The Relationship Between Vitamin D Status and the Clinical Severity of COVID-19 Infection: A Retrospective Single-Center Analysis
et al., Cureus, doi:10.7759/cureus.22385, Feb 2022
Vitamin D for COVID-19
8th treatment shown to reduce risk in
October 2020, now with p < 0.00000000001 from 137 studies, recognized in 18 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
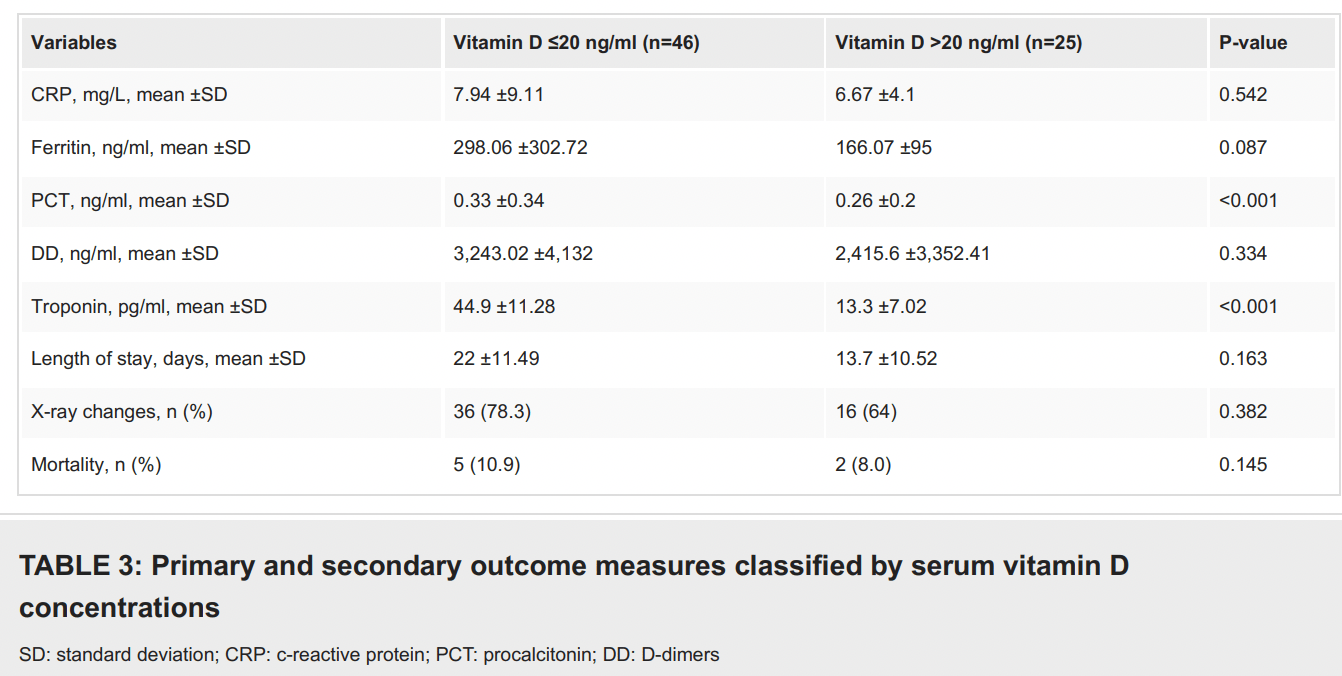
Retrospective 71 hospitalized COVID-19 patients in Greece with vitamin D levels measured within 48 hours of admission, showing longer hospitalization, more radiographic findings, and higher inflammatory and cellular damage markers with vitamin D deficiency. Differences were only statistically significant for troponin and PCT.
This is the 121st of 232 COVID-19 sufficiency studies for vitamin D, which collectively show higher levels reduce risk with p<0.0000000001.
|
risk of death, 26.4% lower, RR 0.74, p = 1.00, high D levels (≥20ng/ml) 2 of 25 (8.0%), low D levels (<20ng/ml) 5 of 46 (10.9%), NNT 35.
|
|
radiographic changes, 18.2% lower, RR 0.82, p = 0.26, high D levels (≥20ng/ml) 16 of 25 (64.0%), low D levels (<20ng/ml) 36 of 46 (78.3%), NNT 7.0.
|
|
hospitalization time, 37.7% lower, relative time 0.62, p = 0.16, high D levels (≥20ng/ml) 25, low D levels (<20ng/ml) 46.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Zidrou et al., 19 Feb 2022, retrospective, Greece, peer-reviewed, 6 authors, study period August 2020 - October 2020.
The Relationship Between Vitamin D Status and the Clinical Severity of COVID-19 Infection: A Retrospective Single-Center Analysis
Cureus, doi:10.7759/cureus.22385
Background and objective Some studies have suggested a potential protective role of vitamin D in coronavirus disease 2019 patients, and this has led to a debate on the topic in the medical community. However, the reported data on the number of hospitalized patients who were vitamin D-deficient is not convincing. In light of this, the aim of the present study was to explore if vitamin D deficiency is correlated with severity and mortality rates of COVID-19 infection in hospitalized COVID-19 patients at a tertiary care hospital in Greece.
Methods We conducted a single-center retrospective study involving 71 patients hospitalized with COVID-19 from August to October 2020. Serum 25-hydroxyvitamin D (25(OH)D) level was assessed in all patients within 48 hours of hospital admission. Serum 25(OH)D level ≤20 ng/ml was defined as a deficiency, while that >20 ng/ml as repletion. The primary outcomes of the infection were classified as partial/complete recovery and mortality during hospitalization. The secondary outcomes were blood markers of inflammation and thrombosis.
Results Among the 71 COVID-19-positive patients [mean age: 63 years, range: 20-97; male (n=47; 66.2%): female (n=24; 33.8%)] who were enrolled in the study, 46 (64.8%) patients had 25(OH)D levels ≤20 ng/ml and 25 (35.2%) had a level >20 ng/ml. According to the patients' medical history, 55 patients (77.5%) had comorbidities. It appears that vitamin D deficiency (<20 ng/ml) significantly correlated with elevated biochemical markers such as procalcitonin and troponin (p<0.001). Moreover, male gender, advanced age (>60 years), and comorbidities were positively associated with more severe COVID-19 infection (elevated inflammation markers, radiographic findings on X-rays, and increased length of hospital stay).
Conclusion These preliminary findings show that vitamin D status among the patients was not related to the severity of COVID-19 infection.
Additional Information Disclosures Human subjects: Consent was obtained or waived by all participants in this study. Hospital Ethics Committee, General Hospital of Thessaloniki "Papageorgiou" issued approval 345/April 2021. This study has been approved by the Hospital Ethics Committee at the General Hospital of Thessaloniki "Papageorgiou". Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue.
Conflicts of interest: In compliance with the ICMJE uniform disclosure form, all authors declare the following: Payment/services info: All authors have declared that no financial support was received from any organization for the submitted work. Financial relationships: All authors have declared that they have no financial relationships at present or within the previous three years with any organizations that might have an interest in the submitted work. Other relationships: All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
References
Abrishami, Dalili, Torbati, Asgari, Arab-Ahmadi et al., Possible association of vitamin D status with lung involvement and outcome in patients with COVID-19: a retrospective study, Eur J Nutr, doi:10.1007/s00394-020-02411-0?utm_medium=email&utm_source=transaction
Arboleda, Urcuqui-Inchima, Vitamin D supplementation: a potential approach for coronavirus/COVID-19 therapeutics?, Front Immunol, doi:10.3389/fimmu.2020.01523?utm_medium=email&utm_source=transaction
Bajgain, Badal, Bajgain, Santana, Prevalence of comorbidities among individuals with COVID-19: a rapid review of current literature, Am J Infect Control, doi:10.1016/j.ajic.2020.06.213?utm_medium=email&utm_source=transaction
Baktash, Hosack, Patel, Vitamin D status and outcomes for hospitalised older patients with COVID-19, Postgrad Med J, doi:10.1136/postgradmedj-2020-138712?utm_medium=email&utm_source=transaction
Burton, Fort, Seoane, Hospitalization and mortality among black patients and white patients with Covid-19, N Engl J Med, doi:10.1056/NEJMsa2011686?utm_medium=email&utm_source=transaction
Campi, Gennari, Merlotti, Vitamin D and COVID-19 severity and related mortality: a prospective study in Italy, BMC Infect Dis, doi:10.1186/s12879-021-06281-7?utm_medium=email&utm_source=transaction
Chen, Zhou, Dong, Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study, Lancet, doi:10.1016/S0140-6736(20)30211-7?utm_medium=email&utm_source=transaction
Daneshkhah, Agrawal, Eshein, Subramanian, Roy et al., The possible role of vitamin D in suppressing cytokine storm and associated mortality in COVID-19 patients, PREPRINT, doi:10.1101/2020.04.08.20058578?utm_medium=email&utm_source=transaction
Garcia, Fumeaux, Guerci, Prognostic factors associated with mortality risk and disease progression in 639 critically ill patients with COVID-19 in Europe: initial report of the international RISC-19-ICU prospective observational cohort, EClinicalMedicine, doi:10.1016/j.eclinm.2020.100449?utm_medium=email&utm_source=transaction
Gebhard, Regitz-Zagrosek, Neuhauser, Morgan, Klein, Impact of sex and gender on COVID-19 outcomes in Europe, Biol Sex Differ, doi:10.1186/s13293-020-00304-9?utm_medium=email&utm_source=transaction
Gönen, Alaylıoğlu, Durcan, Rapid and effective vitamin D supplementation may present better clinical outcomes in COVID-19 (SARS-CoV-2) patients by altering serum INOS1, IL1B, IFNg, cathelicidin-LL37, and ICAM1, Nutrients, doi:10.3390/nu13114047?utm_medium=email&utm_source=transaction
Holick, Vitamin D status: measurement, interpretation, and clinical application, Ann Epidemiol, doi:10.1016/j.annepidem.2007.12.001?utm_medium=email&utm_source=transaction
Isaia, Medico, Associations between hypovitaminosis D and COVID-19: a narrative review, Aging Clin Exp Res, doi:10.1007/s40520-020-01650-9?utm_medium=email&utm_source=transaction
Jakovac, COVID-19 and vitamin D-is there a link and an opportunity for intervention?, Am J Physiol Endocrinol Metab, doi:10.1152/ajpendo.00138.2020?utm_medium=email&utm_source=transaction
Jassil, Sharma, Bikle, Wang, Vitamin D binding protein and 25-hydroxyvitamin D levels: emerging clinical applications, Endocr Pract, doi:10.4158/EP161604.RA?utm_medium=email&utm_source=transaction
Jenei, Jenei, Tamás, COVID-19 mortality is associated with low vitamin D levels in patients with risk factors and/or advanced age, Clin Nutr ESPEN, doi:10.1016/j.clnesp.2021.11.025?utm_medium=email&utm_source=transaction
Jevalikar, Mithal, Singh, Lack of association of baseline 25-hydroxyvitamin D levels with disease severity and mortality in Indian patients hospitalized for COVID-19, Sci Rep, doi:10.1038/s41598-021-85809-y?utm_medium=email&utm_source=transaction
Lagadinou, Zorbas, Velissaris, Vitamin D plasma levels in patients with COVID-19: a case series, Infez Med
Malacova, Cheang, Dunlop, Prevalence and predictors of vitamin D deficiency in a nationally representative sample of adults participating in the 2011-2013 Australian Health Survey, Br J Nutr, doi:10.1017/S0007114519000151?utm_medium=email&utm_source=transaction
Martineau, Forouhi, Vitamin D for COVID-19: a case to answer?, Lancet Diabetes Endocrinol, doi:10.1016/S2213-8587(20)30268-0?utm_medium=email&utm_source=transaction
Martineau, Jolliffe, Hooper, of 7 infections: systematic review and meta-analysis of individual participant data, Cureus, doi:10.1136/bmj.i6583?utm_medium=email&utm_source=transaction
Meltzer, Best, Zhang, Vokes, Arora et al., Association of vitamin d deficiency and treatment with COVID-19 incidence, PREPRINT, doi:10.1101/2020.05.08.20095893?utm_medium=email&utm_source=transaction
Pizzini, Aichner, Sahanic, Impact of vitamin D deficiency on COVID-19-a prospective analysis from the CovILD Registry, Nutrients, doi:10.3390/nu12092775.25?utm_medium=email&utm_source=transaction
Raharusuna, Priambada, Budiarti, Agung, Budi, Patterns of COVID-19 mortality and vitamin D: an Indonesian study
Readcube, None
Richardson, Hirsch, Narasimhan, Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area, JAMA, doi:10.1001/jama.2020.6775?utm_medium=email&utm_source=transaction
Shi, Yu, Zhao, Wang, Zhao et al., Host susceptibility to severe COVID-19 and establishment of a host risk score: findings of 487 cases outside Wuhan, Crit Care, doi:10.1186/s13054-020-2833-7?utm_medium=email&utm_source=transaction
Vasheghani, Jannati, Baghaei, Rezaei, Aliyari et al., The relationship between serum 25-hydroxyvitamin D levels and the severity of COVID-19 disease and its mortality, Sci Rep, doi:10.1038/s41598-021-97017-9?utm_medium=email&utm_source=transaction
Yang, Zheng, Gou, Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis, Int J Infect Dis, doi:10.1016/j.ijid.2020.03.017?utm_medium=email&utm_source=transaction
Zheng, Peng, Xu, Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis, J Infect, doi:10.1016/j.jinf.2020.04.021?utm_medium=email&utm_source=transaction
DOI record:
{
"DOI": "10.7759/cureus.22385",
"ISSN": [
"2168-8184"
],
"URL": "http://dx.doi.org/10.7759/cureus.22385",
"author": [
{
"affiliation": [],
"family": "Zidrou",
"given": "Christiana",
"sequence": "first"
},
{
"affiliation": [],
"family": "Vasiliadis",
"given": "Angelo V",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tsatlidou",
"given": "Maria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Sentona",
"given": "Maria",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vogiatzis",
"given": "Stavros",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Beletsiotis",
"given": "Anastasios",
"sequence": "additional"
}
],
"container-title": [
"Cureus"
],
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2022,
2,
19
]
],
"date-time": "2022-02-19T14:47:31Z",
"timestamp": 1645282051000
},
"deposited": {
"date-parts": [
[
2022,
2,
19
]
],
"date-time": "2022-02-19T14:47:34Z",
"timestamp": 1645282054000
},
"indexed": {
"date-parts": [
[
2022,
2,
19
]
],
"date-time": "2022-02-19T15:10:20Z",
"timestamp": 1645283420851
},
"is-referenced-by-count": 0,
"issn-type": [
{
"type": "print",
"value": "2168-8184"
}
],
"issued": {
"date-parts": [
[
2022,
2,
19
]
]
},
"language": "en",
"link": [
{
"URL": "https://www.cureus.com/articles/85849-the-relationship-between-vitamin-d-status-and-the-clinical-severity-of-covid-19-infection-a-retrospective-single-center-analysis",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "4492",
"original-title": [],
"prefix": "10.7759",
"published": {
"date-parts": [
[
2022,
2,
19
]
]
},
"published-print": {
"date-parts": [
[
2022,
2,
19
]
]
},
"publisher": "Cureus, Inc.",
"reference": [
{
"DOI": "10.1016/S0140-6736(20)30211-7",
"article-title": "Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study",
"author": "Chen N",
"doi-asserted-by": "publisher",
"journal-title": "Lancet",
"key": "ref1",
"unstructured": "Chen N, Zhou M, Dong X, et al.. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020, 395:507-13. 10.1016/S0140-6736(20)30211-7",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/j.ijid.2020.03.017",
"article-title": "Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis",
"author": "Yang J",
"doi-asserted-by": "publisher",
"journal-title": "Int J Infect Dis",
"key": "ref2",
"unstructured": "Yang J, Zheng Y, Gou X, et al.. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis. Int J Infect Dis. 2020, 94:91-5. 10.1016/j.ijid.2020.03.017",
"volume": "94",
"year": "2020"
},
{
"DOI": "10.1186/s13054-020-2833-7",
"article-title": "Host susceptibility to severe COVID-19 and establishment of a host risk score: findings of 487 cases outside Wuhan",
"author": "Shi Y",
"doi-asserted-by": "publisher",
"journal-title": "Crit Care",
"key": "ref3",
"unstructured": "Shi Y, Yu X, Zhao H, Wang H, Zhao R, Sheng J. Host susceptibility to severe COVID-19 and establishment of a host risk score: findings of 487 cases outside Wuhan. Crit Care. 2020, 24:108. 10.1186/s13054-020-2833-7",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.1101/2020.05.08.20095893",
"article-title": "Association of vitamin d deficiency and treatment with COVID-19 incidence [PREPRINT]",
"author": "Meltzer DO",
"doi-asserted-by": "publisher",
"journal-title": "medRxiv",
"key": "ref4",
"unstructured": "Meltzer DO, Best TJ, Zhang H, Vokes T, Arora V, Solway J. Association of vitamin d deficiency and treatment with COVID-19 incidence [PREPRINT]. medRxiv. 2020, 10.1101/2020.05.08.20095893",
"year": "2020"
},
{
"DOI": "10.3390/nu12092775.25",
"article-title": "Impact of vitamin D deficiency on COVID-19—a prospective analysis from the CovILD Registry",
"author": "Pizzini A",
"doi-asserted-by": "publisher",
"journal-title": "Nutrients",
"key": "ref5",
"unstructured": "Pizzini A, Aichner M, Sahanic S, et al.. Impact of vitamin D deficiency on COVID-19—a prospective analysis from the CovILD Registry. Nutrients. 2020, 12:2775. 10.3390/nu12092775.25",
"volume": "12",
"year": "2020"
},
{
"DOI": "10.1136/postgradmedj-2020-138712",
"article-title": "Vitamin D status and outcomes for hospitalised older patients with COVID-19",
"author": "Baktash V",
"doi-asserted-by": "publisher",
"journal-title": "Postgrad Med J",
"key": "ref6",
"unstructured": "Baktash V, Hosack T, Patel N, et al.. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad Med J. 2021, 97:442-7. 10.1136/postgradmedj-2020-138712",
"volume": "97",
"year": "2021"
},
{
"DOI": "10.1007/s00394-020-02411-0",
"article-title": "Possible association of vitamin D status with lung involvement and outcome in patients with COVID-19: a retrospective study",
"author": "Abrishami A",
"doi-asserted-by": "publisher",
"journal-title": "Eur J Nutr",
"key": "ref7",
"unstructured": "Abrishami A, Dalili N, Mohammadi Torbati P, Asgari R, Arab-Ahmadi M, Behnam B, Sanei-Taheri M. Possible association of vitamin D status with lung involvement and outcome in patients with COVID-19: a retrospective study. Eur J Nutr. 2021, 60:2249-57. 10.1007/s00394-020-02411-0",
"volume": "60",
"year": "2021"
},
{
"DOI": "10.4158/EP161604.RA",
"article-title": "Vitamin D binding protein and 25-hydroxyvitamin D levels: emerging clinical applications",
"author": "Jassil NK",
"doi-asserted-by": "publisher",
"journal-title": "Endocr Pract",
"key": "ref8",
"unstructured": "Jassil NK, Sharma A, Bikle D, Wang X. Vitamin D binding protein and 25-hydroxyvitamin D levels: emerging clinical applications. Endocr Pract. 2017, 23:605-13. 10.4158/EP161604.RA",
"volume": "23",
"year": "2017"
},
{
"DOI": "10.1101/2020.04.08.20058578",
"article-title": "The possible role of vitamin D in suppressing cytokine storm and associated mortality in COVID-19 patients [PREPRINT]",
"author": "Daneshkhah A",
"doi-asserted-by": "publisher",
"journal-title": "medRxiv",
"key": "ref9",
"unstructured": "Daneshkhah A, Agrawal V, Eshein A, Subramanian H, Roy HK, Backman V. The possible role of vitamin D in suppressing cytokine storm and associated mortality in COVID-19 patients [PREPRINT]. medRxiv. 2020, 10.1101/2020.04.08.20058578",
"year": "2020"
},
{
"DOI": "10.1136/bmj. i6583",
"article-title": "Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data",
"author": "Martineau AR",
"doi-asserted-by": "publisher",
"journal-title": "BMJ",
"key": "ref10",
"unstructured": "Martineau AR, Jolliffe DA, Hooper RL, et al.. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ. 2017, 356:i6583. 10.1136/bmj. i6583",
"volume": "356",
"year": "2017"
},
{
"DOI": "10.1007/s40520-020-01650-9",
"article-title": "Associations between hypovitaminosis D and COVID-19: a narrative review",
"author": "Isaia G",
"doi-asserted-by": "publisher",
"journal-title": "Aging Clin Exp Res",
"key": "ref11",
"unstructured": "Isaia G, Medico E. Associations between hypovitaminosis D and COVID-19: a narrative review. Aging Clin Exp Res. 2020, 32:1879-81. 10.1007/s40520-020-01650-9",
"volume": "32",
"year": "2020"
},
{
"DOI": "10.1152/ajpendo.00138.2020",
"article-title": "COVID-19 and vitamin D-is there a link and an opportunity for intervention?",
"author": "Jakovac H",
"doi-asserted-by": "publisher",
"journal-title": "Am J Physiol Endocrinol Metab",
"key": "ref12",
"unstructured": "Jakovac H. COVID-19 and vitamin D-is there a link and an opportunity for intervention?. Am J Physiol Endocrinol Metab. 2020, 318:E589. 10.1152/ajpendo.00138.2020",
"volume": "318",
"year": "2020"
},
{
"DOI": "10.3389/fimmu.2020.01523",
"article-title": "Vitamin D supplementation: a potential approach for coronavirus/COVID-19 therapeutics?",
"author": "Arboleda JF",
"doi-asserted-by": "publisher",
"journal-title": "Front Immunol",
"key": "ref13",
"unstructured": "Arboleda JF, Urcuqui-Inchima S. Vitamin D supplementation: a potential approach for coronavirus/COVID-19 therapeutics?. Front Immunol. 2020, 11:1523. 10.3389/fimmu.2020.01523",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/j.annepidem.2007.12.001",
"article-title": "Vitamin D status: measurement, interpretation, and clinical application",
"author": "Holick MF",
"doi-asserted-by": "publisher",
"journal-title": "Ann Epidemiol",
"key": "ref14",
"unstructured": "Holick MF. Vitamin D status: measurement, interpretation, and clinical application. Ann Epidemiol. 2009, 19:73-8. 10.1016/j.annepidem.2007.12.001",
"volume": "19",
"year": "2009"
},
{
"DOI": "10.1016/j.clnesp.2021.11.025",
"article-title": "COVID-19 mortality is associated with low vitamin D levels in patients with risk factors and/or advanced age",
"author": "Jenei T",
"doi-asserted-by": "publisher",
"journal-title": "Clin Nutr ESPEN",
"key": "ref15",
"unstructured": "Jenei T, Jenei S, Tamás LT, et al.. COVID-19 mortality is associated with low vitamin D levels in patients with risk factors and/or advanced age. Clin Nutr ESPEN. 2022, 47:410-3. 10.1016/j.clnesp.2021.11.025",
"volume": "47",
"year": "2022"
},
{
"DOI": "10.1016/S2213-8587(20)30268-0",
"article-title": "Vitamin D for COVID-19: a case to answer?",
"author": "Martineau AR",
"doi-asserted-by": "publisher",
"journal-title": "Lancet Diabetes Endocrinol",
"key": "ref16",
"unstructured": "Martineau AR, Forouhi NG. Vitamin D for COVID-19: a case to answer?. Lancet Diabetes Endocrinol. 2020, 8:735-6. 10.1016/S2213-8587(20)30268-0",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1038/s41598-021-85809-y",
"article-title": "Lack of association of baseline 25-hydroxyvitamin D levels with disease severity and mortality in Indian patients hospitalized for COVID-19",
"author": "Jevalikar G",
"doi-asserted-by": "publisher",
"journal-title": "Sci Rep",
"key": "ref17",
"unstructured": "Jevalikar G, Mithal A, Singh A, et al.. Lack of association of baseline 25-hydroxyvitamin D levels with disease severity and mortality in Indian patients hospitalized for COVID-19. Sci Rep. 2021, 11:6258. 10.1038/s41598-021-85809-y",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1186/s12879-021-06281-7",
"article-title": "Vitamin D and COVID-19 severity and related mortality: a prospective study in Italy",
"author": "Campi I",
"doi-asserted-by": "publisher",
"journal-title": "BMC Infect Dis",
"key": "ref18",
"unstructured": "Campi I, Gennari L, Merlotti D, et al.. Vitamin D and COVID-19 severity and related mortality: a prospective study in Italy. BMC Infect Dis. 2021, 21:566. 10.1186/s12879-021-06281-7",
"volume": "21",
"year": "2021"
},
{
"DOI": "10.3390/nu13114047",
"article-title": "Rapid and effective vitamin D supplementation may present better clinical outcomes in COVID-19 (SARS-CoV-2) patients by altering serum INOS1, IL1B, IFNg, cathelicidin-LL37, and ICAM1",
"author": "Gönen MS",
"doi-asserted-by": "publisher",
"journal-title": "Nutrients",
"key": "ref19",
"unstructured": "Gönen MS, Alaylıoğlu M, Durcan E, et al.. Rapid and effective vitamin D supplementation may present better clinical outcomes in COVID-19 (SARS-CoV-2) patients by altering serum INOS1, IL1B, IFNg, cathelicidin-LL37, and ICAM1. Nutrients. 2021, 13:4047. 10.3390/nu13114047",
"volume": "13",
"year": "2021"
},
{
"article-title": "Patterns of COVID-19 mortality and vitamin D: an Indonesian study [PREPRINT]",
"author": "Raharusuna P",
"journal-title": "Readcube",
"key": "ref20",
"unstructured": "Raharusuna P, Priambada S, Budiarti C, Agung E, Budi C. Patterns of COVID-19 mortality and vitamin D: an Indonesian study [PREPRINT]. Readcube. 2020,",
"year": "2020"
},
{
"article-title": "Vitamin D plasma levels in patients with COVID-19: a case series",
"author": "Lagadinou M",
"journal-title": "Infez Med",
"key": "ref21",
"unstructured": "Lagadinou M, Zorbas B, Velissaris D. Vitamin D plasma levels in patients with COVID-19: a case series. Infez Med. 2021, 29:224-8.",
"volume": "29",
"year": "2021"
},
{
"article-title": "Dietary Reference Intakes for Calcium and Vitamin D",
"key": "ref22",
"unstructured": "Dietary Reference Intakes for Calcium and Vitamin D. Ross AC, Taylor CL, Yaktine AL, Del Valle HB (ed). National Academies Press, Washington, DC; 2011.",
"year": "2011"
},
{
"DOI": "10.1056/NEJMsa2011686",
"article-title": "Hospitalization and mortality among black patients and white patients with Covid-19",
"author": "Price-Haywood EG",
"doi-asserted-by": "publisher",
"journal-title": "N Engl J Med",
"key": "ref23",
"unstructured": "Price-Haywood EG, Burton J, Fort D, Seoane L. Hospitalization and mortality among black patients and white patients with Covid-19. N Engl J Med. 2020, 382:2534-43. 10.1056/NEJMsa2011686",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/j.jinf.2020.04.021",
"article-title": "Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis",
"author": "Zheng Z",
"doi-asserted-by": "publisher",
"journal-title": "J Infect",
"key": "ref24",
"unstructured": "Zheng Z, Peng F, Xu B, et al.. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J Infect. 2020, 81:e16-25. 10.1016/j.jinf.2020.04.021",
"volume": "81",
"year": "2020"
},
{
"DOI": "10.1038/s41598-021-97017-9",
"article-title": "The relationship between serum 25-hydroxyvitamin D levels and the severity of COVID-19 disease and its mortality",
"author": "Vasheghani M",
"doi-asserted-by": "publisher",
"journal-title": "Sci Rep",
"key": "ref25",
"unstructured": "Vasheghani M, Jannati N, Baghaei P, Rezaei M, Aliyari R, Marjani M. The relationship between serum 25-hydroxyvitamin D levels and the severity of COVID-19 disease and its mortality. Sci Rep. 2021, 11:17594. 10.1038/s41598-021-97017-9",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1186/s13293-020-00304-9",
"article-title": "Impact of sex and gender on COVID-19 outcomes in Europe",
"author": "Gebhard C",
"doi-asserted-by": "publisher",
"journal-title": "Biol Sex Differ",
"key": "ref26",
"unstructured": "Gebhard C, Regitz-Zagrosek V, Neuhauser HK, Morgan R, Klein SL. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex Differ. 2020, 11:29. 10.1186/s13293-020-00304-9",
"volume": "11",
"year": "2020"
},
{
"DOI": "10.1016/j.ajic.2020.06.213",
"article-title": "Prevalence of comorbidities among individuals with COVID-19: a rapid review of current literature",
"author": "Bajgain KT",
"doi-asserted-by": "publisher",
"journal-title": "Am J Infect Control",
"key": "ref27",
"unstructured": "Bajgain KT, Badal S, Bajgain BB, Santana MJ. Prevalence of comorbidities among individuals with COVID-19: a rapid review of current literature. Am J Infect Control. 2021, 49:238-46. 10.1016/j.ajic.2020.06.213",
"volume": "49",
"year": "2021"
},
{
"DOI": "10.1016/j.eclinm.2020.100449",
"article-title": "Prognostic factors associated with mortality risk and disease progression in 639 critically ill patients with COVID-19 in Europe: initial report of the international RISC-19-ICU prospective observational cohort",
"author": "Wendel Garcia PD",
"doi-asserted-by": "publisher",
"journal-title": "EClinicalMedicine",
"key": "ref28",
"unstructured": "Wendel Garcia PD, Fumeaux T, Guerci P, et al.. Prognostic factors associated with mortality risk and disease progression in 639 critically ill patients with COVID-19 in Europe: initial report of the international RISC-19-ICU prospective observational cohort. EClinicalMedicine. 2020, 25:100449. 10.1016/j.eclinm.2020.100449",
"volume": "25",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.6775",
"article-title": "Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area",
"author": "Richardson S",
"doi-asserted-by": "publisher",
"journal-title": "JAMA",
"key": "ref29",
"unstructured": "Richardson S, Hirsch JS, Narasimhan M, et al.. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020, 323:2052-9. 10.1001/jama.2020.6775",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1017/S0007114519000151",
"article-title": "Prevalence and predictors of vitamin D deficiency in a nationally representative sample of adults participating in the 2011-2013 Australian Health Survey",
"author": "Malacova E",
"doi-asserted-by": "publisher",
"journal-title": "Br J Nutr",
"key": "ref30",
"unstructured": "Malacova E, Cheang PR, Dunlop E, et al.. Prevalence and predictors of vitamin D deficiency in a nationally representative sample of adults participating in the 2011-2013 Australian Health Survey. Br J Nutr. 2019, 121:894-904. 10.1017/S0007114519000151",
"volume": "121",
"year": "2019"
}
],
"reference-count": 30,
"references-count": 30,
"relation": {},
"score": 1,
"short-container-title": [],
"short-title": [],
"source": "Crossref",
"subject": [
"Aerospace Engineering"
],
"subtitle": [],
"title": [
"The Relationship Between Vitamin D Status and the Clinical Severity of COVID-19 Infection: A Retrospective Single-Center Analysis"
],
"type": "journal-article"
}
