The Quercetin Paradox, Evaluation of its Therapeutic Efficacy as a Nutraceutical in Clinical Trials: Evidence and Speculation
et al., Dose-Response, doi:10.1177/15593258261419728, Jan 2026
Quercetin for COVID-19
36th treatment shown to reduce risk in
January 2022, now with p = 0.0018 from 9 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
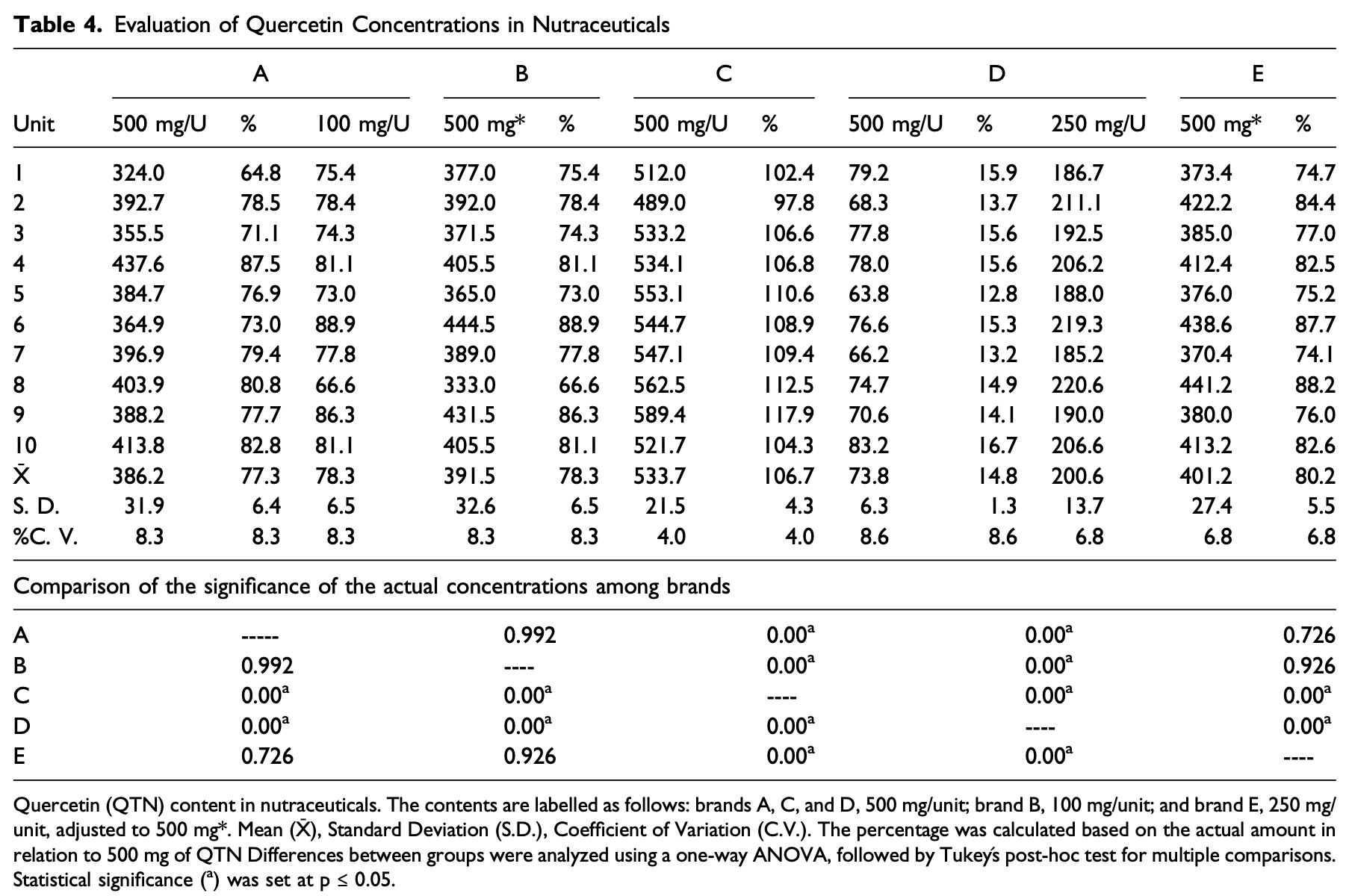
Analysis of five quercetin formulations showing significant variability in quercetin content compared to label claims, with only one brand meeting the labeled concentration, while another brand contained only 15% of the stated amount.
Bioavailability. Quercetin has low bioavailability and studies typically use advanced formulations to improve bioavailability which may be required to reach therapeutic concentrations.
García Alvarez et al., 31 Jan 2026, Mexico, peer-reviewed, 6 authors.
Contact: lili_rives@yahoo.com.
The Quercetin Paradox, Evaluation of its Therapeutic Efficacy as a Nutraceutical in Clinical Trials: Evidence and Speculation
Dose-Response, doi:10.1177/15593258261419728
Introduction: Quercetin, a dietary flavonoid typically consumed as a glycoside, is available in supplements in its aglycone form. This aglycone form is the most studied in preclinical and clinical research because of its antioxidant, anti-inflammatory, and metabolic properties. Several clinical trials have demonstrated the benefits of quercetin for enzymatic, metabolic and cardiovascular health, approximately 60% of these studies fail to verify the actual concentration administered, generating uncertainty on their findings. Objective: This study aimed to evaluate the concentration, stability, and cost-concentration of commercial quercetin nutraceutical formulations. Methodology: The concentrations of five commercial brands were quantified by high-performance liquid chromatography. Stability was assessed over 6 months at both room temperature and 40°C. Results: Only one brand contained the amount labeled; three contained approximately 80%, and one contained merely 14.8%. This study is the first to evaluate the long and thermal stability of quercetin in nutraceutical products, demonstrating that the active ingredient remains stable for at least 6 months. Brand C was identified as the most suitable option. Conclusion: There was significant variability in the quercetin concentration of commercial supplements. These findings underscore the need for stricter regulatory oversight to ensure the quality, therapeutic efficacy, and safety of nutraceutical products.
Declaration of conflicting interests The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Institutional Review Board Statement The
References
Ahmed, Khan, Ali, In silico and in vivo: evaluating the therapeutic potential of kaempferol, quercetin, and catechin to treat chronic epilepsy in a rat model, Front Bioeng Biotechnol, doi:10.3389/fbioe.2021.754952
Almeida, Borge, Piskula, Bioavailability of quercetin in humans with a focus on interindividual variation, Compr Rev Food Sci Food Saf, doi:10.1111/1541-4337.12342
Andres, Pevny, Ziegenhagen, Safety aspects of the use of quercetin as a dietary supplement, Mol Nutr Food Res, doi:10.1002/mnfr.201700447
Batiha, Beshbishy, Ikram, The pharmacological activity, biochemical properties, and pharmacokinetics of the major natural polyphenolic flavonoid: quercetin, Foods, doi:10.3390/foods9030374
Bazzucchi, Patrizio, Ceci, The effects of quercetin supplementation on eccentric exercise-induced muscle damage, Nutrients, doi:10.3390/nu11010205
Bensa, Vovk, Glavnik, Resveratrol food supplement products and the challenges of accurate label information to ensure food safety for consumers, Nutrients, doi:10.3390/nu15020474
Berger, Wein, Blank, Metges, Wolffram, Bioavailability of the flavonol quercetin in cows after intraruminal application of quercetin aglycone and rutin, J Dairy Sci, doi:10.3168/jds.2012-5439
Boots, Drent, De Boer, Bast, Haenen, Quercetin reduces markers of oxidative stress and inflammation in sarcoidosis, Clin Nutr, doi:10.1016/j.clnu.2011.01.010
Burak, Brüll, Langguth, Higher plasma quercetin levels following oral administration of an onion skin extract compared with pure quercetin dihydrate in humans, Eur J Nutr, doi:10.1007/s00394-015-1084-x
Cermak, Breves, Lüpke, In vitro degradation of the flavonol quercetin and of quercetin glycosides in the porcine hindgut, Arch Anim Nutr, doi:10.1080/17450390500467695
Chekalina, Burmak, Petrov, Quercetin reduces the transcriptional activity of NF-kB in stable coronary artery disease, Indian Heart J, doi:10.1016/j.ihj.2018.04.006
Chua, Anderson, Chen, Hu, Quality, labeling accuracy, and cost comparison of purified soy isoflavonoid products, J Alternative Compl Med, doi:10.1089/acm.2004.10.1053
Crozier, Aganath, Michael, Dietary phenolics: chemistry, bioavailability and effects on health, Nat Prod Rep, doi:10.1039/b802662a
Daneshvar, Hariri, Ghiasvand, Effect of eight weeks of quercetin supplementation on exercise performance, muscle damage and body muscle in male badminton players, Int J Prev Med
Dehghani, Seyedi Jandaghi, Janani, Sarebanhassanabadi, Emamat et al., Effects of quercetin supplementation on inflammatory factors and quality of life in post-myocardial infarction patients: a double blind, placebocontrolled, randomized clinical trial, Phyther Res, doi:10.1002/ptr.6955
Duranti, Ceci, Patrizio, Chronic consumption of quercetin reduces erythrocytes oxidative damage: evaluation at resting and after eccentric exercise in humans, Nutr Res, doi:10.1016/j.nutres.2017.12.002
Egert, Wolffram, Bosy-Westphal, Daily quercetin supplementation dose-dependently increases plasma quercetin concentrations in healthy humans, J Nutr, doi:10.1093/jn/138.9.1615
Guiance, Marino, Isern, Flavonoides: aplicaciones medicinales e industriales, Invenio
Gómez-Garduño, Garcia-Alvárez, Pacheco, Cellular toxicity of quercetin and its effect on glucose movilization, Acta Pediátrica México, doi:10.18233/qpm.v46i1.2746
Gómez-Garduño, León-Rodríguez, Medina, Phytochemicals that interfere with drug metabolism and transport, modifying plasma concentration in humans and animals, Dose Response
Heijnen, Haenen, Oostveen, Stalpers, Bast, Protection of flavonoids against lipid peroxidation: the structure activity relationship revisited, Free Radic Res, doi:10.1080/10715760290025951
Javadi, Ahmadzadeh, Eghtesadi, The effect of quercetin on inflammatory factors and clinical symptoms in women with rheumatoid arthritis: a double-blind, randomized controlled trial, J Am Coll Nutr, doi:10.1080/07315724.2016.1140093
Khorshidi, Moini, Alipoor, The effects of quercetin supplementation on metabolic and hormonal parameters as well as plasma concentration and gene expression of resistin in overweight or obese women with polycystic ovary syndrome, Phyther Res, doi:10.1002/ptr.6166
Mantadaki, Linardakis, Tsakiri, Benefits of quercetin on glycated hemoglobin, blood pressure, piKo-6 readings, night-time sleep, anxiety, and quality of life in patients wiah type 2 diabetes mellitus: a randomized controlled trial, J Clin Med, doi:10.3390/jcm13123504
Mantadaki, Linardakis, Vafeiadi, Anastasiou, Tsatsakis et al., The impact of three-month quercetin intake on quality of life and anxiety in patients with type II diabetes mellitus: an early data analysis from a randomized controlled trial, Cureus, doi:10.7759/cureus.58219
Mazloom, Abdollahzadeh, Dabbaghmanesh, The effect of quercetin supplementation on oxidative stress, glycemic control, lipid profile and insulin resistance in type 2 diabetes: a randomized clinical trial, J Heal Sci Surveill Sys
Mehta, Nelson, Loveless, Phase 1 study of quercetin, a natural antioxidant for children and young adults with Fanconi anemia, Blood Adv, doi:10.1182/bloodadvances.2024015053
Olson, Melton, Dong, Bowden, Stabilization of quercetin paradoxically reduces its proapoptotic effect on UVB-Irradiated human keratinocytes, Cancer Prev Res, doi:10.1158/1940-6207.CAPR-08-0101
Pardo-Barrela, Lago-Crespo, Lage-Yusty, Hernández, Comparison of methods for rapid analysis of quercetin, Plant Foods Hum Nutr, doi:10.1007/s11130-014-0454-5
Pfeuffer, Auinger, Bley, Effect of quercetin on traits of the metabolic syndrome, endothelial function and inflammation in men with different APOE isoforms, Nutr Metabol Cardiovasc Dis, doi:10.1016/j.numecd.2011.08.010
Ramaa, Shirode, Mundada, Kadam, Nutraceuticalsan emerging era in the treatment and prevention of cardiovascular diseases, Curr Pharm Biotechnol, doi:10.2174/138920106775789647
Reinboth, Wolffram, Abraham, Ungemach, Cermak, Oral bioavailability of quercetin from different quercetin glycosides in dogs, Br J Nutr, doi:10.1017/S000711451000053X
Rezvan, Moini, Gorgani-Firuzjaee, Mj, Oral quercetin supplementation enhances adiponectin receptor transcript expression in polycystic ovary syndrome patients: a randomized placebo-controlled doubleblind clinical trial, Cell J, doi:10.22074/cellj.2018.4577
Rezvan, Moini, Janani, Effects of quercetin on adiponectin-mediated insulin sensitivity in polycystic ovary syndrome: a randomized placebo-controlled double-blind clinical trial, Horm Metab Res, doi:10.1055/s-0042-118705
Shi, Williamson, Quercetin lowers plasma uric acid in prehyperuricaemic males: a randomised, double-blinded, placebocontrolled, cross-over trial, Br J Nutr, doi:10.1017/S0007114515005310
Shohan, Nashibi, Mahmoudian-Sani, The therapeutic efficacy of quercetin in combination with antiviral drugs in hospitalized COVID-19 patients: a randomized controlled trial, Eur J Pharmacol, doi:10.1016/j.ejphar.2021.174615
Terao, Potential role of quercetin glycosides as antiatherosclerotic food-derived factors for human health, Antioxidants, doi:10.3390/antiox12020258
Tsao, Bernard, Hsu, Hsu, Liao et al., Short-term oral quercetin supplementation improves postexercise insulin sensitivity, antioxidant capacity and enhances subsequent cycling time to exhaustion in healthy adults: a pilot study, Front Nutr, doi:10.3389/fnut.2022.875319
Vida, Fittler, Somogyi-Végh, Dietary quercetin supplements: assessment of online product informations and quantitation of quercetin in the products by high-performance liquid chromatography, Phyther Res, doi:10.1002/ptr.6382
Zhang, Angst, Jenny, Quercetin aglycone is bioavailable in murine pancreas and pancreatic xenografts, J Agric Food Chem, doi:10.1021/jf101192k
DOI record:
{
"DOI": "10.1177/15593258261419728",
"ISSN": [
"1559-3258",
"1559-3258"
],
"URL": "http://dx.doi.org/10.1177/15593258261419728",
"abstract": "<jats:sec>\n <jats:title>Introduction</jats:title>\n <jats:p>Quercetin, a dietary flavonoid typically consumed as a glycoside, is available in supplements in its aglycone form. This aglycone form is the most studied in preclinical and clinical research because of its antioxidant, anti-inflammatory, and metabolic properties. Several clinical trials have demonstrated the benefits of quercetin for enzymatic, metabolic and cardiovascular health, approximately 60% of these studies fail to verify the actual concentration administered, generating uncertainty on their findings.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Objective</jats:title>\n <jats:p>This study aimed to evaluate the concentration, stability, and cost–concentration of commercial quercetin nutraceutical formulations.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Methodology</jats:title>\n <jats:p>The concentrations of five commercial brands were quantified by high-performance liquid chromatography. Stability was assessed over 6 months at both room temperature and 40°C.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>Only one brand contained the amount labeled; three contained approximately 80%, and one contained merely 14.8%. This study is the first to evaluate the long and thermal stability of quercetin in nutraceutical products, demonstrating that the active ingredient remains stable for at least 6 months. Brand C was identified as the most suitable option.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusion</jats:title>\n <jats:p>There was significant variability in the quercetin concentration of commercial supplements. These findings underscore the need for stricter regulatory oversight to ensure the quality, therapeutic efficacy, and safety of nutraceutical products.</jats:p>\n </jats:sec>",
"alternative-id": [
"10.1177/15593258261419728"
],
"article-number": "15593258261419728",
"author": [
{
"ORCID": "https://orcid.org/0000-0003-3563-134X",
"affiliation": [
{
"name": "National Institute of Pediatrics"
}
],
"authenticated-orcid": false,
"family": "García Alvarez",
"given": "Raquel",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0001-6919-4497",
"affiliation": [
{
"name": "National Institute of Pediatrics"
}
],
"authenticated-orcid": false,
"family": "Chávez-Pacheco",
"given": "Juan Luis",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Institute of Pediatrics"
}
],
"family": "Altamirano-Bustamante",
"given": "Nelly Francisca",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-2981-2347",
"affiliation": [
{
"name": "National Institute of Pediatrics"
}
],
"authenticated-orcid": false,
"family": "Solorio-López",
"given": "Edelmira",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-0226-3651",
"affiliation": [
{
"name": "National Institute of Pediatrics"
}
],
"authenticated-orcid": false,
"family": "Gómez-Garduño",
"given": "Josefina",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-1334-0026",
"affiliation": [
{
"name": "National Institute of Pediatrics"
}
],
"authenticated-orcid": false,
"family": "Rivera-Espinosa",
"given": "Liliana",
"sequence": "additional"
}
],
"container-title": "Dose-Response",
"container-title-short": "Dose-Response",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"journals.sagepub.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
11
]
],
"date-time": "2026-03-11T10:54:34Z",
"timestamp": 1773226474000
},
"deposited": {
"date-parts": [
[
2026,
3,
11
]
],
"date-time": "2026-03-11T10:54:36Z",
"timestamp": 1773226476000
},
"funder": [
{
"name": "Program E022 of the National Institute of Pediatrics"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
12
]
],
"date-time": "2026-03-12T01:25:23Z",
"timestamp": 1773278723067,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2026,
1
]
]
},
"journal-issue": {
"issue": "1",
"published-print": {
"date-parts": [
[
2026,
1
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by-nc/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
1,
1
]
],
"date-time": "2026-01-01T00:00:00Z",
"timestamp": 1767225600000
}
},
{
"URL": "https://journals.sagepub.com/page/policies/text-and-data-mining-license",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
1,
1
]
],
"date-time": "2026-01-01T00:00:00Z",
"timestamp": 1767225600000
}
}
],
"link": [
{
"URL": "https://journals.sagepub.com/doi/pdf/10.1177/15593258261419728",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://journals.sagepub.com/doi/full-xml/10.1177/15593258261419728",
"content-type": "application/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://journals.sagepub.com/doi/pdf/10.1177/15593258261419728",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "179",
"original-title": [],
"prefix": "10.1177",
"published": {
"date-parts": [
[
2026,
1
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
6
]
]
},
"published-print": {
"date-parts": [
[
2026,
1
]
]
},
"publisher": "SAGE Publications",
"reference": [
{
"DOI": "10.1002/mnfr.201700447",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_2_2"
},
{
"DOI": "10.3390/foods9030374",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_3_2"
},
{
"DOI": "10.1177/15593258221120485",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_4_2"
},
{
"DOI": "10.1021/jf101192k",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_5_2"
},
{
"DOI": "10.3168/jds.2012-5439",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_6_2"
},
{
"DOI": "10.1158/1940-6207.CAPR-08-0101",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_7_2"
},
{
"DOI": "10.3389/fbioe.2021.754952",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_8_2"
},
{
"DOI": "10.2174/138920106775789647",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_9_2"
},
{
"article-title": "Flavonoides: aplicaciones medicinales e industriales",
"author": "Guiance HSN",
"first-page": "11",
"issue": "40",
"journal-title": "Invenio",
"key": "e_1_3_4_10_2",
"unstructured": "Guiance HSN, Marino L, Isern D, et al. Flavonoides: aplicaciones medicinales e industriales. Invenio. 2019;22(40):11-27.",
"volume": "22",
"year": "2019"
},
{
"DOI": "10.3389/fnut.2022.875319",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_11_2"
},
{
"DOI": "10.1080/10715760290025951",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_12_2"
},
{
"DOI": "10.1016/j.clnu.2011.01.010",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_13_2"
},
{
"DOI": "10.1111/1541-4337.12342",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_14_2"
},
{
"DOI": "10.1007/s00394-015-1084-x",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_15_2"
},
{
"DOI": "10.1039/b802662a",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_16_2"
},
{
"DOI": "10.1080/17450390500467695",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_17_2"
},
{
"DOI": "10.3390/antiox12020258",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_18_2"
},
{
"DOI": "10.1017/S000711451000053X",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_19_2"
},
{
"DOI": "10.1016/j.numecd.2011.08.010",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_20_2"
},
{
"article-title": "Effect of eight weeks of quercetin supplementation on exercise performance, muscle damage and body muscle in male badminton players",
"author": "Daneshvar P",
"first-page": "S53",
"journal-title": "Int J Prev Med",
"key": "e_1_3_4_21_2",
"unstructured": "Daneshvar P, Hariri M, Ghiasvand R, et al. Effect of eight weeks of quercetin supplementation on exercise performance, muscle damage and body muscle in male badminton players. Int J Prev Med. 2013;4:S53-S57.",
"volume": "4",
"year": "2013"
},
{
"DOI": "10.1016/j.nutres.2017.12.002",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_22_2"
},
{
"DOI": "10.3390/nu11010205",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_23_2"
},
{
"DOI": "10.1093/jn/138.9.1615",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_24_2"
},
{
"DOI": "10.1017/S0007114515005310",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_25_2"
},
{
"DOI": "10.1016/j.ihj.2018.04.006",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_26_2"
},
{
"DOI": "10.1002/ptr.6955",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_27_2"
},
{
"DOI": "10.1002/ptr.6166",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_28_2"
},
{
"DOI": "10.1055/s-0042-118705",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_29_2"
},
{
"DOI": "10.1182/bloodadvances.2024015053",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_30_2"
},
{
"DOI": "10.1016/j.ejphar.2021.174615",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_31_2"
},
{
"article-title": "The effect of quercetin supplementation on oxidative stress, glycemic control, lipid profile and insulin resistance in type 2 diabetes: a randomized clinical trial",
"author": "Mazloom Z",
"first-page": "8",
"issue": "1",
"journal-title": "J Heal Sci Surveill Sys",
"key": "e_1_3_4_32_2",
"unstructured": "Mazloom Z, Abdollahzadeh SM, Dabbaghmanesh MH, et al. The effect of quercetin supplementation on oxidative stress, glycemic control, lipid profile and insulin resistance in type 2 diabetes: a randomized clinical trial. J Heal Sci Surveill Sys. 2014;2(1):8-14.",
"volume": "2",
"year": "2014"
},
{
"DOI": "10.7759/cureus.58219",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_33_2"
},
{
"DOI": "10.22074/cellj.2018.4577",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_34_2"
},
{
"DOI": "10.1080/07315724.2016.1140093",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_35_2"
},
{
"DOI": "10.18233/qpm.v46i1.2746",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_36_2"
},
{
"key": "e_1_3_4_37_2",
"unstructured": "Secretaria de Salud. NORMA oficial mexicana NOM-177-SSA1-2013. https://www.dof.gob.mx/nota_detalle.php?codigo=5314833&fecha=20/09/2013#gsc.tab=0 (Accesed 15 12 2024)."
},
{
"key": "e_1_3_4_38_2",
"unstructured": "FDA. Bioanalytical method validation guidance for industry bioanalytical method validation guidance for industry. 2018. https://www.fda.gov/files/drugs/published/Bioanalytical-Method-Validation-Guidance-for-Industry.pdf. Accesed 6 03 2025."
},
{
"key": "e_1_3_4_39_2",
"unstructured": "Secretaría de Salud. Comisión permanente de la Farmacopea de los Estados Unidos Mexicanos. In: Supplement 13.2 13 ed. Farmacopea De Los Estados Unidos Mexicanos. Uniformidad de dosis; 2025. MGA 0299. cpfeum@farmacopea.org.mx."
},
{
"DOI": "10.1089/acm.2004.10.1053",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_40_2"
},
{
"key": "e_1_3_4_41_2",
"unstructured": "Secretaria de Salud. NORMA oficial mexicana NOM-073-SSA1-2015. 2015. https://dof.gob.mx/nota_detalle_popup.php?codigo=5440183. (Accesed 6 12 2024)."
},
{
"key": "e_1_3_4_42_2",
"unstructured": "Secretaria de Salud. COFEPRIS. Guía de Etiquetado de Suplementos Alimenticios. 2022. https://www.gob.mx/cofepris/documentos/consulta-de-clasificacion-de-producto-como-suplemento-alimenticio-2?state=published. Published accesed 12 2024."
},
{
"key": "e_1_3_4_43_2",
"unstructured": "Secretaria de Salud COFEPRIS. Marco jurídico para suplementos alimenticios. 2016. https://www.gob.mx/cofepris/acciones-y-programas/marco-juridico-para-suplementos-alimenticios. (Accesed 15 12 2024)."
},
{
"DOI": "10.1002/ptr.6382",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_44_2"
},
{
"DOI": "10.1007/s11130-014-0454-5",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_45_2"
},
{
"DOI": "10.3390/jcm13123504",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_46_2"
},
{
"key": "e_1_3_4_47_2",
"unstructured": "FDA (U.S. Food and Drug Administration). GRAS notice for high-purity quercetin submitted by Quercegen pharma LLC. Gras notice for high purity quercetin. 2010. https://www.hfpappexternal.fda.gov/scripts/fdcc/index.cfm?set=GrASNotices&id=341 (Accesed 15 11 2025)."
},
{
"DOI": "10.3390/nu15020474",
"doi-asserted-by": "publisher",
"key": "e_1_3_4_48_2"
}
],
"reference-count": 47,
"references-count": 47,
"relation": {},
"resource": {
"primary": {
"URL": "https://journals.sagepub.com/doi/10.1177/15593258261419728"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "The Quercetin Paradox, Evaluation of its Therapeutic Efficacy as a Nutraceutical in Clinical Trials: Evidence and Speculation",
"type": "journal-article",
"update-policy": "https://doi.org/10.1177/sage-journals-update-policy",
"volume": "24"
}
