Canadian adaptive platform trial of treatments for COVID in community settings (CanTreatCOVID): recruitment strategies of a decentralized, national randomized controlled trial for acute SARS-CoV-2
et al., Frontiers in Public Health, doi:10.3389/fpubh.2026.1698604, NCT05614349, Feb 2026
Recruitment strategy analysis for the decentralized adaptive platform trial CanTreatCOVID. This is the second paper published reporting secondary analyses, while authors have not yet published the actual efficacy results of the trial.
Astorga et al., 11 Feb 2026, Randomized Controlled Trial, Canada, peer-reviewed, 5 authors, study period January 2023 - September 2024, trial NCT05614349 (history).
Contact: benita.hosseini@utoronto.ca.
Canadian adaptive platform trial of treatments for COVID in community settings (CanTreatCOVID): recruitment strategies of a decentralized, national randomized controlled trial for acute SARS-CoV-2
Frontiers in Public Health, doi:10.3389/fpubh.2026.1698604
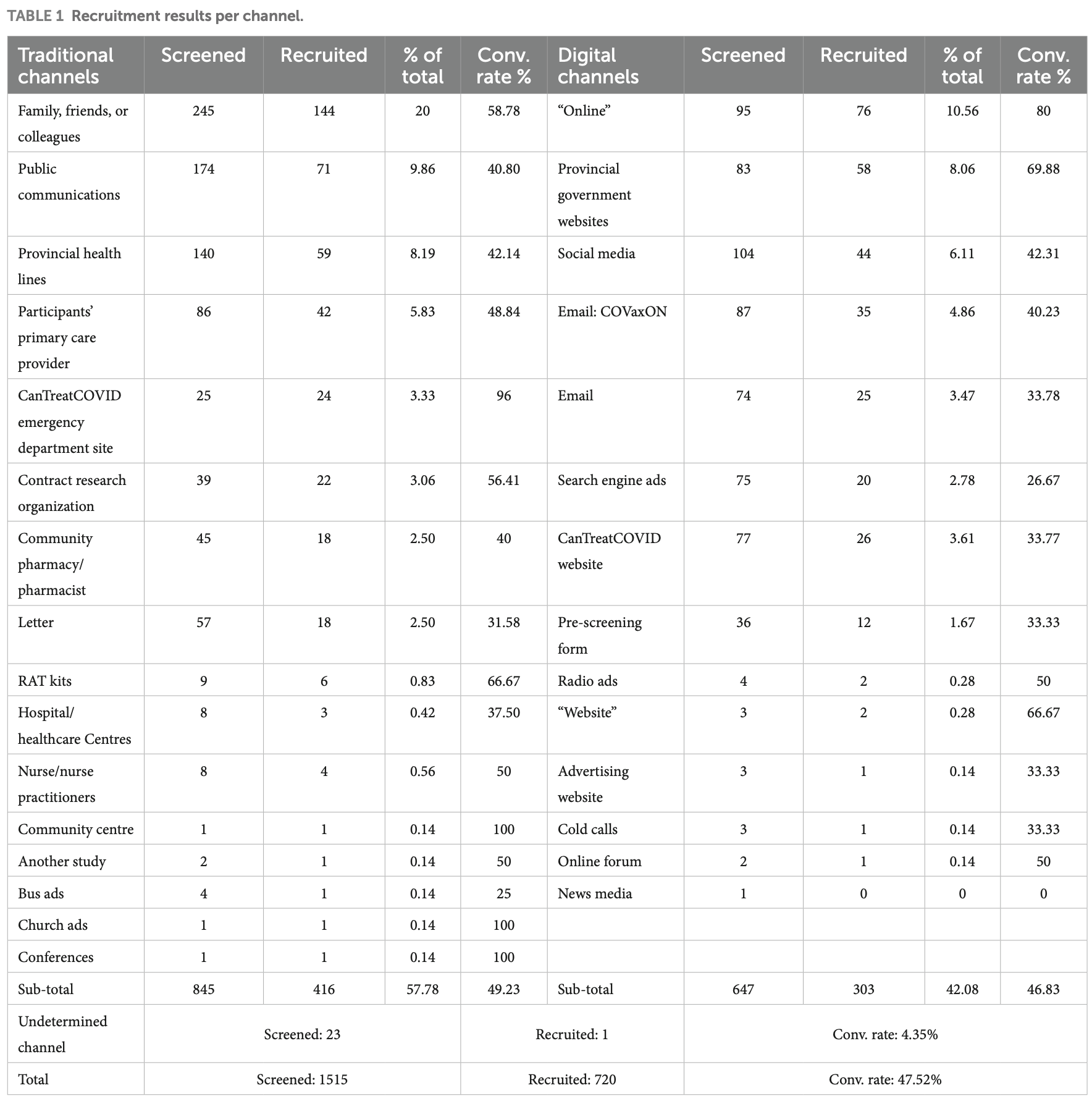
Background: Recruitment remains a challenge in clinical trials. This study describes the use of digital and traditional recruitment channels in a national, community-based adaptive platform trial for COVID-19 in Canada. Methods: Self-reported recruitment sources were collected from participants of a remote, national adaptive platform trial of COVID-19 treatments conducted across six Canadian provinces using a secure web-based application. Recruitment channels were analyzed using descriptive statistics, chi-square tests, and logistic regression to explore associations with province. Results: From January 2023 to September 2024, 1,515 participants completed the pre-screening process, and 720 were randomized. Of them, 416 were recruited through traditional channels, 303 through digital channels, and one through an undetermined source. Recruitment channels varied by province (χ 2 (5) = 81.30, p < 0.001; Cramér's V = 0.34), and digital channels were associated with higher odds of recruitment compared with traditional channels (adjusted OR = 2.78, 95% CI 2.20-3.52). The most frequently reported referral source was 'family, friends, or colleagues' (20%), followed by provincial government websites and health lines (16.25%), online channels (10.56%), and public communications (9.86%). Conclusion: Combining traditional and digital recruitment methods supports recruitment. Trials can raise awareness of the study by leveraging both digital and traditional channels and can move information to action through trusted social groups and institutions. Further research is needed to evaluate the cost-effectiveness, reach, and demographic differences across platforms, informing recruitment strategies in future decentralized and adaptive trials beyond COVID-19.
Frontiers in Public Health 07 frontiersin.org The increased utilization of online platforms during the COVID-19 pandemic introduced affordances for mixed traditional and digital recruitment in trials (13, 24) . While leveraging various channels reaches broad audiences, understanding geographical and other relevant contexts is equally important for successful recruitment (22, 23) . The observed variation in traditional and digital recruitment channel distribution across CanTreatCOVID sites aligns with the moderate association identified in the analysis. Two of the provinces that generated 77% of the recruitment results had different primary sources: traditional for ON and digital for BC, whereas other sites relied on traditional channels. The heterogeneity across provinces suggests that geographical and other characteristics influence recruitment rather than uniform reliance on a single recruitment strategy, and highlights the potential complementary role of traditional and digital channels. A key limitation of this analysis is that recruitment strategies were not randomized, and participants may have been exposed to multiple recruitment channels. As a result, the findings should be interpreted as descriptive comparisons and associations rather than causal or inferential effects of specific recruitment approaches. This study reports on a wide range of recruitment channels and provides insights into their effectiveness in engaging with potential participants. Future..
References
Amstutz, Schandelmaier, Frei, Surina, Agarwal et al., Discontinuation and non-publication of randomised clinical trials supported by the main public funding body in Switzerland: a retrospective cohort study, BMJ Open, doi:10.1136/bmjopen-2017-016216
Aysola, Tahirovic, Troxel, Asch, Gangemi et al., A randomized controlled trial of opt-in versus opt-out enrollment into a diabetes behavioral intervention, Am J Health Promot: AJHP, doi:10.1177/0890117116671673
Bengtsson, How to plan and perform a qualitative study using content analysis, Nurs Open, doi:10.1016/j.npls.2016.01.001
Bikou, Deligianni, Dermiki-Gkana, Liappas, Teriús-Padrón et al., Improving participant recruitment in clinical trials: comparative analysis of innovative digital platforms, J Med Internet Res, doi:10.2196/60504
Boulware, Pullen, Bangdiwala, Pastick, Lofgren et al., A randomized trial of hydroxychloroquine as postexposure prophylaxis for COVID-19, N Engl J Med, doi:10.1056/nejmoa2016638
Briel, Elger, Mclennan, Schandelmaier, Elm et al., Exploring reasons for recruitment failure in clinical trials: a qualitative study with clinical trial stakeholders in Switzerland, Germany, and Canada, Trials, doi:10.1186/s13063-021-05818-0
Buckley, Mchugh, Riordan, What works to recruit general practices to trials? A rapid review, HRB Open Res, doi:10.12688/hrbopenres.13650.1
Buenconsejo, Liao, Lin, Singh, Cooner et al., Platform trials to evaluate the benefit-risk of COVID-19 therapeutics: successes, learnings, and recommendations for future pandemics, Contemp Clin Trials, doi:10.1016/j.cct.2023.107292
Cunningham-Erves, Kusnoor, Villalta-Gil, Stallings, Ichimura et al., Development and pilot implementation of guidelines for culturally tailored research recruitment materials for African Americans and Latinos, BMC Med Res Methodol, doi:10.1186/s12874-022-01724-4
Firmino-Machado, Varela, Mendes, Moreira, Lunet et al., Stepwise strategy to improve cervical cancer screening adherence (SCAN-cervical Cancer) -automated text messages, phone calls and reminders: population based randomized controlled trial, Prev Med, doi:10.1016/j.ypmed.2018.06.004
Frampton, Shepherd, Pickett, Griffiths, Wyatt, Digital tools for the recruitment and retention of participants in randomised controlled trials: a systematic map, Trials, doi:10.1186/s13063-020-04358-3
Golinelli, Boetto, Carullo, Nuzzolese, Landini et al., Adoption of digital technologies in health care during the COVID-19 pandemic: systematic review of early scientific literature, J Med Internet Res, doi:10.2196/22280
Greenough, Sajjadi, Rucker, Vassar, Hartwell, The use of telecommunication and virtualization among ongoing and discontinued COVID-19 clinical trials: a cross-sectional analysis, Contemp Clin Trials, doi:10.1016/j.cct.2022.106681
Griessbach, Schönenberger, Heravi, Gloy, Agarwal et al., Characteristics, progression, and output of randomized platform trials: a systematic review, JAMA Netw Open, doi:10.1001/jamanetworkopen.2024.3109
Hartman, Patel, Rao, Hagen, Saveraid et al., A comparison of recruitment methods for a remote, nationwide clinical trial for COVID-19 treatment, Open Forum Infect Dis, doi:10.1093/ofid/ofae224
Hosseini, Condon, Brda, Daley, Greiver et al., Canadian adaptive platform trial of treatments for COVID in community settings (CanTreatCOVID): protocol for a randomised controlled adaptive platform trial of treatments for acute SARS-CoV-2 infection in community settings, BMJ Open, doi:10.1136/bmjopen-2024-097134
Hu, Tardif, Huber, Daly, Langford et al., Chasing the storm: recruiting non-hospitalized patients for a multi-site randomized controlled trial in the United States during the COVID-19 pandemic, Clin Transl Sci, doi:10.1111/cts.13211
Hulstaert, Twick, Sarsour, Verstraete, Enhancing site selection strategies in clinical trial recruitment using real-world data modeling, PLoS One, doi:10.1371/journal.pone.0300109
Khalil, Marcucci, Liu, Leveraging new methodologies for public health crisis management, Front Public Health, doi:10.3389/fpubh.2024.1508417
Kumkale, Albarracín, Seignourel, The effects of source credibility in the presence or absence of prior attitudes: implications for the design of persuasive communication campaigns, J Appl Soc Psychol, doi:10.1111/j.1559-1816.2010.00620.x
Morgan, Harrison, Wright, Jia, Deal et al., The role of perceived expertise and trustworthiness in research study and clinical trial recruitment: perspectives of clinical research coordinators and African American and black Caribbean patients, PLoS One
Moseson, Kumar, Juusola, Comparison of study samples recruited with virtual versus traditional recruitment methods, Contemp Clin Trials Commun, doi:10.1016/j.conctc.2020.100590
Occa, Merritt, Leip, Stapleton, What influences trust in and understanding of clinical trials? An analysis of information and communication technology use and online health behavior from the health information National Trends Survey, Clin Trials, doi:10.1177/17407745231204813
Tomiwa, Wong, Miller, Ogungbe, Byiringiro et al., Leveraging digital tools to enhance diversity and inclusion in clinical trial recruitment, Front Public Health
Treweek, Briel, Digital tools for trial recruitment and retention-plenty of tools but rigorous evaluation is in short supply, Trials, doi:10.1186/s13063-020-04361-8
Vanderbeek, Bliss, Yin, Yap, Implementation of platform trials in the COVID-19 pandemic: a rapid review, Contemp Clin Trials, doi:10.1016/j.cct.2021.106625
DOI record:
{
"DOI": "10.3389/fpubh.2026.1698604",
"ISSN": [
"2296-2565"
],
"URL": "http://dx.doi.org/10.3389/fpubh.2026.1698604",
"abstract": "<jats:sec>\n <jats:title>Background</jats:title>\n <jats:p>Recruitment remains a challenge in clinical trials. This study describes the use of digital and traditional recruitment channels in a national, community-based adaptive platform trial for COVID-19 in Canada.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Methods</jats:title>\n <jats:p>Self-reported recruitment sources were collected from participants of a remote, national adaptive platform trial of COVID-19 treatments conducted across six Canadian provinces using a secure web-based application. Recruitment channels were analyzed using descriptive statistics, chi-square tests, and logistic regression to explore associations with province.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>\n From January 2023 to September 2024, 1,515 participants completed the pre-screening process, and 720 were randomized. Of them, 416 were recruited through traditional channels, 303 through digital channels, and one through an undetermined source. Recruitment channels varied by province (\n <jats:italic>χ</jats:italic>\n <jats:sup>2</jats:sup>\n (5) = 81.30,\n <jats:italic>p</jats:italic>\n &lt; 0.001; Cramér’s\n <jats:italic>V</jats:italic>\n = 0.34), and digital channels were associated with higher odds of recruitment compared with traditional channels (adjusted OR = 2.78, 95% CI 2.20–3.52). The most frequently reported referral source was ‘family, friends, or colleagues’ (20%), followed by provincial government websites and health lines (16.25%), online channels (10.56%), and public communications (9.86%).\n </jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusion</jats:title>\n <jats:p>Combining traditional and digital recruitment methods supports recruitment. Trials can raise awareness of the study by leveraging both digital and traditional channels and can move information to action through trusted social groups and institutions. Further research is needed to evaluate the cost-effectiveness, reach, and demographic differences across platforms, informing recruitment strategies in future decentralized and adaptive trials beyond COVID-19.</jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Clinical trial registration</jats:title>\n <jats:p>\n <jats:ext-link>https://clinicaltrials.gov/study/NCT05614349</jats:ext-link>\n , Identifier NCT05614349.\n </jats:p>\n </jats:sec>",
"alternative-id": [
"10.3389/fpubh.2026.1698604"
],
"article-number": "1698604",
"author": [
{
"affiliation": [
{
"name": "Upstream Lab, Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Unity Health Toronto",
"place": [
"Toronto, ON, Canada"
]
}
],
"family": "Astorga",
"given": "Geil Han",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Upstream Lab, Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Unity Health Toronto",
"place": [
"Toronto, ON, Canada"
]
},
{
"name": "Department of Family and Community Medicine, St. Michael’s Hospital, Unity Health Toronto",
"place": [
"Toronto, ON, Canada"
]
},
{
"name": "Department of Family and Community Medicine, Faculty of Medicine, University of Toronto",
"place": [
"Toronto, ON, Canada"
]
},
{
"name": "Division of Clinical Public Health, Dalla Lana School of Public Health, University of Toronto",
"place": [
"Toronto, ON, Canada"
]
},
{
"name": "Institute of Health Policy, Management, and Evaluation, Dalla Lana School of Public Health, University of Toronto",
"place": [
"Toronto, ON, Canada"
]
}
],
"family": "Pinto",
"given": "Andrew D.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Upstream Lab, Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Unity Health Toronto",
"place": [
"Toronto, ON, Canada"
]
}
],
"family": "Sivayoganathan",
"given": "Kawsika",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Upstream Lab, Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Unity Health Toronto",
"place": [
"Toronto, ON, Canada"
]
}
],
"family": "Maruthananth",
"given": "Kevin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Upstream Lab, Li Ka Shing Knowledge Institute, St. Michael’s Hospital, Unity Health Toronto",
"place": [
"Toronto, ON, Canada"
]
},
{
"name": "Department of Family and Community Medicine, Faculty of Medicine, University of Toronto",
"place": [
"Toronto, ON, Canada"
]
},
{
"name": "Division of Clinical Public Health, Dalla Lana School of Public Health, University of Toronto",
"place": [
"Toronto, ON, Canada"
]
},
{
"name": "Institute of Medical Science, Faculty of Medicine, University of Toronto",
"place": [
"Toronto, ON, Canada"
]
}
],
"family": "Hosseini",
"given": "Banafshe",
"sequence": "additional"
}
],
"container-title": "Frontiers in Public Health",
"container-title-short": "Front. Public Health",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2026,
2,
11
]
],
"date-time": "2026-02-11T06:50:52Z",
"timestamp": 1770792652000
},
"deposited": {
"date-parts": [
[
2026,
2,
24
]
],
"date-time": "2026-02-24T10:22:45Z",
"timestamp": 1771928565000
},
"indexed": {
"date-parts": [
[
2026,
2,
24
]
],
"date-time": "2026-02-24T11:11:00Z",
"timestamp": 1771931460717,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
2,
11
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
11
]
],
"date-time": "2026-02-11T00:00:00Z",
"timestamp": 1770768000000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fpubh.2026.1698604/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2026,
2,
11
]
]
},
"published-online": {
"date-parts": [
[
2026,
2,
11
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"DOI": "10.1016/j.cct.2023.107292",
"article-title": "Platform trials to evaluate the benefit-risk of COVID-19 therapeutics: successes, learnings, and recommendations for future pandemics",
"author": "Buenconsejo",
"doi-asserted-by": "publisher",
"first-page": "107292",
"journal-title": "Contemp Clin Trials",
"key": "ref1",
"volume": "132",
"year": "2023"
},
{
"DOI": "10.1016/j.cct.2021.106625",
"article-title": "Implementation of platform trials in the COVID-19 pandemic: a rapid review",
"author": "Vanderbeek",
"doi-asserted-by": "publisher",
"first-page": "106625",
"journal-title": "Contemp Clin Trials",
"key": "ref2",
"volume": "112",
"year": "2022"
},
{
"DOI": "10.1186/s13063-021-05818-0",
"article-title": "Exploring reasons for recruitment failure in clinical trials: a qualitative study with clinical trial stakeholders in Switzerland, Germany, and Canada",
"author": "Briel",
"doi-asserted-by": "publisher",
"first-page": "844",
"journal-title": "Trials",
"key": "ref3",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.1001/jamanetworkopen.2024.3109",
"article-title": "Characteristics, progression, and output of randomized platform trials: a systematic review",
"author": "Griessbach",
"doi-asserted-by": "publisher",
"first-page": "e243109",
"journal-title": "JAMA Netw Open",
"key": "ref4",
"volume": "7",
"year": "2024"
},
{
"DOI": "10.1371/journal.pone.0300109",
"article-title": "Enhancing site selection strategies in clinical trial recruitment using real-world data modeling",
"author": "Hulstaert",
"doi-asserted-by": "publisher",
"first-page": "e0300109",
"journal-title": "PLoS One",
"key": "ref5",
"volume": "19",
"year": "2024"
},
{
"DOI": "10.12688/hrbopenres.13650.1",
"article-title": "What works to recruit general practices to trials? A rapid review",
"author": "Buckley",
"doi-asserted-by": "publisher",
"first-page": "13",
"journal-title": "HRB Open Res",
"key": "ref6",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.1093/ofid/ofae224",
"article-title": "A comparison of recruitment methods for a remote, nationwide clinical trial for COVID-19 treatment",
"author": "Hartman",
"doi-asserted-by": "publisher",
"first-page": "ofae224",
"journal-title": "Open Forum Infect Dis",
"key": "ref7",
"volume": "11",
"year": "2024"
},
{
"DOI": "10.1136/bmjopen-2017-016216",
"article-title": "Discontinuation and non-publication of randomised clinical trials supported by the main public funding body in Switzerland: a retrospective cohort study",
"author": "Amstutz",
"doi-asserted-by": "publisher",
"first-page": "e016216",
"journal-title": "BMJ Open",
"key": "ref8",
"volume": "7",
"year": "2017"
},
{
"DOI": "10.3389/fpubh.2024.1508417",
"article-title": "Leveraging new methodologies for public health crisis management",
"author": "Khalil",
"doi-asserted-by": "publisher",
"first-page": "1508417",
"journal-title": "Front Public Health",
"key": "ref9",
"volume": "12",
"year": "2024"
},
{
"DOI": "10.1186/s13063-020-04361-8",
"article-title": "Digital tools for trial recruitment and retention—plenty of tools but rigorous evaluation is in short supply",
"author": "Treweek",
"doi-asserted-by": "publisher",
"first-page": "476",
"journal-title": "Trials",
"key": "ref10",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.1177/0890117116671673",
"article-title": "A randomized controlled trial of opt-in versus opt-out enrollment into a diabetes behavioral intervention",
"author": "Aysola",
"doi-asserted-by": "publisher",
"first-page": "745",
"journal-title": "Am J Health Promot: AJHP",
"key": "ref11",
"volume": "32",
"year": "2018"
},
{
"DOI": "10.1016/j.ypmed.2018.06.004",
"article-title": "Stepwise strategy to improve cervical cancer screening adherence (SCAN-cervical Cancer) - automated text messages, phone calls and reminders: population based randomized controlled trial",
"author": "Firmino-Machado",
"doi-asserted-by": "publisher",
"first-page": "123",
"journal-title": "Prev Med",
"key": "ref12",
"volume": "114",
"year": "2018"
},
{
"DOI": "10.1056/nejmoa2016638",
"article-title": "A randomized trial of hydroxychloroquine as postexposure prophylaxis for COVID-19",
"author": "Boulware",
"doi-asserted-by": "publisher",
"first-page": "517",
"journal-title": "N Engl J Med",
"key": "ref13",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1186/s13063-020-04358-3",
"article-title": "Digital tools for the recruitment and retention of participants in randomised controlled trials: a systematic map",
"author": "Frampton",
"doi-asserted-by": "publisher",
"first-page": "478",
"journal-title": "Trials",
"key": "ref14",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.2196/22280",
"article-title": "Adoption of digital technologies in health care during the COVID-19 pandemic: systematic review of early scientific literature",
"author": "Golinelli",
"doi-asserted-by": "publisher",
"first-page": "e22280",
"journal-title": "J Med Internet Res",
"key": "ref15",
"volume": "22",
"year": "2020"
},
{
"DOI": "10.1016/j.cct.2022.106681",
"article-title": "The use of telecommunication and virtualization among ongoing and discontinued COVID-19 clinical trials: a cross-sectional analysis",
"author": "Greenough",
"doi-asserted-by": "publisher",
"first-page": "106681",
"journal-title": "Contemp Clin Trials",
"key": "ref16",
"volume": "114",
"year": "2022"
},
{
"DOI": "10.1016/j.conctc.2020.100590",
"article-title": "Comparison of study samples recruited with virtual versus traditional recruitment methods",
"author": "Moseson",
"doi-asserted-by": "publisher",
"first-page": "100590",
"journal-title": "Contemp Clin Trials Commun",
"key": "ref17",
"volume": "19",
"year": "2020"
},
{
"DOI": "10.2196/60504",
"article-title": "Improving participant recruitment in clinical trials: comparative analysis of innovative digital platforms",
"author": "Bikou",
"doi-asserted-by": "publisher",
"first-page": "e60504",
"journal-title": "J Med Internet Res",
"key": "ref18",
"volume": "26",
"year": "2024"
},
{
"DOI": "10.1136/bmjopen-2024-097134",
"article-title": "Canadian adaptive platform trial of treatments for COVID in community settings (CanTreatCOVID): protocol for a randomised controlled adaptive platform trial of treatments for acute SARS-CoV-2 infection in community settings",
"author": "Hosseini",
"doi-asserted-by": "publisher",
"first-page": "e097134",
"journal-title": "BMJ Open",
"key": "ref19",
"volume": "15",
"year": "2025"
},
{
"DOI": "10.1016/j.npls.2016.01.001",
"article-title": "How to plan and perform a qualitative study using content analysis",
"author": "Bengtsson",
"doi-asserted-by": "publisher",
"first-page": "8",
"journal-title": "Nurs Open",
"key": "ref20",
"volume": "2",
"year": "2016"
},
{
"DOI": "10.1371/journal.pone.0275770",
"article-title": "The role of perceived expertise and trustworthiness in research study and clinical trial recruitment: perspectives of clinical research coordinators and African American and black Caribbean patients",
"author": "Morgan",
"doi-asserted-by": "crossref",
"first-page": "e0275770",
"journal-title": "PLoS One",
"key": "ref21",
"volume": "18",
"year": "2023"
},
{
"DOI": "10.1186/s12874-022-01724-4",
"article-title": "Development and pilot implementation of guidelines for culturally tailored research recruitment materials for African Americans and Latinos",
"author": "Cunningham-Erves",
"doi-asserted-by": "publisher",
"first-page": "248",
"journal-title": "BMC Med Res Methodol",
"key": "ref22",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.3389/fpubh.2024.1483367",
"article-title": "Leveraging digital tools to enhance diversity and inclusion in clinical trial recruitment",
"author": "Tomiwa",
"doi-asserted-by": "crossref",
"first-page": "1483367",
"journal-title": "Front Public Health",
"key": "ref23",
"volume": "12",
"year": "2024"
},
{
"DOI": "10.1111/cts.13211",
"article-title": "Chasing the storm: recruiting non-hospitalized patients for a multi-site randomized controlled trial in the United States during the COVID-19 pandemic",
"author": "Hu",
"doi-asserted-by": "publisher",
"first-page": "831",
"journal-title": "Clin Transl Sci",
"key": "ref24",
"volume": "15",
"year": "2022"
},
{
"DOI": "10.1177/17407745231204813",
"article-title": "What influences trust in and understanding of clinical trials? An analysis of information and communication technology use and online health behavior from the health information National Trends Survey",
"author": "Occa",
"doi-asserted-by": "publisher",
"first-page": "95",
"journal-title": "Clin Trials",
"key": "ref25",
"volume": "21",
"year": "2024"
},
{
"DOI": "10.1111/j.1559-1816.2010.00620.x",
"article-title": "The effects of source credibility in the presence or absence of prior attitudes: implications for the design of persuasive communication campaigns",
"author": "Kumkale",
"doi-asserted-by": "publisher",
"first-page": "1325",
"journal-title": "J Appl Soc Psychol",
"key": "ref26",
"volume": "40",
"year": "2010"
}
],
"reference-count": 26,
"references-count": 26,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fpubh.2026.1698604/full"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Canadian adaptive platform trial of treatments for COVID in community settings (CanTreatCOVID): recruitment strategies of a decentralized, national randomized controlled trial for acute SARS-CoV-2",
"type": "journal-article",
"update-policy": "https://doi.org/10.3389/crossmark-policy",
"volume": "14"
}