue": "2",
"journal-title": "J Biomed Inform",
"key": "10.1016/j.chest.2020.05.580_bib8",
"volume": "42",
"year": "2009"
},
{
"DOI": "10.1016/j.jbi.2019.103208",
"doi-asserted-by": "crossref",
"key": "10.1016/j.chest.2020.05.580_bib9",
"unstructured": "Harris PA, Taylor R, Minor BL, et al. The REDCap consortium: building an international community of software partners, J Biomed Inform. 2019;95:103208."
},
{
"DOI": "10.1001/jama.1982.03320430047030",
"article-title": "Evaluating the yield of medical tests",
"author": "Harrell",
"doi-asserted-by": "crossref",
"first-page": "2543",
"issue": "18",
"journal-title": "JAMA",
"key": "10.1016/j.chest.2020.05.580_bib10",
"volume": "247",
"year": "1982"
},
{
"DOI": "10.1186/s41512-018-0029-2",
"article-title": "The index of prediction accuracy: an intuitive measure useful for evaluating risk prediction models",
"author": "Kattan",
"doi-asserted-by": "crossref",
"first-page": "7",
"journal-title": "Diagn Progn Res",
"key": "10.1016/j.chest.2020.05.580_bib11",
"volume": "2",
"year": "2018"
},
{
"DOI": "10.1097/EDE.0b013e3181c30fb2",
"article-title": "Assessing the performance of prediction models: a framework for traditional and novel measures",
"author": "Steyerberg",
"doi-asserted-by": "crossref",
"first-page": "128",
"issue": "1",
"journal-title": "Epidemiology",
"key": "10.1016/j.chest.2020.05.580_bib12",
"volume": "21",
"year": "2010"
},
{
"DOI": "10.1136/gutjnl-2020-320926",
"article-title": "Epidemiological, clinical and virological characteristics of 74 cases of coronavirus-infected disease 2019 (COVID-19) with gastrointestinal symptoms",
"author": "Jin",
"doi-asserted-by": "crossref",
"first-page": "1002",
"issue": "6",
"journal-title": "Gut",
"key": "10.1016/j.chest.2020.05.580_bib14",
"volume": "69",
"year": "2020"
},
{
"key": "10.1016/j.chest.2020.05.580_bib15",
"unstructured": "Sun Y, Koh V, Marimuthu K, et al. Epidemiological and clinical predictors of COVID-19. Clin Infect Dis. 2020;ciaa322."
},
{
"DOI": "10.1016/S0140-6736(20)30633-4",
"article-title": "Clinical course and mortality risk of severe COVID-19",
"author": "Weiss",
"doi-asserted-by": "crossref",
"first-page": "1014",
"issue": "10229",
"journal-title": "Lancet",
"key": "10.1016/j.chest.2020.05.580_bib16",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1186/s13054-020-2833-7",
"article-title": "Host susceptibility to severe COVID-19 and establishment of a host risk score: findings of 487 cases outside Wuhan",
"author": "Shi",
"doi-asserted-by": "crossref",
"first-page": "108",
"issue": "1",
"journal-title": "Crit Care",
"key": "10.1016/j.chest.2020.05.580_bib17",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1093/cid/ciaa272",
"doi-asserted-by": "crossref",
"key": "10.1016/j.chest.2020.05.580_bib18",
"unstructured": "Wang Z, Yang B, Li Q, Wen L, Zhang R. Clinical features of 69 cases with coronavirus disease 2019 in Wuhan, China. Clin Infect Dis. 2020;ciaa272."
},
{
"DOI": "10.1016/S0140-6736(20)30566-3",
"doi-asserted-by": "crossref",
"key": "10.1016/j.chest.2020.05.580_bib19",
"unstructured": "Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study [published correction appears in Lancet. 2020 Mar 28;395(10229):1038]. Lancet. 2020;395(10229):1054-1062."
},
{
"key": "10.1016/j.chest.2020.05.580_bib20",
"unstructured": "Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;e200994."
},
{
"DOI": "10.1016/S2213-2600(20)30079-5",
"doi-asserted-by": "crossref",
"key": "10.1016/j.chest.2020.05.580_bib21",
"unstructured": "Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study [published correction appears in Lancet Respir Med. 2020 Apr;8(4):e26]. Lancet Respir Med. 2020;8(5):475-481."
},
{
"key": "10.1016/j.chest.2020.05.580_bib22",
"unstructured": "Zhang JJ, Dong X, Cao YY, et al. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy. In press."
},
{
"DOI": "10.1016/S0140-6736(20)30211-7",
"article-title": "Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study",
"author": "Chen",
"doi-asserted-by": "crossref",
"first-page": "507",
"issue": "10223",
"journal-title": "Lancet",
"key": "10.1016/j.chest.2020.05.580_bib23",
"volume": "395",
"year": "2020"
},
{
"key": "10.1016/j.chest.2020.05.580_bib24",
"unstructured": "World Health Organization. http://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/weekly-surveillance-report. Accessed March 28, 2020."
},
{
"article-title": "Age-associated changes in the vascular renin-angiotensin system in mice",
"author": "Yoon",
"first-page": "6731093",
"journal-title": "Oxide Med Cell Longev",
"key": "10.1016/j.chest.2020.05.580_bib25",
"volume": "2016",
"year": "2016"
},
{
"DOI": "10.1016/j.exger.2020.110892",
"article-title": "Ageing reduces angiotensin II type 1 receptor antagonism mediated pre-conditioning effects in ischemic kidneys by inducing oxidative and inflammatory stress",
"author": "Ogbadu",
"doi-asserted-by": "crossref",
"first-page": "110892",
"journal-title": "Exp Gerontol",
"key": "10.1016/j.chest.2020.05.580_bib26",
"volume": "135",
"year": "2020"
},
{
"DOI": "10.1128/JVI.03337-14",
"article-title": "Synthetic toll-like receptor 4 (TLR4) and TLR7 ligands as influenza virus vaccine adjuvants induce rapid, sustained, and broadly protective responses",
"author": "Goff",
"doi-asserted-by": "crossref",
"first-page": "3221",
"issue": "6",
"journal-title": "J Virol",
"key": "10.1016/j.chest.2020.05.580_bib27",
"volume": "89",
"year": "2015"
},
{
"DOI": "10.1155/2019/7932046",
"article-title": "Carvedilol inhibits angiotensin ii-induced proliferation and contraction in hepatic stellate cells through the RhoA/Rho-Kinase pathway",
"author": "Wu",
"doi-asserted-by": "crossref",
"first-page": "7932046",
"journal-title": "Biomed Res Int",
"key": "10.1016/j.chest.2020.05.580_bib28",
"volume": "2019",
"year": "2019"
}
],
"reference-count": 27,
"references-count": 27,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S0012369220316548"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Cardiology and Cardiovascular Medicine",
"Critical Care and Intensive Care Medicine",
"Pulmonary and Respiratory Medicine"
],
"subtitle": [],
"title": "Individualizing Risk Prediction for Positive Coronavirus Disease 2019 Testing",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "158"
}
