Vitamin D3 Supplementation Enhances Symptom Relief and Modulates Microbiome and Immune Responses in Pediatric Post-Acute Sequelae of COVID-19: A Randomized Controlled Trial
et al., MDPI AG, doi:10.20944/preprints202602.0361.v1, NCT05633472, Feb 2026
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
RCT 33 pediatric outpatients with post-acute sequelae of COVID-19 (PASC) showing significant symptom improvement with vitamin D3 treatment.
Chen et al., 5 Feb 2026, Double Blind Randomized Controlled Trial, placebo-controlled, Taiwan, preprint, 15 authors, study period 1 November, 2022 - 31 December, 2023, trial NCT05633472 (history).
Contact: lshwu@hotmail.com (corresponding author), wangjy@mail.cmu.edu.tw.
Vitamin D3 Supplementation Enhances Symptom Relief and Modulates Microbiome and Immune Responses in Pediatric Post-Acute Sequelae of COVID-19: A Randomized Controlled Trial
doi:10.20944/preprints202602.0361.v1
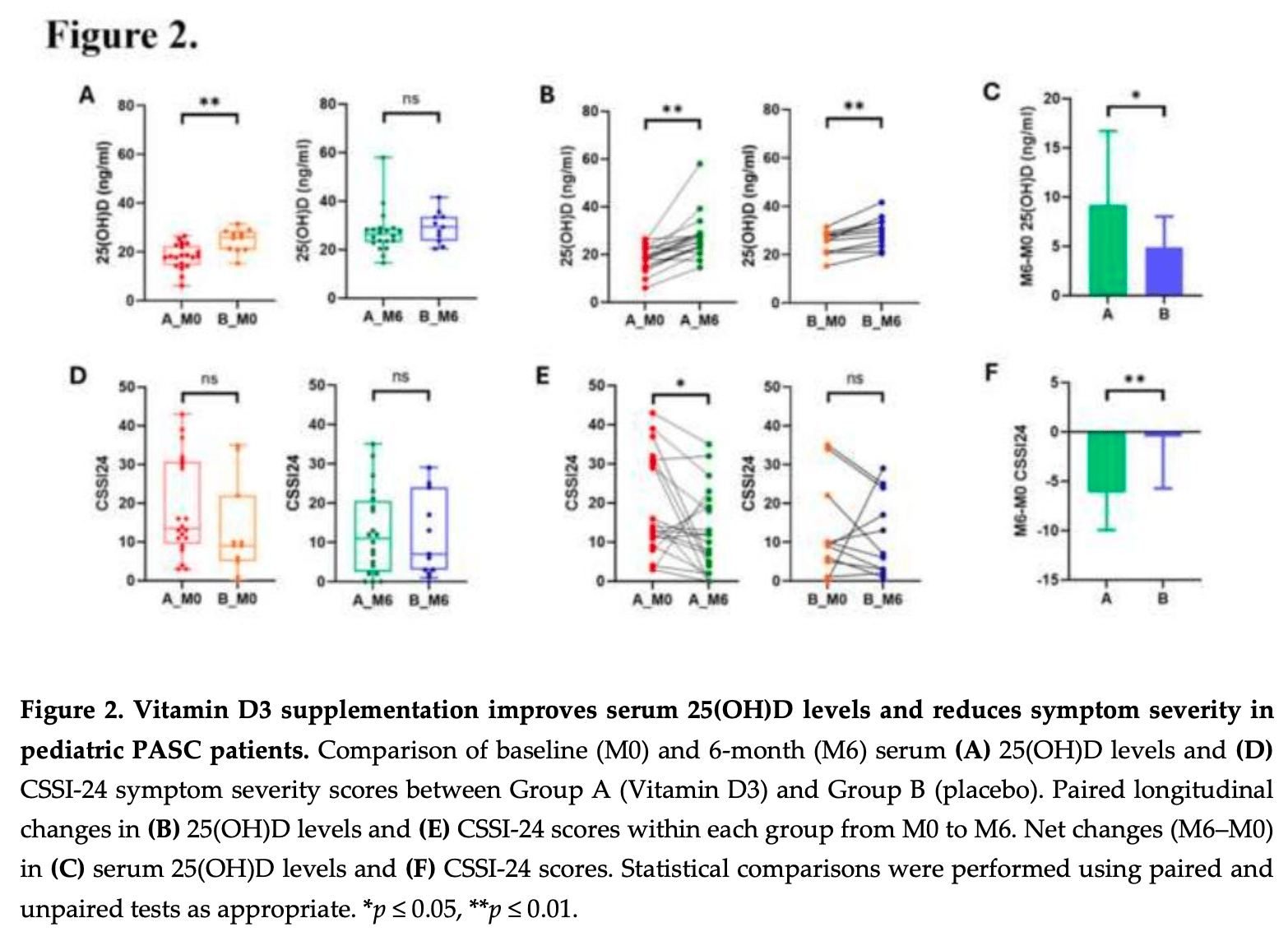
Background: Post-acute sequelae ofCOVID-19 (PASC) in children present persistent symptoms that impair quality of life. Vitamin D's immunomodulatory and microbiome-modulating properties suggest its potential to alleviate PASC symptoms. This study evaluated the efficacy of Vitamin D3 supplementation in alleviating symptom severity and to explore associated immunological and microbiome alterations in children with PASC. Methods: In a double-blind randomized controlled clinical trial, 33 children with PASC were assigned to receive 2,000 IU/day of oral vitamin D (Group A, n=21) or placebo (Group B, n=12) for 6 months. Serum 25-hydroxyvitamin D [25(OH)D] levels, symptom severity (Children's Somatic Symptom Inventory-24, CSSI-24), immune cytokines, and checkpoint proteins were assessed at baseline (M0) and 6 months (M6). Nasal and rectal microbiota were analyzed using 16S rRNA sequencing to evaluate composition and predict functional pathways. Results: Serum 25(OH)D increased significantly in Group A compared with Group B (p < 0.01). Group A demonstrated significant reductions in CSSI-24 scores (p < 0.05), with improvements in neuropsychiatric, respiratory and cardiovascular symptoms. Pro-inflammatory cytokines including IFN-γ (FDR = 0.01) and MIP-1α (FDR = 0.0004) decreased, while checkpoint proteins TIM-3 and HVEM increased (both FDR < 0.05). Nasal microbiota in Group A at M6 showed increased richness and enrichment of Sphingomonas, Paenibacillus, Ralstonia, and Sphingobium, with functional pathways related to xenobiotic metabolism altered after supplementation. Coprobacillus abundance in rectal samples positively correlated with 25(OH)D levels and negatively with IFN-γ (p = 0.02). Pooling participants from both time points across group A indicated that higher 25(OH)D levels were associated with lower symptom scores. Conclusions: Vitamin D3 supplementation improved
Authors' contributions: PCC, YLH, LSHW, and JYW designed the study. YLH, MHH, and YSH performed clinical data collections. PCC and YLH coordinated and supervised data collection. TL and YCC analyzed the microbiome data. PCC, YHC, CYL, XLL, HJT, and LSHW analyzed the clinical data. PCC wrote the initial draft of the manuscript. TL, LSHW, and JYW reviewed and revised the manuscript. WSK, HFK, and SDW provided critical assessments during the revision process leading to the final submitted manuscript. All authors have reviewed and approved the final version of this manuscript.
References
Akimbekov, Digel, Sherelkhan, Lutfor, Razzaque, Vitamin D and the Host-Gut Microbiome: A Brief Overview, Acta Histochem Cytochem
Baktash, Hosack, Patel, Shah, Kandiah et al., Vitamin D status and outcomes for hospitalised older patients with COVID-19, Postgrad Med J
Bellerba, Muzio, Gnagnarella, Facciotti, Chiocca et al., The Association between Vitamin D and Gut Microbiota: A Systematic Review of Human Studies, Nutrients
Cantorna, Mcdaniel, Bora, Chen, James, Vitamin D, immune regulation, the microbiota, and inflammatory bowel disease, Exp Biol Med
Castillo, Costa, Barrios, Díaz, Miranda et al., Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study, J Steroid Biochem Mol Biol
Cătălina, Gheorman, Gheorman, Forțofoiu, The Role of Neuroinflammation in the Comorbidity of Psychiatric Disorders and Internal Diseases, Healthcare
Davis, Assaf, Mccorkell, Wei, Low et al., Characterizing long COVID in an international cohort: 7 months of symptoms and their impact, EClinicalMedicine
Durack, Lynch, Nariya, Bhakta, Beigelman et al., Features of the bronchial bacterial microbiome associated with atopy, asthma, and responsiveness to inhaled corticosteroid treatment, J Allergy Clin Immunol
Giannos, Prokopidis, Gut dysbiosis and long COVID-19: Feeling gutted, J Med Virol
Guo, Yi, Wu, Lu, The microbiome in post-acute infection syndrome (PAIS), Comput Struct Biotechnol J
Holick, Vitamin D deficiency, N Engl J Med
Hsu, Chen, Tsai, Wei, Wu et al., Clinical Features and Vaccination Effects among Children with Post-Acute Sequelae of COVID-19 in Taiwan, Vaccines
Kaufman, Niles, Kroll, Bi, Holick, SARS-CoV-2 positivity rates associated with circulating 25-hydroxyvitamin D levels, PLoS One
Kenny, Townsend, Savinelli, Mallon, Long COVID: Clinical characteristics, proposed pathogenesis and potential therapeutic targets, Front Mol Biosci
Liu, Mak, Su, Yeoh, Lui et al., Gut microbiota dynamics in a prospective cohort of patients with post-acute COVID-19 syndrome, Gut
Louis, Flint, Formation of propionate and butyrate by the human colonic microbiota, Environ Microbiol
Martens, Gysemans, Verstuyf, Mathieu, Vitamin D's Effect on Immune Function, Nutrients
Martineau, Jolliffe, Hooper, Greenberg, Aloia et al., Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data, Bmj
Meo, Alhusseini, Ibrahim, Meo, Ahsan et al., Clinical manifestations of Long-COVID: an observational perspective, Front Med
Mercola, Grant, Wagner, Evidence Regarding Vitamin D and Risk of COVID-19 and Its Severity, Nutrients
Nalbandian, Sehgal, Gupta, Madhavan, Mcgroder et al., Post-acute COVID-19 syndrome, Nat Med
Ooi, Chen, Cantorna, Vitamin D regulation of immune function in the gut: why do T cells have vitamin D receptors?, Mol Aspects Med
Phetsouphanh, Darley, Wilson, Howe, Munier et al., Immunological dysfunction persists for 8 months following initial mild-to-moderate SARS-CoV-2 infection, Nat Immunol
Pichler, Coskun, Ortega-Arbulú, Conci, Wörheide et al., A 16S rRNA gene sequencing and analysis protocol for the Illumina MiniSeq platform, Microbiologyopen
Quesada-Gomez, Entrenas-Castillo, Bouillon, Vitamin D receptor stimulation to reduce acute respiratory distress syndrome (ARDS) in patients with coronavirus SARS-CoV-2 infections: Revised Ms SBMB 2020_166, J Steroid Biochem Mol Biol
Raj, Bruce, Anbalagan, Srinivasan, Chinnappan et al., COVID-19 influenced gut dysbiosis, post-acute sequelae, immune regulation, and therapeutic regimens, Front Cell Infect Microbiol
Rao, Gross, Mohandas, Stein, Case et al., Postacute Sequelae of SARS-CoV-2 in Children, Pediatrics
Singh, Rawat, Alwakeel, Sharif, Khodor, The potential role of vitamin D supplementation as a gut microbiota modifier in healthy individuals, Sci Rep
Stephenson, Shafran, Ladhani, Long COVID in children and adolescents, Curr Opin Infect Dis
Torres-Sánchez, Ruiz-Rodríguez, Ortiz, Moreno, Ampatzoglou et al., Exploring Next Generation Probiotics for Metabolic and Microbiota Dysbiosis Linked to Xenobiotic Exposure: Holistic Approach, Int J Mol Sci
Ullah, Gut-vitamin D interplay: key to mitigating immunosenescence and promoting healthy ageing, Immun Ageing
Walker, Beck, Garber, Children's Somatization Inventory: psychometric properties of the revised form (CSI-24), J Pediatr Psychol
Wykes, Lewin, Immune checkpoint blockade in infectious diseases, Nat Rev Immunol
Yong, Long COVID or post-COVID-19 syndrome: putative pathophysiology, risk factors, and treatments, Infect Dis (Lond)
Álvarez-Santacruz, Tyrkalska, Candel, The Microbiota in Long COVID, Int J Mol Sci
DOI record:
{
"DOI": "10.20944/preprints202602.0361.v1",
"URL": "http://dx.doi.org/10.20944/preprints202602.0361.v1",
"abstract": "<jats:p>Background :Post-acute sequelae of COVID-19 (PASC) in children present persistent symptoms that impair quality of life. Vitamin D’s immunomodulatory and microbiome-modulating properties suggest its potential to alleviate PASC symptoms. This study evaluated the efficacy of Vitamin D3 supplementation in alleviating symptom severity and to explore associated immunological and microbiome alterations in children with PASC. Methods: In a double-blind randomized controlled clinical trial, 33 children with PASC were assigned to receive 2,000 IU/day of oral vitamin D (Group A, n=21) or placebo (Group B, n=12) for 6 months. Serum 25-hydroxyvitamin D [25(OH)D] levels, symptom severity (Children’s Somatic Symptom Inventory-24, CSSI-24), immune cytokines, and checkpoint proteins were assessed at baseline (M0) and 6 months (M6). Nasal and rectal microbiota were analyzed using 16S rRNA sequencing to evaluate composition and predict functional pathways. Results: Serum 25(OH)D increased significantly in Group A compared with Group B (p &lt; 0.01). Group A demonstrated significant reductions in CSSI-24 scores (p &lt; 0.05), with improvements in neuropsychiatric, respiratory and cardiovascular symptoms. Pro-inflammatory cytokines including IFN-γ (FDR = 0.01) and MIP-1α (FDR = 0.0004) decreased, while checkpoint proteins TIM-3 and HVEM increased (both FDR &lt; 0.05). Nasal microbiota in Group A at M6 showed increased richness and enrichment of Sphingomonas, Paenibacillus, Ralstonia, and Sphingobium, with functional pathways related to xenobiotic metabolism altered after supplementation. Coprobacillus abundance in rectal samples positively correlated with 25(OH)D levels and negatively with IFN-γ (p = 0.02). Pooling participants from both time points across group A indicated that higher 25(OH)D levels were associated with lower symptom scores. Conclusions: Vitamin D3 supplementation improved symptom severity, modulated immune responses, and altered microbiota profiles in children with PASC. These findings support Vitamin D3 as a potential adjunctive therapy for pediatric PASC and highlight host–microbe–immune interactions as therapeutic targets.</jats:p>",
"accepted": {
"date-parts": [
[
2026,
2,
4
]
]
},
"author": [
{
"affiliation": [],
"family": "Chen",
"given": "Pei-Chi",
"sequence": "first"
},
{
"affiliation": [],
"family": "Liu",
"given": "Tsunglin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chuang",
"given": "Yu-Ching",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-3010-2767",
"affiliation": [],
"authenticated-orcid": false,
"family": "Hsu",
"given": "Yu-Lung",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chen",
"given": "Yen-Hsi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lin",
"given": "Chih-Yu",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hsieh",
"given": "Miao-Hsi",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ho",
"given": "Yu-Shan",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0000-3860-975X",
"affiliation": [],
"authenticated-orcid": false,
"family": "Liu",
"given": "Xiao-Ling",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kuo",
"given": "Wen-Shuo",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kao",
"given": "Hui-Fang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Shulhn-Der",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-5338-6117",
"affiliation": [],
"authenticated-orcid": false,
"family": "Tsai",
"given": "Hui-Ju",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0001-6275-8368",
"affiliation": [],
"authenticated-orcid": false,
"family": "Wu",
"given": "Lawrence Shih-Hsin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Jiu-Yao",
"sequence": "additional"
}
],
"container-title": [],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
2,
9
]
],
"date-time": "2026-02-09T00:12:46Z",
"timestamp": 1770595966000
},
"deposited": {
"date-parts": [
[
2026,
2,
9
]
],
"date-time": "2026-02-09T00:13:02Z",
"timestamp": 1770595982000
},
"group-title": "Medicine and Pharmacology",
"indexed": {
"date-parts": [
[
2026,
2,
9
]
],
"date-time": "2026-02-09T17:05:14Z",
"timestamp": 1770656714425,
"version": "3.49.0"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
2,
5
]
]
},
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0",
"content-version": "unspecified",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
5
]
],
"date-time": "2026-02-05T00:00:00Z",
"timestamp": 1770249600000
}
}
],
"member": "1968",
"original-title": [],
"posted": {
"date-parts": [
[
2026,
2,
5
]
]
},
"prefix": "10.20944",
"published": {
"date-parts": [
[
2026,
2,
5
]
]
},
"publisher": "MDPI AG",
"reference-count": 0,
"references-count": 0,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.preprints.org/manuscript/202602.0361/v1"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"subtype": "preprint",
"title": "Vitamin D3 Supplementation Enhances Symptom Relief and Modulates Microbiome and Immune Responses in Pediatric Post-Acute Sequelae of COVID-19: A Randomized Controlled Trial",
"type": "posted-content"
}
