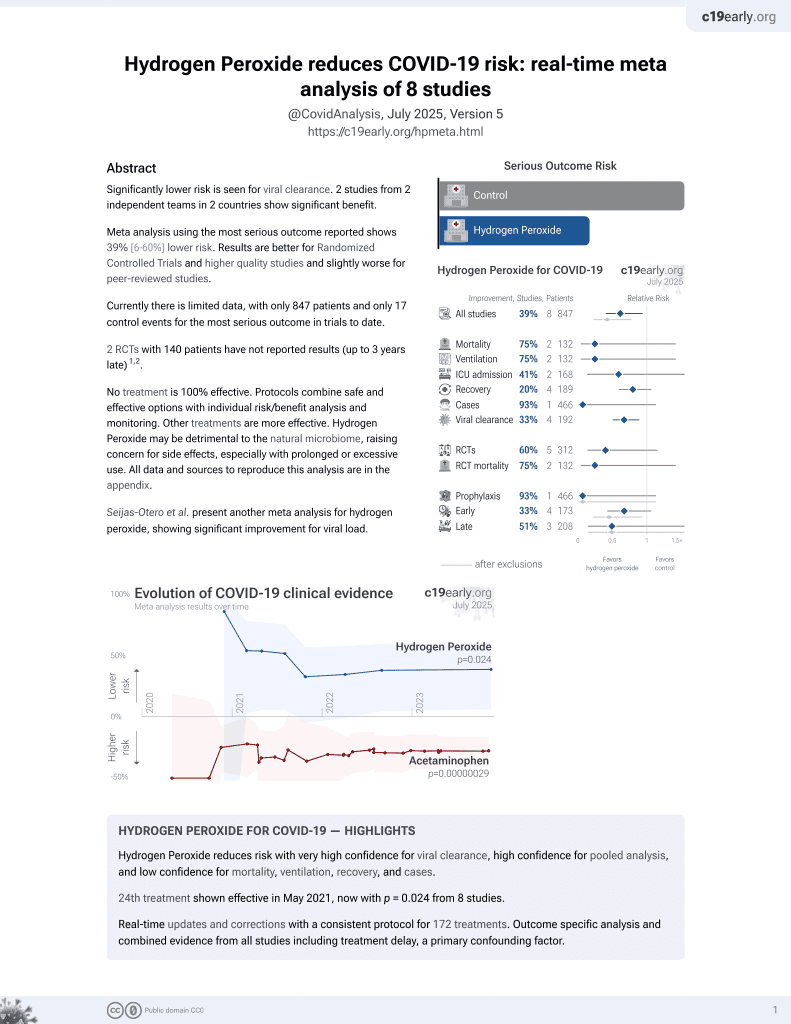
Estimating salivary carriage of severe acute respiratory syndrome coronavirus 2 in nonsymptomatic people and efficacy of mouthrinse in reducing viral load
et al., The Journal of the American Dental Association, doi:10.1016/j.adaj.2021.05.021, NCT04603794, Jun 2021
23rd treatment shown to reduce risk in
May 2021, now with p = 0.024 from 8 studies.
Lower risk for viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
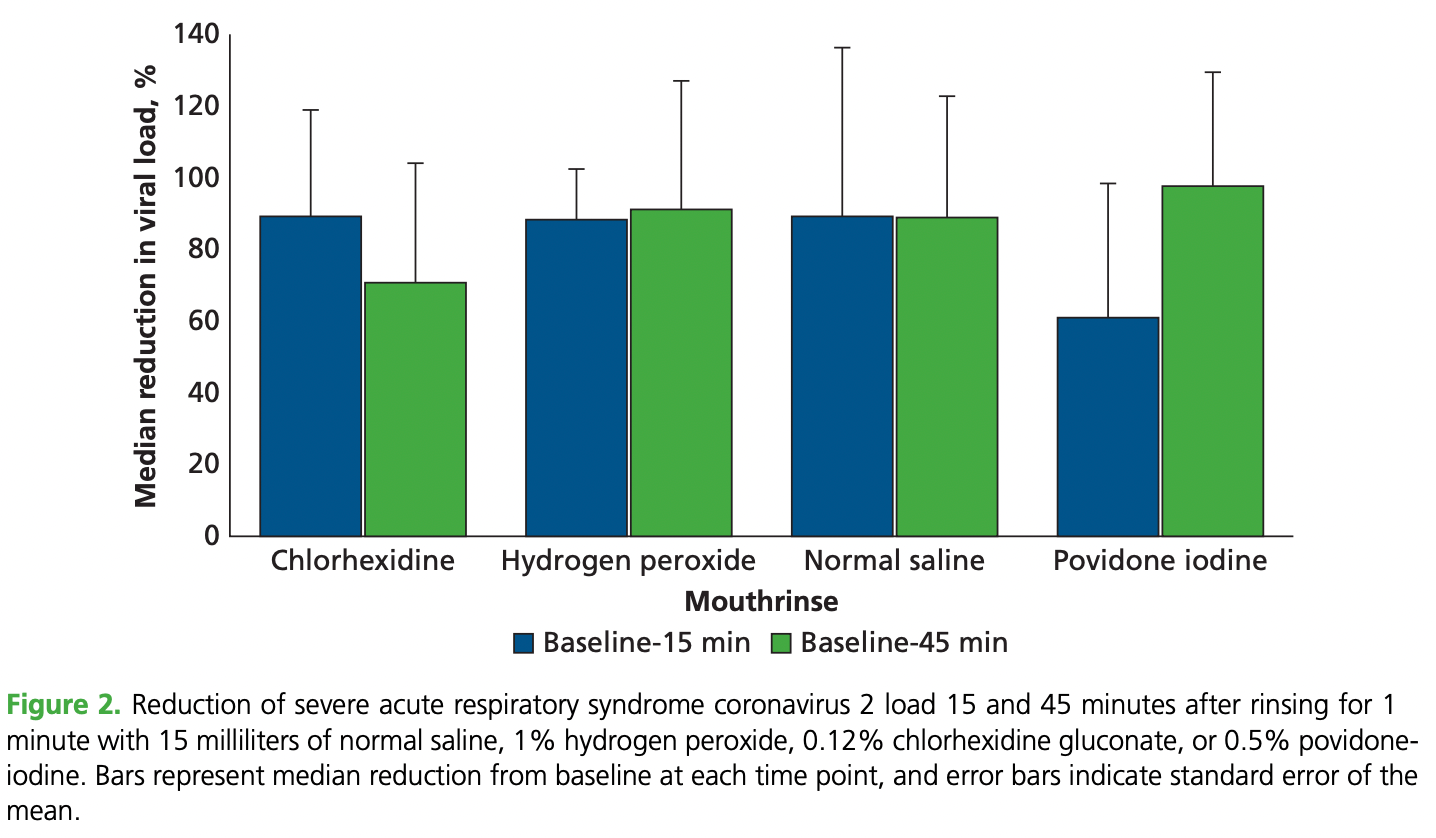
Small RCT comparing mouthrinsing with saline, 1% hydrogen peroxide, 0.12% chlorhexidine, and 0.5% povidone-iodine, showing significant reductions in salivary viral load for all 4 mouthrinses at 15 and 45 minutes after rinsing.
Analysis of short-term changes in viral load using PCR may not detect
effective treatments because PCR is unable to differentiate between intact
infectious virus and non-infectious or destroyed virus particles. For example
Tarragó-Gil, Alemany perform RCTs with cetylpyridinium chloride
(CPC) mouthwash that show no difference in PCR viral load, however there was
significantly increased detection of SARS-CoV-2 nucleocapsid protein,
indicating viral lysis. CPC inactivates SARS-CoV-2 by degrading its membrane,
exposing the nucleocapsid of the virus. To better estimate changes in viral
load and infectivity, methods like viral culture that can
differentiate intact vs. degraded virus are preferred.
Study covers hydrogen peroxide and povidone-iodine.
Chaudhary et al., 11 Jun 2021, Randomized Controlled Trial, USA, peer-reviewed, 17 authors, trial NCT04603794 (history).
Estimating salivary carriage of severe acute respiratory syndrome coronavirus 2 in nonsymptomatic people and efficacy of mouthrinse in reducing viral load
The Journal of the American Dental Association, doi:10.1016/j.adaj.2021.05.021
Background. Many people infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) never develop substantial symptoms. With more than 34 million people in the United States already infected and highly transmissible variants rapidly emerging, it is highly probable that post-and presymptomatic people will form an important fraction of those seeking dental care. Salivary carriage rates in these populations are not known. Moreover, although preventing transmission is critical for controlling spread, the efficacy of mouthrinses in reducing oral viral load is poorly studied. Methods. The authors recruited 201 asymptomatic, presymptomatic, postsymptomatic, and symptomatic people and measured copy numbers of SARS-CoV-2 in unstimulated saliva using realtime reverse transcriptase quantitative polymerase chain reaction. Subsequently, the authors inducted 41 symptomatic people into a randomized, triple-blinded study and instructed them to rinse with saline, 1% hydrogen peroxide, 0.12% chlorhexidine, or 0.5% povidone-iodine for 60 seconds. The authors measured viral load 15 and 45 minutes after rinsing. Results. Salivary SARS-CoV-2 was detected in 23% of asymptomatic, 60% of postsymptomatic, and 28% of presymptomatic participants. Neither carriage rate nor viral load correlated with COVID-19 symptomatology, age, sex, or race or ethnicity. All 4 mouthrinses decreased viral load by 61% through 89% at 15 minutes and by 70% through 97% at 45 minutes. The extent of reduction correlated significantly with initial viral load. Conclusions. Nonsymptomatic people can pose a risk of transmitting the virus, and mouthrinses are simple and efficacious means of reducing this risk, especially when the load is less than 10 4 copies per milliliter. Practical Implications. At a time when resources are stretched, the findings of this study contribute to evidence-based selection of personal protection equipment and simple infectioncontrol practices to reduce contagion at source. This clinical trial was registered at ClinicalTrials.gov. The registration number is NCT04603794.
References
Dr, Woolsey, General Practice Residency Program
Feres, Figueiredo, Faveri, Stewart, De Vizio, The effectiveness of a preprocedural mouthrinse containing cetylpyridinium chloride in reducing bacteria in the dental office, JADA
García-Caballero, Quintas, Prada-López, Seoane, Donos et al., Chlorhexidine substantivity on salivary flora and plaque-like biofilm: an in situ model, PLoS One
Gottsauner, Michaelides, Schmidt, A prospective clinical pilot study on the effects of a hydrogen peroxide mouthrinse on the intraoral viral load of SARS-CoV-2, Clin Oral Investig
Goyal, Reeves, Cardozo-Ojeda, Schiffer, Mayer, Viral load and contact heterogeneity predict SARS-CoV-2 transmission and super-spreading events, eLife
Hassandarvish, Tiong, Mohamed, In vitro virucidal activity of povidone iodine gargle and mouthwash against SARS-CoV-2: implications for dental practice, Br Dent J. Published online, doi:10.1038/s41415-020-2402-0
Hoffmann, Kleine-Weber, Schroeder, SARS-CoV-2 Cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor, Cell
Joshi, Padhye, Gupta, Efficacy of two pre-procedural rinses at two different temperatures in reducing aerosol contamination produced during ultrasonic scaling in a dental set-up: a microbiological study, J Int Acad Periodontol
Kalenderian, Xiao, Spallek, Franklin, Olsen et al., Analysis of anxiety-related factors amongst frontline dental staff during the COVID-19 pandemic in Yichang, China, BMC Oral Health
Liu, Yi, Zhang, Lv, Zhu et al., Viral Load dynamics in sputum and nasopharyngeal swab in patients with COVID-19, J Dent Res
Meethil, Saraswat, Chaudhary, Dabdoub, Kumar et al., Estimating COVID-19 prevalence and infection control practices among US dentists, J Dent Res, doi:10.1177/00220345211015948
Murakami, Ghatak-Roy, Popova, COVID-19 infection among emergency department healthcare providers in a large tertiary academic medical center following the peak of the pandemic, Am J Emerg Med
Navazesh, Methods for collecting saliva, Ann N Y Acad Sci
Peditto, Scapellato, Marcianò, Costa, Oteri et al., SARS-CoV-2 prevalence in saliva and gastric and intestinal fluid in patients undergoing gastrointestinal endoscopy in COVID-19 endemic areas: prospective cross-sectional study in Japan, Int J Environ Res Public Health, doi:10.1111/den.13945
Seneviratne, Balan, Ko, Efficacy of commercial mouth-rinses on SARS-CoV-2 viral load in saliva: randomized control trial in Singapore, Infection
Srinivasan, Zunt, Goldblatt, Oral epithelial expression of angiotensin converting enzyme-2: implications for COVID-19 diagnosis and prognosis, bioRxiv, doi:10.1101/2020.06.22.165035
Teo, Choudhury, Tan, Saliva is more sensitive than nasopharyngeal or nasal swabs for diagnosis of asymptomatic and mild COVID-19 infection, Sci Rep
Vogels, Brackney, Kalinich, High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa, Int J Oral Sci
Wölfel, Corman, Guggemos, Virological assessment of hospitalized patients with COVID-2019, Nature
Xu, Wang, Hoskin, Differential effects on antiseptic mouth rinses on SARS-CoV-2 infectivity in vitro, Pathogens
DOI record:
{
"DOI": "10.1016/j.adaj.2021.05.021",
"ISSN": [
"0002-8177"
],
"URL": "http://dx.doi.org/10.1016/j.adaj.2021.05.021",
"alternative-id": [
"S000281772100355X"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Estimating salivary carriage of severe acute respiratory syndrome coronavirus 2 in nonsymptomatic people and efficacy of mouthrinse in reducing viral load"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "The Journal of the American Dental Association"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.adaj.2021.05.021"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 American Dental Association. All rights reserved."
}
],
"author": [
{
"affiliation": [],
"family": "Chaudhary",
"given": "PremPrashant",
"sequence": "first"
},
{
"affiliation": [],
"family": "Melkonyan",
"given": "Arsen",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Meethil",
"given": "Archana",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Saraswat",
"given": "Shweta",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hall",
"given": "David L.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cottle",
"given": "James",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wenzel",
"given": "Mark",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ayouty",
"given": "Nadine",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Bense",
"given": "Spenser",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Casanova",
"given": "Fabiola",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chaney",
"given": "Matthew",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chase",
"given": "Hannah",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hermel",
"given": "Rebecca",
"sequence": "additional"
},
{
"affiliation": [],
"family": "McClement",
"given": "Matthew",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sesson",
"given": "Claire",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Woolsey",
"given": "Bryce",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Kumar",
"given": "Purnima",
"sequence": "additional"
}
],
"container-title": [
"The Journal of the American Dental Association"
],
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.jp",
"jada.ada.org",
"clinicalkey.com",
"clinicalkey.es",
"clinicalkey.com.au",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2021,
6,
11
]
],
"date-time": "2021-06-11T09:19:18Z",
"timestamp": 1623403158000
},
"deposited": {
"date-parts": [
[
2021,
10,
25
]
],
"date-time": "2021-10-25T22:14:40Z",
"timestamp": 1635200080000
},
"indexed": {
"date-parts": [
[
2021,
12,
20
]
],
"date-time": "2021-12-20T19:29:19Z",
"timestamp": 1640028559970
},
"is-referenced-by-count": 3,
"issn-type": [
{
"type": "print",
"value": "0002-8177"
}
],
"issue": "11",
"issued": {
"date-parts": [
[
2021,
11
]
]
},
"journal-issue": {
"issue": "11",
"published-print": {
"date-parts": [
[
2021,
11
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
11,
1
]
],
"date-time": "2021-11-01T00:00:00Z",
"timestamp": 1635724800000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S000281772100355X?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S000281772100355X?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "903-908",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
11
]
]
},
"published-print": {
"date-parts": [
[
2021,
11
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"author": "Kalenderian",
"key": "10.1016/j.adaj.2021.05.021_bib1"
},
{
"DOI": "10.1186/s12903-020-01335-9",
"article-title": "Analysis of anxiety-related factors amongst frontline dental staff during the COVID-19 pandemic in Yichang, China",
"author": "Zhao",
"doi-asserted-by": "crossref",
"first-page": "342",
"issue": "1",
"journal-title": "BMC Oral Health",
"key": "10.1016/j.adaj.2021.05.021_bib3",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.3390/ijerph17093325",
"article-title": "Dentistry during the COVID-19 epidemic: an Italian workflow for the management of dental practice",
"author": "Peditto",
"doi-asserted-by": "crossref",
"first-page": "3325",
"issue": "9",
"journal-title": "Int J Environ Res Public Health",
"key": "10.1016/j.adaj.2021.05.021_bib4",
"volume": "17",
"year": "2020"
},
{
"DOI": "10.2139/ssrn.3700893",
"doi-asserted-by": "crossref",
"key": "10.1016/j.adaj.2021.05.021_bib6",
"unstructured": "Miyake S, Ashikari K, Kato S, et al. SARS-CoV-2 prevalence in saliva and gastric and intestinal fluid in patients undergoing gastrointestinal endoscopy in COVID-19 endemic areas: prospective cross-sectional study in Japan. Dig Endosc. Published online February 6, 2021. https://doi.org/10.1111/den.13945"
},
{
"DOI": "10.1016/j.ajem.2020.11.064",
"article-title": "COVID-19 infection among emergency department healthcare providers in a large tertiary academic medical center following the peak of the pandemic",
"author": "Murakami",
"doi-asserted-by": "crossref",
"first-page": "27",
"journal-title": "Am J Emerg Med",
"key": "10.1016/j.adaj.2021.05.021_bib7",
"volume": "40",
"year": "2021"
},
{
"DOI": "10.1038/s41598-021-82787-z",
"article-title": "Saliva is more sensitive than nasopharyngeal or nasal swabs for diagnosis of asymptomatic and mild COVID-19 infection",
"author": "Teo",
"doi-asserted-by": "crossref",
"first-page": "3134",
"issue": "1",
"journal-title": "Sci Rep",
"key": "10.1016/j.adaj.2021.05.021_bib8",
"volume": "11",
"year": "2021"
},
{
"article-title": "The effectiveness of a preprocedural mouthrinse containing cetylpyridinium chloride in reducing bacteria in the dental office",
"author": "Feres",
"first-page": "415",
"issue": "4",
"journal-title": "JADA",
"key": "10.1016/j.adaj.2021.05.021_bib9",
"volume": "141",
"year": "2010"
},
{
"DOI": "10.1371/journal.pone.0083522",
"article-title": "Chlorhexidine substantivity on salivary flora and plaque-like biofilm: an in situ model",
"author": "García-Caballero",
"doi-asserted-by": "crossref",
"issue": "12",
"journal-title": "PLoS One",
"key": "10.1016/j.adaj.2021.05.021_bib10",
"volume": "8",
"year": "2013"
},
{
"article-title": "Efficacy of two pre-procedural rinses at two different temperatures in reducing aerosol contamination produced during ultrasonic scaling in a dental set-up: a microbiological study",
"author": "Joshi",
"first-page": "138",
"issue": "4",
"journal-title": "J Int Acad Periodontol",
"key": "10.1016/j.adaj.2021.05.021_bib11",
"volume": "19",
"year": "2017"
},
{
"article-title": "In vitro virucidal activity of povidone iodine gargle and mouthwash against SARS-CoV-2: implications for dental practice",
"author": "Hassandarvish",
"journal-title": "Br Dent J. Published online December",
"key": "10.1016/j.adaj.2021.05.021_bib12",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.3390/pathogens10030272",
"article-title": "Differential effects on antiseptic mouth rinses on SARS-CoV-2 infectivity in vitro",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "272",
"issue": "3",
"journal-title": "Pathogens",
"key": "10.1016/j.adaj.2021.05.021_bib13",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1007/s00784-020-03549-1",
"article-title": "A prospective clinical pilot study on the effects of a hydrogen peroxide mouthrinse on the intraoral viral load of SARS-CoV-2",
"author": "Gottsauner",
"doi-asserted-by": "crossref",
"first-page": "3707",
"issue": "10",
"journal-title": "Clin Oral Investig",
"key": "10.1016/j.adaj.2021.05.021_bib14",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1007/s15010-020-01563-9",
"article-title": "Efficacy of commercial mouth-rinses on SARS-CoV-2 viral load in saliva: randomized control trial in Singapore",
"author": "Seneviratne",
"doi-asserted-by": "crossref",
"first-page": "305",
"issue": "2",
"journal-title": "Infection",
"key": "10.1016/j.adaj.2021.05.021_bib15",
"volume": "49",
"year": "2021"
},
{
"key": "10.1016/j.adaj.2021.05.021_bib25",
"unstructured": "American Dental Association. Return to work toolkit. Accessed January 6, 2020. https://success.ada.org/∼/media/CPS/Files/Open%20Files/ADA_Return_to_Work_Toolkit.pdf"
},
{
"DOI": "10.1111/j.1749-6632.1993.tb18343.x",
"article-title": "Methods for collecting saliva",
"author": "Navazesh",
"doi-asserted-by": "crossref",
"first-page": "72",
"issue": "1",
"journal-title": "Ann N Y Acad Sci",
"key": "10.1016/j.adaj.2021.05.021_bib16",
"volume": "694",
"year": "1993"
},
{
"author": "Vogels",
"key": "10.1016/j.adaj.2021.05.021_bib17"
},
{
"DOI": "10.1038/s41368-020-0074-x",
"article-title": "High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "8",
"issue": "1",
"journal-title": "Int J Oral Sci",
"key": "10.1016/j.adaj.2021.05.021_bib18",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1101/2020.06.22.165035",
"doi-asserted-by": "crossref",
"key": "10.1016/j.adaj.2021.05.021_bib19",
"unstructured": "Srinivasan M, Zunt SL, Goldblatt LI. Oral epithelial expression of angiotensin converting enzyme-2: implications for COVID-19 diagnosis and prognosis. bioRxiv. June 23, 2020. Preprint. Version 1. Withdrawal status: active. https://doi.org/10.1101/2020.06.22.165035"
},
{
"DOI": "10.1016/j.cell.2020.02.052",
"article-title": "SARS-CoV-2 Cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor",
"author": "Hoffmann",
"doi-asserted-by": "crossref",
"first-page": "271",
"issue": "2",
"journal-title": "Cell",
"key": "10.1016/j.adaj.2021.05.021_bib20",
"volume": "181",
"year": "2020"
},
{
"DOI": "10.7554/eLife.63537",
"article-title": "Viral load and contact heterogeneity predict SARS-CoV-2 transmission and super-spreading events",
"author": "Goyal",
"doi-asserted-by": "crossref",
"journal-title": "eLife",
"key": "10.1016/j.adaj.2021.05.021_bib21",
"volume": "10",
"year": "2021"
},
{
"DOI": "10.1038/s41586-020-2196-x",
"article-title": "Virological assessment of hospitalized patients with COVID-2019",
"author": "Wölfel",
"doi-asserted-by": "crossref",
"first-page": "465",
"issue": "7809",
"journal-title": "Nature",
"key": "10.1016/j.adaj.2021.05.021_bib22",
"volume": "581",
"year": "2020"
},
{
"DOI": "10.1177/00220345211015948",
"article-title": "Sources of SARS-CoV-2 and other microorganisms in dental aerosols",
"author": "Meethil",
"doi-asserted-by": "crossref",
"first-page": "817",
"issue": "8",
"journal-title": "J Dent Res",
"key": "10.1016/j.adaj.2021.05.021_bib26",
"volume": "100",
"year": "2021"
},
{
"article-title": "Estimating COVID-19 prevalence and infection control practices among US dentists",
"author": "Estrich",
"first-page": "815",
"issue": "11",
"journal-title": "JADA",
"key": "10.1016/j.adaj.2021.05.021_bib23",
"volume": "151",
"year": "2020"
},
{
"DOI": "10.1177/0022034520946251",
"article-title": "Viral Load dynamics in sputum and nasopharyngeal swab in patients with COVID-19",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "1239",
"issue": "11",
"journal-title": "J Dent Res",
"key": "10.1016/j.adaj.2021.05.021_bib24",
"volume": "99",
"year": "2020"
}
],
"reference-count": 24,
"references-count": 24,
"relation": {},
"score": 1,
"short-container-title": [
"The Journal of the American Dental Association"
],
"short-title": [],
"source": "Crossref",
"subject": [
"General Dentistry"
],
"subtitle": [],
"title": [
"Estimating salivary carriage of severe acute respiratory syndrome coronavirus 2 in nonsymptomatic people and efficacy of mouthrinse in reducing viral load"
],
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "152"
}
chaudhary