Tocilizumab Efficacy Across Inflammatory Subphenotypes in COVID-19-Related Acute Respiratory Distress Syndrome
et al., Critical Care Explorations, doi:10.1097/cce.0000000000001392, Mar 2026
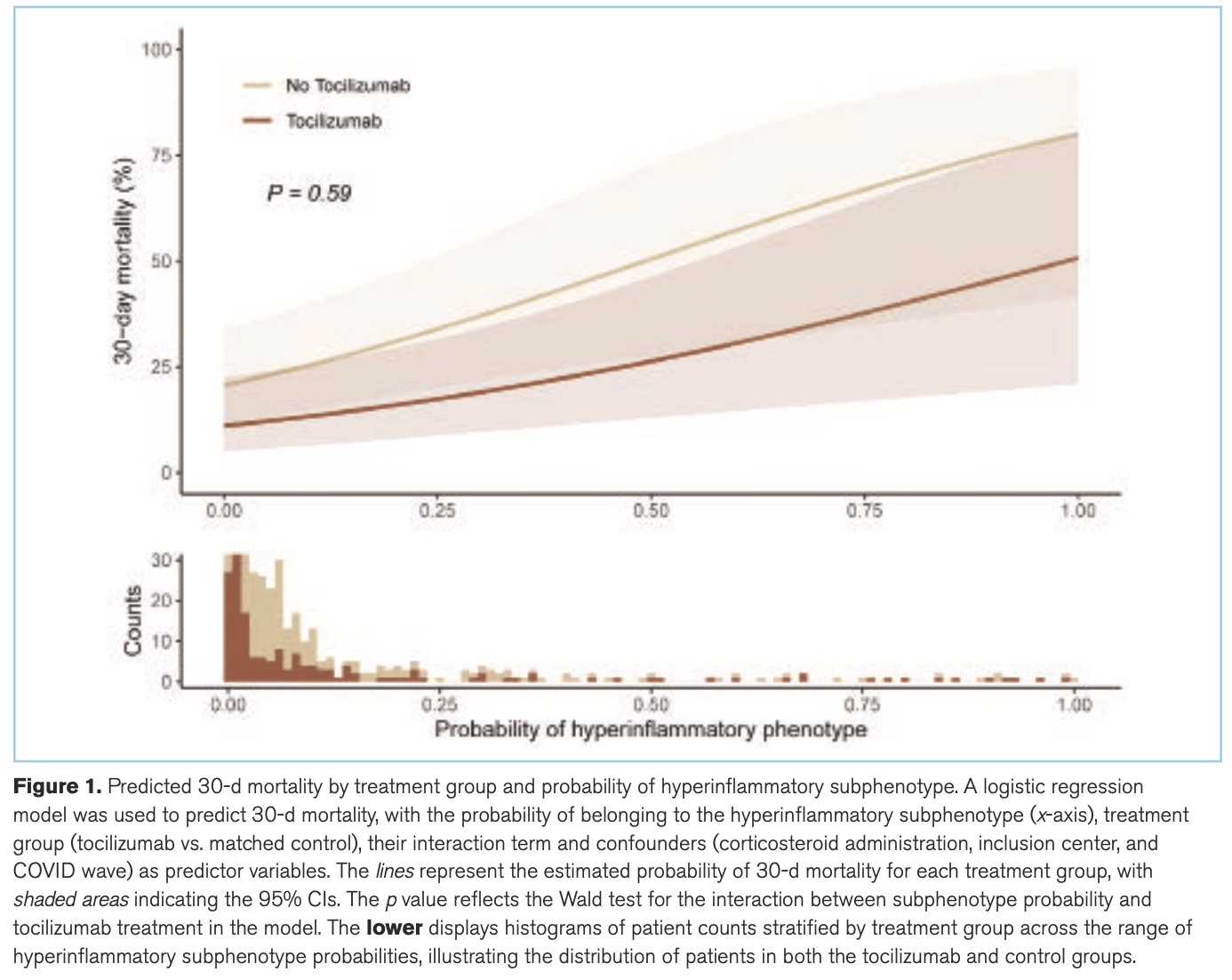
Retrospective 561 mechanically ventilated COVID-19 ARDS patients across three Dutch university hospital ICUs, showing significant reduction in 30-day mortality with tocilizumab, with no significant difference in efficacy between hypoinflammatory and hyperinflammatory subphenotypes.
Severe imbalance in COVID wave timing despite propensity matching: authors state outcomes improved in later waves, yet the 'Wave and strain' variable shows a very large mismatch (SMD 1.10) between the tocilizumab group (64% Alpha, 1% Original) and matched controls (27% Alpha, 59% Original). This temporal confounding severely biases the mortality outcome. The regression adjustment should partially correct for this confounding, but is limited by collinearity and the fact that the wave is a crude proxy for the real confounders such as evolving clinical protocols, staff experience, changing viral variants, shifts in admission thresholds.
Column sums in Table 1 exceed the stated sample size: in Table 1, the 'Wave and strain' categories for the matched controls (n=344) list counts of 160, 95, 87, and 52. These sum up to 394, more than the number of patients.
Errors in percentages in Table 1: For the comorbidity 'Hypertension', the tocilizumab group lists 35 cases out of 172, which is 20.3%, but reports it as 36%. For the controls, it lists 70 cases out of 344, which is 20.3%, but reports it as 46%.
Rounding inconsistencies in hypoinflammatory percentages: in Table 1, 153/172 is 88.95%, but is reported as 90%.
Filippini et al., 16 Mar 2026, retrospective, Netherlands, peer-reviewed, 10 authors.
Tocilizumab Efficacy Across Inflammatory Subphenotypes in COVID-19-Related Acute Respiratory Distress Syndrome
Critical Care Explorations, doi:10.1097/cce.0000000000001392
OBJECTIVES: This study evaluated whether the established efficacy of tocilizumab, an interleukin-6 (IL-6) receptor antagonist, differs between the hypoinflammatory and hyperinflammatory subphenotypes. DESIGN: Retrospective analysis of data from three biobanks. SETTING: ICUs of three university teaching hospitals in the Netherlands.
PATIENTS: Mechanically ventilated patients with COVID-19.
INTERVENTIONS: Tocilizumab administration vs. no administration.
MEASUREMENTS AND MAIN RESULTS: A total of 561 patients were included. Based on a classifier model incorporating IL-6, tumor necrosis factor receptor 1, and bicarbonate, 95% were classified as Hypoinflammatory and 5% as Hyperinflammatory. Tocilizumab was associated with a significant reduction in 30-day mortality in the overall cohort, even after adjustment for confounders (p = 0.014). However, there was no evidence that treatment effectiveness differed between the two subphenotypes (p = 0.59).
CONCLUSIONS: In this cohort, tocilizumab significantly reduced 30-day mortality overall. Although the number of Hyperinflammatory patients was low, there was no evidence that its efficacy differed between inflammatory subphenotypes. These findings underscore the importance of including both subphenotypes in future trials evaluating the differential effects of tocilizumab.
APPENDIX A INSTITUTIONAL REVIEW BOARD APPROVAL
References
Abani, Abbas, Abbas, Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial, Lancet
Calfee, Delucchi, Parsons, ARDS Network: Subphenotypes in acute respiratory distress syndrome: Latent class analysis of data from two randomised controlled trials, Lancet Respir Med
Calfee, Delucchi, Sinha, Irish Critical Care Trials Group: Acute respiratory distress syndrome subphenotypes and differential response to simvastatin: Secondary analysis of a randomised controlled trial, Lancet Respir Med
Filippini, Smit, Bos, Subphenotypes in acute respiratory distress syndrome: Universal steps toward treatable traits, Anesth Analg, doi:10.1213/ANE.0000000000006727
Neurology, Endry, Lim, Wiersinga, Marry Smit
Nishimoto, Terao, Mima, Mechanisms and pathologic significances in increase in serum interleukin-6 (IL-6) and soluble IL-6 receptor after administration of an anti-IL-6 receptor antibody, tocilizumab, in patients with rheumatoid arthritis and Castleman disease, Blood
Sinha, Calfee, Cherian, Prevalence of phenotypes of acute respiratory distress syndrome in critically ill patients with COVID-19: A prospective observational study, Lancet Respir Med
Sinha, Delucchi, Mcauley, Development and validation of parsimonious algorithms to classify acute respiratory distress syndrome phenotypes: A secondary analysis of randomised controlled trials, Lancet Respir Med
Sinha, Furfaro, Cummings, Latent class analysis reveals COVID-19-related acute respiratory distress syndrome subgroups with differential responses to corticosteroids, Am J Respir Crit Care Med
Sinha, Kerchberger, Willmore, Identifying molecular phenotypes in sepsis: An analysis of two prospective observational cohorts and secondary analysis of two randomised controlled trials, Lancet Respir Med
Van Amstel, Bartek, Vlaar, Temporal transitions of the hyperinflammatory and hypoinflammatory phenotypes in critical illness, Am J Respir Crit Care Med
Van Amstel, Van Vught, Bos, Integrating biology into clinical trial design, Curr Opin Crit Care
DOI record:
{
"DOI": "10.1097/cce.0000000000001392",
"ISSN": [
"2639-8028"
],
"URL": "http://dx.doi.org/10.1097/cce.0000000000001392",
"abstract": "<jats:sec>\n <jats:title>OBJECTIVES:</jats:title>\n <jats:p>This study evaluated whether the established efficacy of tocilizumab, an interleukin-6 (IL-6) receptor antagonist, differs between the hypoinflammatory and hyperinflammatory subphenotypes.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>DESIGN:</jats:title>\n <jats:p>Retrospective analysis of data from three biobanks.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>SETTING:</jats:title>\n <jats:p>ICUs of three university teaching hospitals in the Netherlands.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>PATIENTS:</jats:title>\n <jats:p>Mechanically ventilated patients with COVID-19.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>INTERVENTIONS:</jats:title>\n <jats:p>Tocilizumab administration vs. no administration.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>MEASUREMENTS AND MAIN RESULTS:</jats:title>\n <jats:p>\n A total of 561 patients were included. Based on a classifier model incorporating IL-6, tumor necrosis factor receptor 1, and bicarbonate, 95% were classified as Hypoinflammatory and 5% as Hyperinflammatory. Tocilizumab was associated with a significant reduction in 30-day mortality in the overall cohort, even after adjustment for confounders (\n <jats:italic toggle=\"yes\">p</jats:italic>\n = 0.014). However, there was no evidence that treatment effectiveness differed between the two subphenotypes (\n <jats:italic toggle=\"yes\">p</jats:italic>\n = 0.59).\n </jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>CONCLUSIONS:</jats:title>\n <jats:p>In this cohort, tocilizumab significantly reduced 30-day mortality overall. Although the number of Hyperinflammatory patients was low, there was no evidence that its efficacy differed between inflammatory subphenotypes. These findings underscore the importance of including both subphenotypes in future trials evaluating the differential effects of tocilizumab.</jats:p>\n </jats:sec>",
"author": [
{
"ORCID": "https://orcid.org/0000-0003-0937-8563",
"affiliation": [
{
"name": "Department of Intensive Care Medicine, Amsterdam UMC, University of Amsterdam, Amsterdam, The Netherlands."
}
],
"authenticated-orcid": false,
"family": "Filippini",
"given": "Daan F. L.",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Department of Intensive Care Medicine, Erasmus MC, Erasmus University Rotterdam, Rotterdam, The Netherlands."
}
],
"family": "Khyali",
"given": "Jessica",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Intensive Care Medicine, Erasmus MC, Erasmus University Rotterdam, Rotterdam, The Netherlands."
},
{
"name": "Department of Internal Medicine, Erasmus MC, Erasmus University Rotterdam, Rotterdam, The Netherlands."
}
],
"family": "Janssen",
"given": "Malou",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Intensive Care Medicine, UMC Utrecht, Utrecht University, Utrecht, The Netherlands."
},
{
"name": "Julius Center for Health Sciences and Primary Care, UMC Utrecht, Utrecht, The Netherlands."
}
],
"family": "Rademaker",
"given": "Emma",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Intensive Care Medicine, UMC Utrecht, Utrecht University, Utrecht, The Netherlands."
},
{
"name": "Julius Center for Health Sciences and Primary Care, UMC Utrecht, Utrecht, The Netherlands."
}
],
"family": "Cremer",
"given": "Olaf L.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Center for Infection and Molecular Medicine (C.I.M.M.), Amsterdam UMC, University of Amsterdam, Amsterdam, The Netherlands."
},
{
"name": "Division of Infectious Diseases, Amsterdam UMC, University of Amsterdam, Amsterdam, The Netherlands."
}
],
"family": "van der Poll",
"given": "Tom",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Intensive Care Medicine, Amsterdam UMC, University of Amsterdam, Amsterdam, The Netherlands."
}
],
"family": "van Amstel",
"given": "Rombout B. E.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Intensive Care Medicine, Erasmus MC, Erasmus University Rotterdam, Rotterdam, The Netherlands."
},
{
"name": "Department of Intensive Care Medicine, OLVG, Amsterdam, The Netherlands."
}
],
"family": "Endeman",
"given": "Henrik",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Intensive Care Medicine, Amsterdam UMC, University of Amsterdam, Amsterdam, The Netherlands."
},
{
"name": "Department of Pulmonology, Amsterdam UMC, University of Amsterdam, Amsterdam, The Netherlands."
},
{
"name": "Laboratory of Experimental Intensive Care and Anesthesiology (L.E.I.C.A.), University of Amsterdam, Amsterdam, The Netherlands."
}
],
"family": "Bos",
"given": "Lieuwe D. J.",
"sequence": "additional"
},
{
"affiliation": [],
"name": "on behalf of the Amsterdam UMC COVID-19 Biobank Study Group",
"sequence": "additional"
}
],
"container-title": "Critical Care Explorations",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"lww.com",
"ovid.com"
]
},
"created": {
"date-parts": [
[
2026,
3,
16
]
],
"date-time": "2026-03-16T16:00:09Z",
"timestamp": 1773676809000
},
"deposited": {
"date-parts": [
[
2026,
3,
16
]
],
"date-time": "2026-03-16T16:00:11Z",
"timestamp": 1773676811000
},
"indexed": {
"date-parts": [
[
2026,
3,
16
]
],
"date-time": "2026-03-16T16:51:08Z",
"timestamp": 1773679868823,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issue": "3",
"issued": {
"date-parts": [
[
2026,
3
]
]
},
"journal-issue": {
"issue": "3",
"published-online": {
"date-parts": [
[
2026
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "unspecified",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
1
]
],
"date-time": "2026-03-01T00:00:00Z",
"timestamp": 1772323200000
}
}
],
"link": [
{
"URL": "https://journals.lww.com/10.1097/CCE.0000000000001392",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "276",
"original-title": [],
"page": "e1392",
"prefix": "10.1097",
"published": {
"date-parts": [
[
2026,
3
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
16
]
]
},
"publisher": "Ovid Technologies (Wolters Kluwer Health)",
"reference": [
{
"DOI": "10.1016/S0140-6736(21)00676-0",
"article-title": "Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial.",
"author": "Abani",
"doi-asserted-by": "crossref",
"first-page": "1637",
"journal-title": "Lancet",
"key": "R1-20260316",
"volume": "397",
"year": "2021"
},
{
"DOI": "10.1016/S2213-2600(14)70097-9",
"article-title": "Subphenotypes in acute respiratory distress syndrome: Latent class analysis of data from two randomised controlled trials.",
"author": "Calfee",
"doi-asserted-by": "crossref",
"first-page": "611",
"journal-title": "Lancet Respir Med",
"key": "R2-20260316",
"volume": "2",
"year": "2014"
},
{
"DOI": "10.1213/ANE.0000000000006727",
"article-title": "Subphenotypes in acute respiratory distress syndrome: Universal steps toward treatable traits.",
"author": "Filippini",
"doi-asserted-by": "crossref",
"journal-title": "Anesth Analg",
"key": "R3-20260316",
"year": "2024"
},
{
"DOI": "10.1164/rccm.202406-1241OC",
"article-title": "Temporal transitions of the hyperinflammatory and hypoinflammatory phenotypes in critical illness.",
"author": "van Amstel",
"doi-asserted-by": "crossref",
"first-page": "347",
"journal-title": "Am J Respir Crit Care Med",
"key": "R4-20260316",
"volume": "211",
"year": "2025"
},
{
"DOI": "10.1016/S2213-2600(19)30369-8",
"article-title": "Development and validation of parsimonious algorithms to classify acute respiratory distress syndrome phenotypes: A secondary analysis of randomised controlled trials.",
"author": "Sinha",
"doi-asserted-by": "crossref",
"first-page": "247",
"journal-title": "Lancet Respir Med",
"key": "R5-20260316",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1164/rccm.202105-1302OC",
"article-title": "Latent class analysis reveals COVID-19-related acute respiratory distress syndrome subgroups with differential responses to corticosteroids.",
"author": "Sinha",
"doi-asserted-by": "crossref",
"first-page": "1274",
"journal-title": "Am J Respir Crit Care Med",
"key": "R6-20260316",
"volume": "204",
"year": "2021"
},
{
"DOI": "10.1016/S2213-2600(18)30177-2",
"article-title": "Acute respiratory distress syndrome subphenotypes and differential response to simvastatin: Secondary analysis of a randomised controlled trial.",
"author": "Calfee",
"doi-asserted-by": "crossref",
"first-page": "691",
"journal-title": "Lancet Respir Med",
"key": "R7-20260316",
"volume": "6",
"year": "2018"
},
{
"DOI": "10.1016/S2213-2600(20)30366-0",
"article-title": "Prevalence of phenotypes of acute respiratory distress syndrome in critically ill patients with COVID-19: A prospective observational study.",
"author": "Sinha",
"doi-asserted-by": "crossref",
"first-page": "1209",
"journal-title": "Lancet Respir Med",
"key": "R8-20260316",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(23)00237-0",
"article-title": "Identifying molecular phenotypes in sepsis: An analysis of two prospective observational cohorts and secondary analysis of two randomised controlled trials.",
"author": "Sinha",
"doi-asserted-by": "crossref",
"first-page": "965",
"journal-title": "Lancet Respir Med",
"key": "R9-20260316",
"volume": "11",
"year": "2023"
},
{
"article-title": "Integrating biology into clinical trial design.",
"author": "van Amstel",
"first-page": "26",
"journal-title": "Curr Opin Crit Care",
"key": "R10-20260316",
"volume": "29",
"year": "2023"
},
{
"DOI": "10.1182/blood-2008-05-155846",
"article-title": "Mechanisms and pathologic significances in increase in serum interleukin-6 (IL-6) and soluble IL-6 receptor after administration of an anti-IL-6 receptor antibody, tocilizumab, in patients with rheumatoid arthritis and Castleman disease.",
"author": "Nishimoto",
"doi-asserted-by": "crossref",
"first-page": "3959",
"journal-title": "Blood",
"key": "R11-20260316",
"volume": "112",
"year": "2008"
}
],
"reference-count": 11,
"references-count": 11,
"relation": {},
"resource": {
"primary": {
"URL": "https://journals.lww.com/10.1097/CCE.0000000000001392"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Tocilizumab Efficacy Across Inflammatory Subphenotypes in COVID-19-Related Acute Respiratory Distress Syndrome",
"type": "journal-article",
"update-policy": "https://doi.org/10.1097/lww.0000000000001000",
"volume": "8"
}