Local budesonide therapy in the management of persistent hyposmia in suspected non-severe COVID-19 patients: Results of a randomized controlled trial
et al., International Journal of Infectious Diseases, doi:10.1016/j.ijid.2023.08.022, COVIDORL, NCT04361474, Aug 2023
Budesonide for COVID-19
27th treatment shown to reduce risk in
September 2021, now with p = 0.000003 from 15 studies, recognized in 10 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
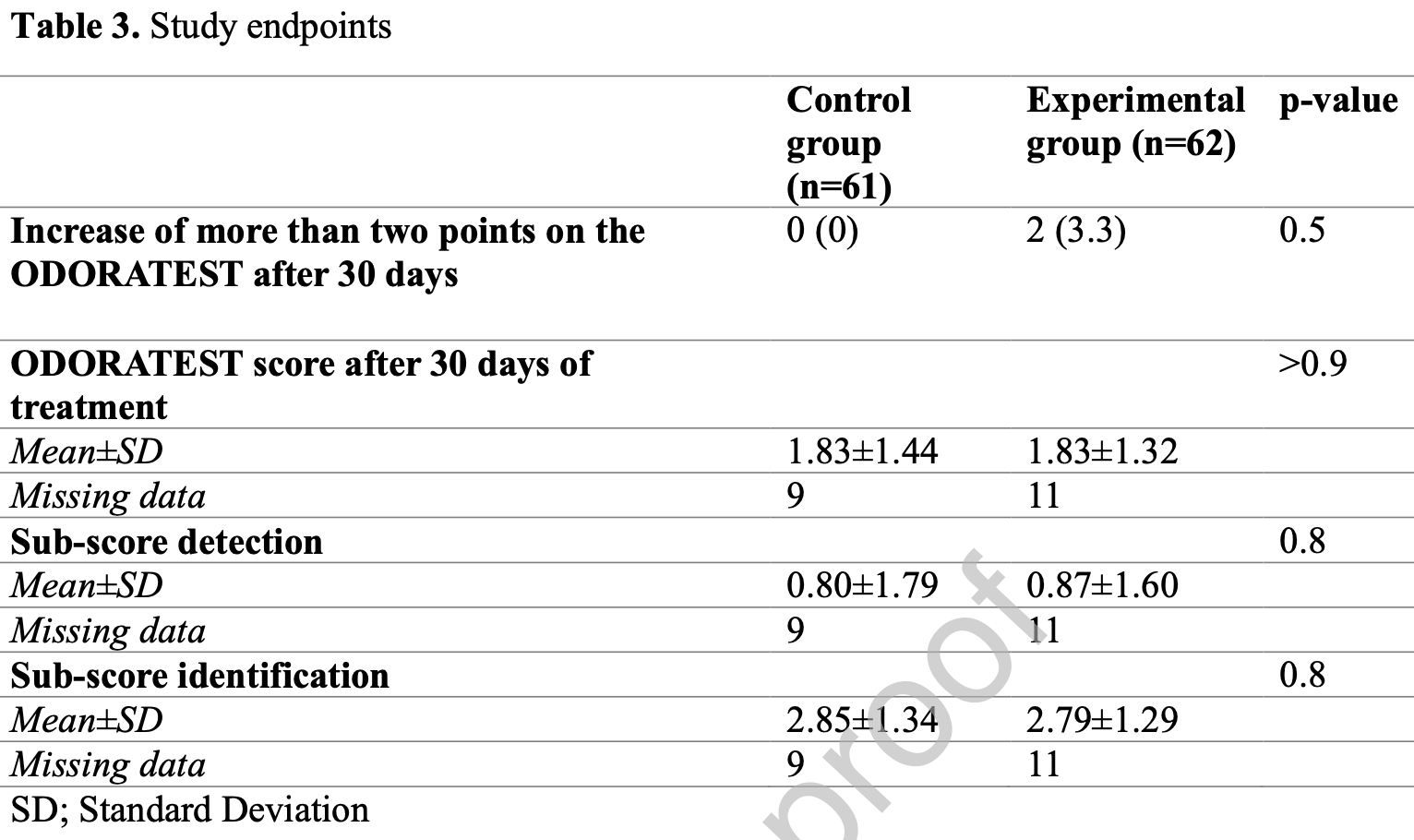
RCT 123 post-COVID-19 hyposmia patients, showing no significant difference in smell recovery with budesonide. 2 patients experienced improvement of more than two points on the ODORATEST score compared with 0 in the control group.
Hautefort et al., 31 Aug 2023, Randomized Controlled Trial, France, peer-reviewed, 16 authors, trial NCT04361474 (history) (COVIDORL).
Contact: mdaval@for.paris.
Local budesonide therapy in the management of persistent hyposmia in suspected non-severe COVID-19 patients: Results of a randomized controlled trial
International Journal of Infectious Diseases, doi:10.1016/j.ijid.2023.08.022
This is a PDF file of an article that has undergone enhancements after acceptance, such as the addition of a cover page and metadata, and formatting for readability, but it is not yet the definitive version of record. This version will undergo additional copyediting, typesetting and review before it is published in its final form, but we are providing this version to give early visibility of the article. Please note that, during the production process, errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of interests: The authors declare that they had no competing interests in relation to this study.
Funding: The study was funded by the French Department of Health (Innovarc). Data sharing: Each investigator will have access to the final trial dataset. Project data sets are housed on the Project Accept Web site (Clinfile). All data sets are password protected. Project Principal Investigators have direct access to their own site's data sets. To ensure confidentiality, data provided to project team members will be anonymized. Data will be made available from the author upon reasonable request (mdaval@for.paris). The authors declare that they had no competing interests in relation to this study.
Ethical approval:
References
Abdelalim, Mohamady, Elsayed, Elawady, Ghallab, Corticosteroid nasal spray for recovery of smell sensation in COVID-19 patients: A randomized controlled trial, American Journal of Otolaryngology, doi:10.1016/j.amjoto.2020.102884
Abdelazim, Abdelazim, Effect of Sodium Gluconate on Decreasing Elevated Nasal Calcium and Improving Olfactory Function Post COVID-19 Infection, Am J Rhinol Allergy, doi:10.1177/19458924221120116
Abdelazim, Abdelazim, Ismaiel, Alsobky, Younes et al., Effect of intra-nasal nitrilotriacetic acid trisodium salt in lowering elevated calcium cations and improving olfactory dysfunction in COVID-19 patients, Eur Arch Otorhinolaryngol, doi:10.1007/s00405-022-07424-5
Abdelazim, Abdelazim, Moneir, The effect of intra-nasal tetra sodium pyrophosphate on decreasing elevated nasal calcium and improving olfactory function post COVID-19: a randomized controlled trial, Allergy Asthma Clin Immunol, doi:10.1186/s13223-022-00711-0
Abdelazim, Mandour, Abdelazim, Ismaiel, Gamal et al., Intra Nasal Use of Ethylene Diamine Tetra Acetic Acid for Improving Olfactory Dysfunction Post COVID-19, Am J Rhinol Allergy, doi:10.1177/19458924231184055
Agusti, Torres, Faner, Early treatment with inhaled budesonide to prevent clinical deterioration in patients with COVID-19, The Lancet Respiratory Medicine, doi:10.1016/S2213-2600(21)00171-5
Auinger, Besser, Liu, Renner, Mueller, Long-term impact of olfactory dysfunction on daily life, Wien Klin Wochenschr, doi:10.1007/s00508-020-01751-5
Butowt, Bartheld, Anosmia in COVID-19: Underlying Mechanisms and Assessment of an Olfactory Route to Brain Infection, Neuroscientist, doi:10.1177/1073858420956905
Doty, Olfactory dysfunction and its measurement in the clinic, World j Otorhinolaryngol-Head Neck Surg, doi:10.1016/j.wjorl.2015.09.007
Eloit, Trotier, A new clinical olfactory test to quantify olfactory deficiencies, Rhinology
Han, Huang, Jiang, Dong, Peng et al., Early Clinical and CT Manifestations of Coronavirus Disease 2019 (COVID-19) Pneumonia, AJR Am J Roentgenol, doi:10.2214/AJR.20.22961
Hura, Xie, Choby, Schlosser, Orlov et al., Treatment of post-viral olfactory dysfunction: an evidence-based review with recommendations, Int Forum Allergy Rhinol, doi:10.1002/alr.22624
Imam, Abdelazim, Abdelazim, Ismaiel, Gamal et al., Efficacy of pentasodium diethylenetriamine pentaacetate in ameliorating anosmia post COVID-19, Am J Otolaryngol, doi:10.1016/j.amjoto.2023.103871
Jafar, Lasso, Shorr, Hutton, Kilty, Olfactory recovery following infection with COVID-19: A systematic review, PLoS ONE, doi:10.1371/journal.pone.0259321
Kohli, Soler, Nguyen, Muus, Schlosser, The Association Between Olfaction and Depression: A Systematic Review, Chem Senses, doi:10.1093/chemse/bjw061
Kokubo, Carvalho, Fornazieri, Gomes, De et al., Effects of septorhinoplasty on smell perception, Eur Arch Otorhinolaryngol, doi:10.1007/s00405-019-05356-1
Kubba, Spinou, Robertson, The Effect of Head Position on the Distribution of Drops within the Nose, American Journal of Rhinology, doi:10.2500/105065800781692949
Lechien, Chiesa-Estomba, Siati, Horoi, Bon et al., Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): a multicenter European study, Eur Arch Otorhinolaryngol, doi:10.1007/s00405-020-05965-1
Lee, Son, Han, Jung, Park, Association between Inhaled Corticosteroid Use and SARS-CoV-2 Infection: A Nationwide Population-Based Study in South Korea, Tuberc Respir Dis, doi:10.4046/trd.2021.0102
Mahase, Covid-19: Budesonide shortens recovery time in patients not admitted to hospital, study finds, BMJ, doi:10.1136/bmj.n957
Mao, Wang, Hu, Chen, He, Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease, JAMA Neurol, doi:10.1001/jamaneurol.2020.1127
Mcgettigan, Dhuibhir, Barrett, Sui, Balding et al., Subjective and Objective Assessment of Taste and Smell Sensation in Advanced Cancer, Am J Hosp Palliat Care, doi:10.1177/1049909119832836
Nguyen, Patel, Budesonide irrigation with olfactory training improves outcomes compared with olfactory training alone in patients with olfactory loss: Budesonide and training, Int Forum Allergy Rhinol, doi:10.1002/alr.22140
O'byrne, Webster, Mackeith, Philpott, Hopkins et al., Interventions for the treatment of persistent post-COVID-19 olfactory dysfunction, Cochrane Database of Systematic Reviews, doi:10.1002/14651858.CD013876.pub2
Qiu, Cui, Hautefort, Haehner, Zhao et al., Olfactory and Gustatory Dysfunction as an Early Identifier of COVID-19 in Adults and Children: An International Multicenter Study, Otolaryngol Head Neck Surg, doi:10.1177/0194599820934376
Rashid, Zgair, Rm, Effect of nasal corticosteroid in the treatment of anosmia due to COVID-19: A randomised double-blind placebo-controlled study, American Journal of Otolaryngology, doi:10.1016/j.amjoto.2021.103033
Scheibe, Bethge, Witt, Hummel, Intranasal Administration of Drugs, Arch Otolaryngol Head Neck Surg, doi:10.1001/archotol.134.6.643
Teaima, Salem, Teama, Mansour, Taha et al., Patterns and clinical outcomes of olfactory and gustatory disorders in six months: Prospective study of 1031 COVID-19 patients, American Journal of Otolaryngology, doi:10.1016/j.amjoto.2021.103259
Vaira, Hopkins, Petrocelli, Lechien, Cutrupi et al., Efficacy of corticosteroid therapy in the treatment of long-lasting olfactory disorders in COVID-19 patients, Rhin, doi:10.4193/Rhin20.515
Vaira, Salzano, Deiana, Riu, Anosmia and Ageusia: Common Findings in COVID-19 Patients, Laryngoscope, doi:10.1002/lary.28692
Wang, Zhang, Sang, Ye, Ruan et al., Kinetics of viral load and antibody response in relation to COVID-19 severity, Journal of Clinical Investigation, doi:10.1172/JCI138759
Yan, Overdevest, Patel, Therapeutic use of steroids in non-chronic rhinosinusitis olfactory dysfunction: a systematic evidence-based review with recommendations, Int Forum Allergy Rhinol, doi:10.1002/alr.22240
Yu, Bafadhel, Dorward, Hayward, Saville et al., Inhaled budesonide for COVID-19 in people at high risk of complications in the community in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial, The Lancet, doi:10.1016/S0140-6736(21)01744-X
Zhou, Yu, Du, Fan, Liu et al., Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study, The Lancet, doi:10.1016/S0140-6736(20)30566-3
DOI record:
{
"DOI": "10.1016/j.ijid.2023.08.022",
"ISSN": [
"1201-9712"
],
"URL": "http://dx.doi.org/10.1016/j.ijid.2023.08.022",
"alternative-id": [
"S1201971223007075"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Local budesonide therapy in the management of persistent hyposmia in suspected non-severe COVID-19 patients: Results of a randomized controlled trial"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "International Journal of Infectious Diseases"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.ijid.2023.08.022"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2023 Published by Elsevier Ltd on behalf of International Society for Infectious Diseases."
}
],
"author": [
{
"affiliation": [],
"family": "Hautefort",
"given": "Charlotte",
"sequence": "first"
},
{
"affiliation": [],
"family": "Corré",
"given": "Alain",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Poillon",
"given": "Guillaume",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Jourdaine",
"given": "Clément",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Housset",
"given": "Juliette",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Eliezer",
"given": "Michael",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Verillaud",
"given": "Benjamin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Slama",
"given": "Dorsaf",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ayache",
"given": "Denis",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Herman",
"given": "Philippe",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0002-9911-8556",
"affiliation": [],
"authenticated-orcid": false,
"family": "Yavchitz",
"given": "Amélie",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Guillaume",
"given": "Jessica",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hervé",
"given": "Camille",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bakkouri",
"given": "Wissame El",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Salmon",
"given": "Dominique",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Daval",
"given": "Mary",
"sequence": "additional"
}
],
"container-title": "International Journal of Infectious Diseases",
"container-title-short": "International Journal of Infectious Diseases",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.fr",
"clinicalkey.jp",
"clinicalkey.es",
"clinicalkey.com.au",
"clinicalkey.com",
"ijidonline.com",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2023,
8,
29
]
],
"date-time": "2023-08-29T15:40:55Z",
"timestamp": 1693323655000
},
"deposited": {
"date-parts": [
[
2023,
8,
31
]
],
"date-time": "2023-08-31T22:41:22Z",
"timestamp": 1693521682000
},
"indexed": {
"date-parts": [
[
2023,
9,
1
]
],
"date-time": "2023-09-01T11:41:59Z",
"timestamp": 1693568519647
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2023,
8
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2023,
8,
1
]
],
"date-time": "2023-08-01T00:00:00Z",
"timestamp": 1690848000000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 26,
"start": {
"date-parts": [
[
2023,
8,
27
]
],
"date-time": "2023-08-27T00:00:00Z",
"timestamp": 1693094400000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971223007075?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S1201971223007075?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"prefix": "10.1016",
"published": {
"date-parts": [
[
2023,
8
]
]
},
"published-print": {
"date-parts": [
[
2023,
8
]
]
},
"publisher": "Elsevier BV",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S1201971223007075"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Infectious Diseases",
"Microbiology (medical)",
"General Medicine"
],
"subtitle": [],
"title": "Local budesonide therapy in the management of persistent hyposmia in suspected non-severe COVID-19 patients: Results of a randomized controlled trial",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy"
}
