A Randomized, Comparative Clinical Study to Evaluate the Activity of CurvicTM Formulation for Management of SARS-COV-2 Infection (COVID-19)
et al., Journal of Clinical Trials, S3:004, CTRI/2020/07/026839, Nov 2020
Curcumin for COVID-19
16th treatment shown to reduce risk in
February 2021, now with p = 0.0000000061 from 28 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT 200 COVID-19 positive patients in India, 100 treated with curcumin, vitamin C, vitamin K2-7, and selenomethionine, showing faster recovery with treatment.
This study is excluded in meta-analysis:
many combined treatments which may significantly contribute to the effect seen.
Study covers curcumin and vitamin K.
|
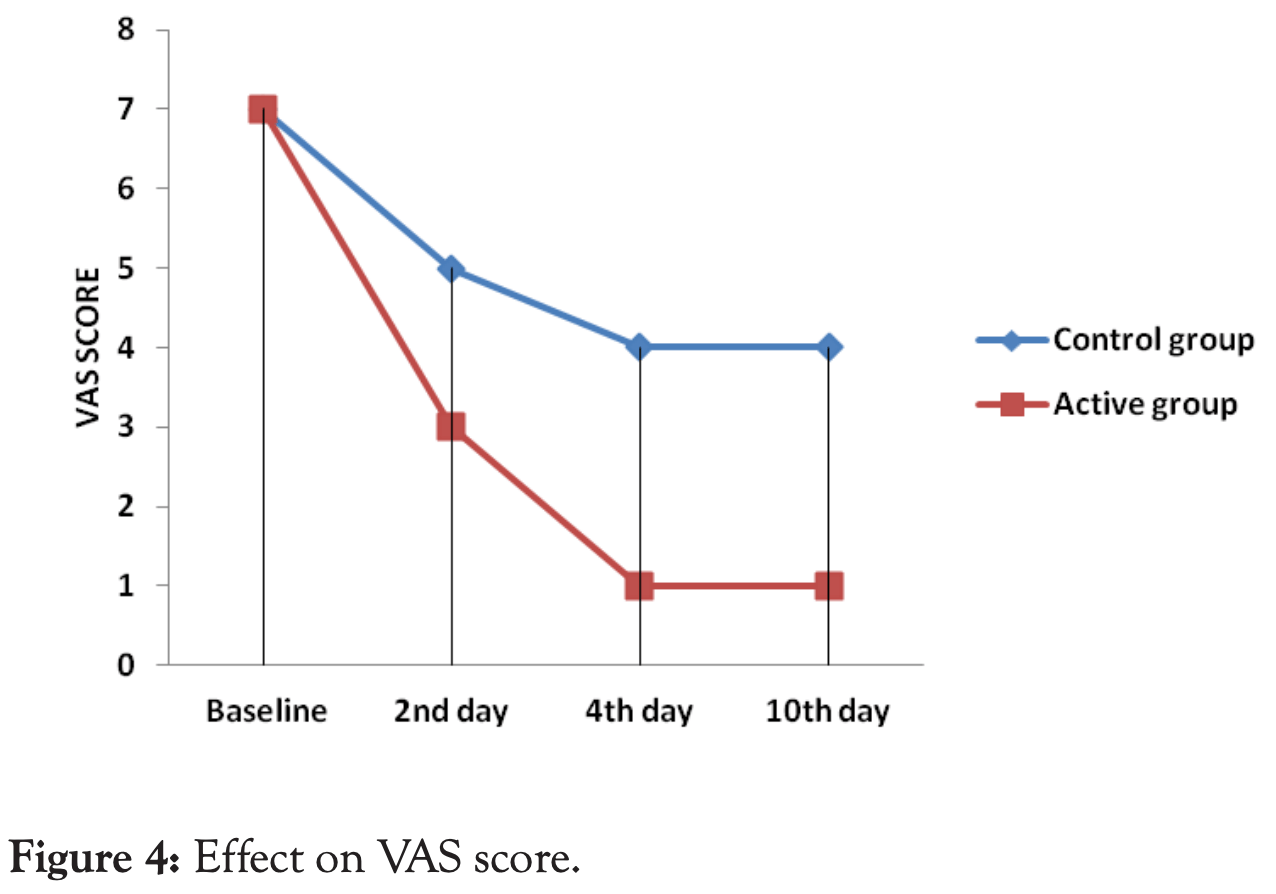
relative VAS score, 75.0% better, RR 0.25, p = 0.001, treatment 100, control 100, inverted to make RR<1 favor treatment, day 10.
|
|
relative VAS score, 75.0% better, RR 0.25, p = 0.001, treatment 100, control 100, inverted to make RR<1 favor treatment, day 4.
|
|
relative VAS score, 40.0% better, RR 0.60, p = 0.001, treatment 100, control 100, day 2.
|
|
median 6-point ordinal scale, 100% lower, RR < 0.001, p < 0.001, treatment 100, control 100.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Dound et al., 16 Nov 2020, Randomized Controlled Trial, India, peer-reviewed, 5 authors, study period August 2020 - September 2020, this trial uses multiple treatments in the treatment arm (combined with vitamin C, vitamin K2-7, and l-selenomethionine) - results of individual treatments may vary, trial CTRI/2020/07/026839.
A Randomized, Comparative Clinical Study to Evaluate the Activity of Curvic TM Formulation for Management of SARS-COV-2 Infection (COVID-19)
Objective: To evaluate role of Curvic TM in the management of COVID-19 and its tolerability in comparison with standard treatment available from Ministry of Health and Family Welfare (MOHFW).
Study design and methodology: In a randomized, two arm, comparative study, COVID-19 positive patients (n=200) were enrolled during the month of August and September 2020 from Radiant Plus Hospital, Nashik, Maharashtra, India. The enrolled patients were administered either Curvic TM 500 mg tablets twice daily or given standard treatment protocol designed by MOHFW for 10 days. The patients were evaluated for decrease in oral temperature; SpO 2 and VAS score for cough and respiratory distress. The underlying mechanism was assessed by evaluating markers such as Interleukin-6, Homocysteine, D-dimer, Ferritin and C Reactive Protein.
Results: In the Curvic TM group, within two days, temperature reduced considerably to afebrile level in all the subjects and remained afebrile till end of study. While in the standard treatment group, temperature reduced to afebrile stage in all the subjects by 4 days and there after remained afebrile till end of study. The elevated levels of serum Interleukin-6, Homocysteine, Di-dimer, Ferritin and C-Reactive Protein at baseline dropped considerably within normal limits within 10 days in the Curvic TM group in comparison to standard treatment group.
Conclusion: Curvic TM has shown for the first time to be useful in management of COVID-19 positive patients along with improvement in the immunity, within 48 hours of starting treatment. Curvic TM was well tolerated without any side effects in any of the patients treated.
OPEN ACCESS Freely available online J Clin Trials, Vol.S3 No: 1000004 as compared standard treatment regimen.
CONCLUSION Curvic TM formulation, consisting of liposomal curcumin, vitamin C, K2-7, Selenomethionine and Zinc reduced inflammatory markers, improved patients disease condition to asymptomatic with complete relief from symptoms could be developed into a valid therapeutic option.
References
Abdollahi, Momtazi, Johnston, Sahebkar, Therapeutic effects of curcumin in inflammatory and immune-mediated diseases: A nature-made jack-of-all-trades?, J Cell Physiol
Ali, Elevated level of C-reactive protein may be an early marker to predict risk for severity of COVID-19, J Med Virol
Avasarala, Zhang, Liu, Wang, London, Correction: Curcumin modulates the inflammatory response and inhibits subsequent fibrosis in a mouse model of viral-induced acute respiratory distress syndrome, PLOS ONE
Bermano, Méplan, Mercer, Hesketh, Selenium and viral infection: Are there lessons for COVID-19?, Br J Nutr
Carr, Maggini, Vitamin C and immune function, Nutrients
Chen, Luo, Yuan, Wang, Yang, Vitamin C mitigates oxidative stress and tumor necrosis factor-alpha in severe community-acquired pneumonia and LPS-induced macrophages, Mediators Inflamm
Das, Anu, Birangal, Nikam, Pandey, Role of comorbidities like diabetes on severe acute respiratory syndrome coronavirus-2: A review, Life Sci
Dofferhoff, Piscaer, Schurgers, Visser, Van Den Ouweland, Reduced vitamin k status as a potentially modifiable risk factor of severe coronavirus disease 2019, Clin Infect Dis
Dound, Chaudhary, Chaudhary, Ahmad, Geesi, Preventive Strategies for Management of COVID-19 Using Natural Molecules
Dound, Chaudhary, Sehgal, Chaudhary, Dound, Plant based molecules for the management of Covid-19, J Infect Dis Ther
Feyaerts, Luyten, Vitamin C as prophylaxis and adjunctive medical treatment for COVID-19?, Nutrition
Fogarty, Townsend, Cheallaigh, Bergin, Martin-Loeches, COVID19 coagulopathy in caucasian patients, Br J Haematol
Hemilä, Vitamin C and SARS coronavirus, J Antimicrob Chemother
Jaiswal, Saxena, Classical Coronaviruses
Keogh-Brown, Smith, The economic impact of SARS: How does the reality match the predictions?, Health Policy
Kernan, Carcillo, Hyperferritinemia and inflammation, Int Immunol
Kieliszek, Lipinski, Selenium supplementation in the prevention of coronavirus infections (COVID-19), Med Hypotheses
Kunnumakkara, Anand, Aggarwal, Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signalling proteins, Cancer Lett
Li, De Clercq, Therapeutic options for the 2019 novel coronavirus (2019-nCoV), Nat Rev Drug Discov
Li, Huang, Wang, Wang, Liang et al., COVID-19 patients' clinical characteristics, discharge rate, and fatality rate of meta-analysis, J Med Virol
Lythgoe, Middleton, Ongoing clinical trials for the management of the COVID-19 pandemic, Trends Pharmacol Sci
Mehta, Mcauley, Brown, Sanchez, Tattersall, COVID-19: Consider cytokine storm syndromes and immunosuppression, Lancet
Moghadamtousi, Kadir, Hassandarvish, Tajik, Abubakar, A review on antibacterial, antiviral, and antifungal activity of curcumin, BioMed
Paliogiannis, Mangoni, Dettori, Nasrallah, Pintus, D-Dimer Concentrations and COVID-19 Severity: A Systematic Review and Meta-Analysis, Front Public Health
Paules, Coronavirus infections: More than just the common cold, JAMA
Pepys, Hirschfield, C-reactive protein: A critical update, J Clin Invest
Ponti, Ruini, Tomasi, Homocysteine as a potential predictor of cardiovascular risk in patients with COVID-19, Med Hypotheses
Puar, Shanmugam, Fan, Arfuso, Sethi, Evidence for the involvement of the master transcription factor NFκB in cancer initiation and progression, Biomed
Satarker, Nampoothiri, Structural proteins in severe acute respiratory syndrome coronavirus-2, Arch Med Res
Sato, Inaba, Yamashita, MK-7 and its effects on bone quality and strength, Nutrients
Song, From SARS to MERS, thrusting coronaviruses into the spotlight, Viruses
Ullrich, Nitsche, The SARS-CoV-2 main protease as drug target, Bioorg Med Chem Lett
Vargas-Vargas, Cortés-Rojo, Ferritin levels and COVID-19, Rev Panam Salud Publica
Vidali, Morosetti, Cossu, Luisi, Pancani, D-dimer as an indicator of prognosis in SARS-CoV-2 infection: A systematic review, ERJ Open Res
Wit, SARS and MERS: Recent insights into emerging coronaviruses, Nat Rev Microbiol
Zahedipour, Hosseini, Sathyapalan, Majeed, Jamialahmadi, Potential effects of curcumin in the treatment of COVID-19 infection, Phytother Res
Zhang, Saad, Taylor, Rayman, Selenium and selenoproteins in viral infection with potential relevance to COVID-19, Redox Biol
