Exploratory pharmacokinetic-pharmacodynamic characterization and safety of standardized Andrographis paniculata aqueous extract capsules in patients with mild COVID-19
et al., Frontiers in Pharmacology, doi:10.3389/fphar.2026.1781740, TCTR20241123001, Mar 2026
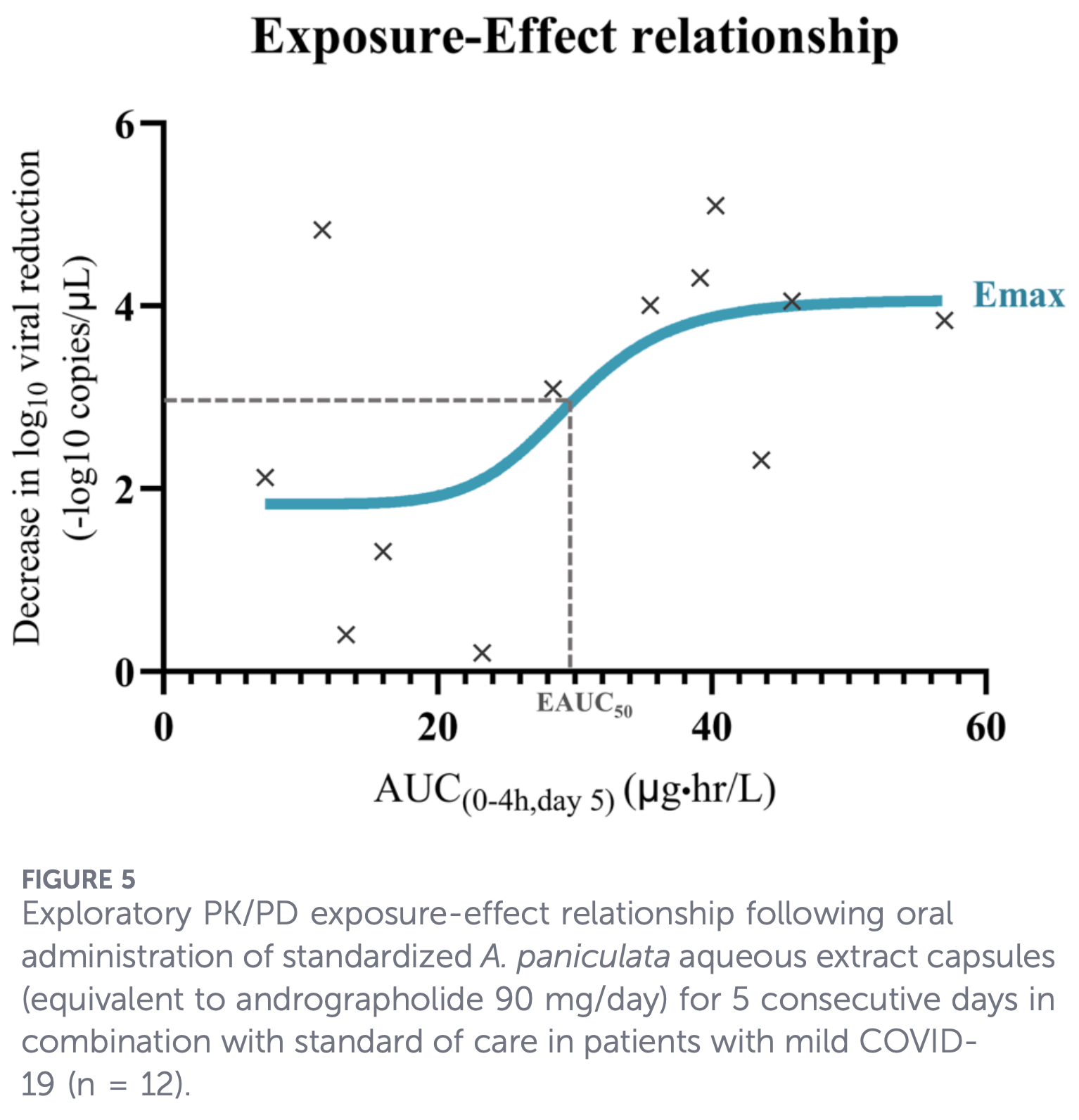
Exploratory open-label PK/PD clinical trial of 12 outpatients with mild COVID-19 showing a -2.96 log10 reduction in viral load over 5 days with standardized Andrographis paniculata aqueous extract capsules (90 mg/day andrographolide). There was no placebo group. The four major diterpenoids (AP1, AP3, AP4, AP6) showed rapid absorption and elimination with limited systemic exposure, and the mean AUC0-4h of andrographolide on day 5 approached the estimated EAUC50 from a sigmoidal Emax PK/PD model (r²=0.4). Adverse events were mild, occurring in 4/12 participants (gastrointestinal symptoms, dizziness, eye discharge), with no hepatotoxicity or renal toxicity observed at this dose level (in contrast to the higher 180 mg/day dose used in a prior healthy volunteer study which caused reversible liver enzyme elevations).
Songvut et al., 13 Mar 2026, retrospective, placebo-controlled, Thailand, peer-reviewed, mean age 34.1, 10 authors, trial TCTR20241123001.
Contact: jutamaad@cri.or.th.
Abstract:
OPEN ACCESS
EDITED BY Jie Liu, Zunyi Medical University, China
REVIEWED BY
Alexander George Panossian, Phytomed AB, Sweden Tam Nguyen, Can Tho University of Medicine and Pharmacy, Vietnam
*CORRESPONDENCE
Jutamaad Satayavivad, jutamaad@cri.or.th
RECEIVED 06 January 2026 REVISED 12 February 2026 ACCEPTED 13 February 2026 PUBLISHED 13 March 2026
CITATION
Songvut P, Payoong P, Okada PA, Rittapai N, Suntararuks S, Akanimanee J, Rangkadilok N, Panomvana D, Puranajoti P and Satayavivad J (2026) Exploratory pharmacokineticpharmacodynamic characterization and safety of standardized Andrographis paniculata aqueous extract capsules in patients with mild COVID-19. Front. Pharmacol. 17:1781740. doi: 10.3389/fphar.2026.1781740
COPYRIGHT
© 2026 Songvut, Payoong, Okada, Rittapai, Suntararuks, Akanimanee, Rangkadilok, Panomvana, Puranajoti and Satayavivad. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
[Exploratory pharmacokineticpharmacodynamic characterization and safety of standardized Andrographis paniculata aqueous extract capsules in patients with mild COVID-19](https://www.frontiersin.org/articles/10.3389/fphar.2026.1781740/full)
Phanit Songvut 1,2 , Paruspak Payoong 3 , Pilailuk Akkapaiboon Okada 4 , Noppawan Rittapai 1 , Sumitra Suntararuks 1 , Jaratluck Akanimanee 1 , Nuchanart Rangkadilok 1,2 , Duangchit Panomvana 5 , Porranee Puranajoti 5 and Jutamaad Satayavivad 1,2 *
1 Laboratory of Pharmacology, Chulabhorn Research Institute, Bangkok, Thailand, 2 Center of Excellence on Environmental Health and Toxicology (EHT), OPS, MHESI, Bangkok, Thailand, 3 Department of Medicine, Chulabhorn Hospital, Chulabhorn Royal Academy, Bangkok, Thailand, 4 National Institute of Health, Department of Medical Sciences, Ministry of Public Health, Nonthaburi, Thailand, 5 Translational Research Unit, Chulabhorn Research Institute, Bangkok, Thailand
Introduction: Andrographis paniculata has been used in the management of COVID-19-related conditions during the SARS-CoV-2 pandemic. However, human pharmacokinetic-pharmacodynamic (PK/PD) evidence to support a rationale for safe and evidence-based dosing of standardized extracts in clinical use remains limited. This exploratory study aimed to investigate the pharmacokinetics, preliminary PK/PD relationship, and short-term safety profile of standardized A. paniculata aqueous extract capsules in patients with mild COVID-19.
Methods: A single-center PK/PD clinical trial with two sequential phases (singledose followed by multiple-dose) was conducted. Patients with mild COVID-19 received standardized A. paniculata aqueous extract capsules equivalent to 30 mg of andrographolide every 8 h (90 mg/day) for 5 consecutive days, alongside standard symptomatic treatment.
Results: Rapid absorption and elimination with limited systemic exposure supported repeated dosing to maintain exposure over the 5-day period. PK/PD analysis demonstrated a sigmoidal, saturable exposure-response relationship. The mean area under the concentration-time curve (AUC0-4 h) on day 5..
DOI record:
{
"DOI": "10.3389/fphar.2026.1781740",
"ISSN": [
"1663-9812"
],
"URL": "http://dx.doi.org/10.3389/fphar.2026.1781740",
"abstract": "<jats:sec>\n <jats:title>Introduction</jats:title>\n <jats:p>\n <jats:italic>Andrographis paniculata</jats:italic>\n has been used in the management of COVID-19-related conditions during the SARS-CoV-2 pandemic. However, human pharmacokinetic-pharmacodynamic (PK/PD) evidence to support a rationale for safe and evidence-based dosing of standardized extracts in clinical use remains limited. This exploratory study aimed to investigate the pharmacokinetics, preliminary PK/PD relationship, and short-term safety profile of standardized\n <jats:italic>A. paniculata</jats:italic>\n aqueous extract capsules in patients with mild COVID-19.\n </jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Methods</jats:title>\n <jats:p>\n A single-center PK/PD clinical trial with two sequential phases (single-dose followed by multiple-dose) was conducted. Patients with mild COVID-19 received standardized\n <jats:italic>A. paniculata</jats:italic>\n aqueous extract capsules equivalent to 30 mg of andrographolide every 8 h (90 mg/day) for 5 consecutive days, alongside standard symptomatic treatment.\n </jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Results</jats:title>\n <jats:p>\n Rapid absorption and elimination with limited systemic exposure supported repeated dosing to maintain exposure over the 5-day period. PK/PD analysis demonstrated a sigmoidal, saturable exposure-response relationship. The mean area under the concentration–time curve (AUC\n <jats:sub>0-4 h</jats:sub>\n ) on day 5 approached the estimated EAUC\n <jats:sub>50</jats:sub>\n (half-maximal effective exposure) derived from the Emax model. A −2.96 log\n <jats:sub>10</jats:sub>\n reduction in viral load was observed; however, without a placebo control group, it is not possible to conclusively attribute this reduction to the investigational\n <jats:italic>A. paniculata</jats:italic>\n aqueous extract capsules. Adverse events were mild, with no hepatotoxicity or renal toxicity observed.\n </jats:p>\n </jats:sec>\n <jats:sec>\n <jats:title>Conclusion</jats:title>\n <jats:p>\n These findings provide preliminary evidence regarding the pharmacokinetics, dose-exposure characteristics, and short-term tolerability of standardized\n <jats:italic>A. paniculata</jats:italic>\n aqueous extract capsules, supporting further investigation in patients with mild COVID-19. Larger, placebo-controlled trials are warranted to establish causality and to evaluate clinical benefits.\n </jats:p>\n </jats:sec>",
"alternative-id": [
"10.3389/fphar.2026.1781740"
],
"article-number": "1781740",
"author": [
{
"affiliation": [
{
"name": "Laboratory of Pharmacology, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
},
{
"name": "Center of Excellence on Environmental Health and Toxicology (EHT), OPS, MHESI",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Songvut",
"given": "Phanit",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Department of Medicine, Chulabhorn Hospital, Chulabhorn Royal Academy",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Payoong",
"given": "Paruspak",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "National Institute of Health, Department of Medical Sciences, Ministry of Public Health",
"place": [
"Nonthaburi, Thailand"
]
}
],
"family": "Okada",
"given": "Pilailuk Akkapaiboon",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Laboratory of Pharmacology, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Rittapai",
"given": "Noppawan",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Laboratory of Pharmacology, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Suntararuks",
"given": "Sumitra",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Laboratory of Pharmacology, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Akanimanee",
"given": "Jaratluck",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Laboratory of Pharmacology, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
},
{
"name": "Center of Excellence on Environmental Health and Toxicology (EHT), OPS, MHESI",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Rangkadilok",
"given": "Nuchanart",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Translational Research Unit, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Panomvana",
"given": "Duangchit",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Translational Research Unit, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Puranajoti",
"given": "Porranee",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Laboratory of Pharmacology, Chulabhorn Research Institute",
"place": [
"Bangkok, Thailand"
]
},
{
"name": "Center of Excellence on Environmental Health and Toxicology (EHT), OPS, MHESI",
"place": [
"Bangkok, Thailand"
]
}
],
"family": "Satayavivad",
"given": "Jutamaad",
"sequence": "additional"
}
],
"container-title": "Frontiers in Pharmacology",
"container-title-short": "Front. Pharmacol.",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"frontiersin.org"
]
},
"created": {
"date-parts": [
[
2026,
3,
13
]
],
"date-time": "2026-03-13T10:40:21Z",
"timestamp": 1773398421000
},
"deposited": {
"date-parts": [
[
2026,
3,
13
]
],
"date-time": "2026-03-13T10:40:24Z",
"timestamp": 1773398424000
},
"funder": [
{
"DOI": "10.13039/501100004704",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100004704",
"id-type": "DOI"
}
],
"name": "National Research Council of Thailand"
},
{
"DOI": "10.13039/501100007959",
"doi-asserted-by": "publisher",
"id": [
{
"asserted-by": "publisher",
"id": "10.13039/501100007959",
"id-type": "DOI"
}
],
"name": "Chulabhorn Research Institute"
}
],
"indexed": {
"date-parts": [
[
2026,
3,
13
]
],
"date-time": "2026-03-13T14:51:40Z",
"timestamp": 1773413500264,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issued": {
"date-parts": [
[
2026,
3,
13
]
]
},
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
3,
13
]
],
"date-time": "2026-03-13T00:00:00Z",
"timestamp": 1773360000000
}
}
],
"link": [
{
"URL": "https://www.frontiersin.org/articles/10.3389/fphar.2026.1781740/full",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1965",
"original-title": [],
"prefix": "10.3389",
"published": {
"date-parts": [
[
2026,
3,
13
]
]
},
"published-online": {
"date-parts": [
[
2026,
3,
13
]
]
},
"publisher": "Frontiers Media SA",
"reference": [
{
"DOI": "10.26502/aimr.0146",
"article-title": "Effect of Andrographis paniculata treatment for nonimmune patients with early-stage COVID-19 on the prevention of pneumonia: a retrospective cohort study",
"author": "Benjaponpitak",
"doi-asserted-by": "publisher",
"first-page": "35",
"journal-title": "Arch. Intern. Med. Res.",
"key": "B1",
"volume": "6",
"year": "2023"
},
{
"DOI": "10.3109/13880209.2013.836544",
"article-title": "Pharmacokinetic analysis and tissue distribution of andrographolide in rat by a validated LC–MS/MS method",
"author": "Bera",
"doi-asserted-by": "publisher",
"first-page": "321",
"journal-title": "Pharm. Biol.",
"key": "B2",
"volume": "52",
"year": "2014"
},
{
"DOI": "10.1111/cts.70184",
"article-title": "Comparison of molnupiravir exposure‐response relationships for virology response and mechanism of action biomarkers with clinical outcomes in treatment of COVID‐19",
"author": "Chawla",
"doi-asserted-by": "publisher",
"first-page": "e70184",
"journal-title": "Clin. Transl. Sci.",
"key": "B3",
"volume": "18",
"year": "2025"
},
{
"DOI": "10.3389/fphar.2022.950012",
"article-title": "Therapeutic effects of herbal-medicine combined therapy for COVID-19: a systematic review and meta-analysis of randomized controlled trials",
"author": "Chien",
"doi-asserted-by": "publisher",
"first-page": "950012",
"journal-title": "Front. Pharmacol.",
"key": "B4",
"volume": "13",
"year": "2022"
},
{
"DOI": "10.1080/10408398.2018.1501657",
"article-title": "Overview of pharmacological activities of Andrographis paniculata and its major compound andrographolide",
"author": "Dai",
"doi-asserted-by": "publisher",
"first-page": "S17",
"journal-title": "Crit. Rev. Food Sci. Nutr.",
"key": "B5",
"volume": "59",
"year": "2019"
},
{
"key": "B6",
"volume-title": "Thai herbal pharmacopoeia",
"year": "2021"
},
{
"DOI": "10.1078/094471102321616391",
"article-title": "A double blind, placebo-controlled study of Andrographis paniculata fixed combination Kan Jang in the treatment of acute upper respiratory tract infections including sinusitis",
"author": "Gabrielian",
"doi-asserted-by": "publisher",
"first-page": "589",
"journal-title": "Phytomedicine",
"key": "B7",
"volume": "9",
"year": "2002"
},
{
"DOI": "10.1371/journal.ppat.1010802",
"article-title": "Infectious viral shedding of SARS-CoV-2 Delta following vaccination: a longitudinal cohort study",
"author": "Garcia-Knight",
"doi-asserted-by": "publisher",
"first-page": "e1010802",
"journal-title": "PLoS Pathog.",
"key": "B8",
"volume": "18",
"year": "2022"
},
{
"DOI": "10.3390/molecules27144479",
"article-title": "A comprehensive review of Andrographis paniculata (Burm. f.) Nees and its constituents as potential lead compounds for COVID-19 drug discovery",
"author": "Intharuksa",
"doi-asserted-by": "publisher",
"first-page": "4479",
"journal-title": "Molecules",
"key": "B9",
"volume": "27",
"year": "2022"
},
{
"DOI": "10.1016/j.jtcme.2022.02.001",
"article-title": "Natural products as a source of novel drugs for treating SARS-CoV2 infection",
"author": "Isidoro",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "J. Tradit. Complement. Med.",
"key": "B10",
"volume": "12",
"year": "2022"
},
{
"DOI": "10.1038/s41598-025-13652-6",
"article-title": "Degradation kinetics of andrographolide in aqueous solution, product identification and biological activity evaluation",
"author": "Jaidee",
"doi-asserted-by": "publisher",
"first-page": "28856",
"journal-title": "Sci. Rep.",
"key": "B11",
"volume": "15",
"year": "2025"
},
{
"DOI": "10.1039/d0ra10529e",
"article-title": "Combination of system biology to probe the anti-viral activity of andrographolide and its derivative against COVID-19",
"author": "Khanal",
"doi-asserted-by": "publisher",
"first-page": "5065",
"journal-title": "RSC Adv.",
"key": "B12",
"volume": "11",
"year": "2021"
},
{
"DOI": "10.1016/j.jtcme.2024.05.004",
"article-title": "In vivo evaluation of Andrographis paniculata and Boesenbergia rotunda extract activity against SARS-CoV-2 Delta variant in golden Syrian hamsters",
"author": "Kongsomros",
"doi-asserted-by": "publisher",
"first-page": "598",
"journal-title": "J. Tradit. Complement. Med.",
"key": "B13",
"volume": "14",
"year": "2024"
},
{
"DOI": "10.1007/s10096-020-03913-9",
"article-title": "Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards",
"author": "La Scola",
"doi-asserted-by": "publisher",
"first-page": "1059",
"journal-title": "Eur. J. Clin. Microbiol. Infect. Dis.",
"key": "B14",
"volume": "39",
"year": "2020"
},
{
"DOI": "10.1111/j.1440-1681.2011.05633.x",
"article-title": "Andrographolide and its analogues: versatile bioactive molecules for combating inflammation and cancer",
"author": "Lim",
"doi-asserted-by": "publisher",
"first-page": "300",
"journal-title": "Clin. Exp. Pharmacol. Physiol.",
"key": "B15",
"volume": "39",
"year": "2012"
},
{
"DOI": "10.3389/fimmu.2022.1062067",
"article-title": "Robust humoral and cellular recall responses to AZD1222 attenuate breakthrough SARS-CoV-2 infection compared to unvaccinated",
"author": "Maaske",
"doi-asserted-by": "publisher",
"first-page": "1062067",
"journal-title": "Front. Immunol.",
"key": "B16",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1002/jmv.27307",
"article-title": "High proportion of low cycle threshold value as an early indicator of COVID‐19 surge",
"author": "Mishra",
"doi-asserted-by": "publisher",
"first-page": "240",
"journal-title": "J. Med. Virol.",
"key": "B17",
"volume": "94",
"year": "2022"
},
{
"DOI": "10.1038/s41586-022-05522-2",
"article-title": "The WHO estimates of excess mortality associated with the COVID-19 pandemic",
"author": "Msemburi",
"doi-asserted-by": "publisher",
"first-page": "130",
"journal-title": "Nature",
"key": "B18",
"volume": "613",
"year": "2023"
},
{
"DOI": "10.1080/07391102.2020.1777901",
"article-title": "Computational investigation on Andrographis paniculata phytochemicals to evaluate their potency against SARS-CoV-2 in comparison to known antiviral compounds in drug trials",
"author": "Murugan",
"doi-asserted-by": "publisher",
"first-page": "4415",
"journal-title": "J. Biomol. Struct. Dyn.",
"key": "B19",
"volume": "39",
"year": "2021"
},
{
"DOI": "10.1016/S0944-7113(00)80054-9",
"article-title": "Pharmacokinetic and oral bioavailability of andrographolide from Andrographis paniculata fixed combination Kan Jang in rats and humans",
"author": "Panossian",
"doi-asserted-by": "publisher",
"first-page": "351",
"journal-title": "Phytomedicine",
"key": "B20",
"volume": "7",
"year": "2000"
},
{
"DOI": "10.1186/1749-8546-8-2",
"article-title": "Changes in the contents of four active diterpenoids at different growth stages in Andrographis paniculata (Burm.f.) nees (Chuanxinlian)",
"author": "Pholphana",
"doi-asserted-by": "publisher",
"first-page": "2",
"journal-title": "Chin. Med.",
"key": "B21",
"volume": "8",
"year": "2013"
},
{
"DOI": "10.1016/S2213-2600(20)30527-0",
"article-title": "Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: a nationwide, population-based retrospective cohort study",
"author": "Piroth",
"doi-asserted-by": "publisher",
"first-page": "251",
"journal-title": "Lancet Respir. Med.",
"key": "B22",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1002/pca.3364",
"article-title": "Comparison of diterpenoid contents and dissolution profiles of selected Andrographis paniculata crude and extract capsules",
"author": "Rangkadilok",
"doi-asserted-by": "publisher",
"first-page": "1309",
"journal-title": "Phytochem. Anal.",
"key": "B23",
"volume": "35",
"year": "2024"
},
{
"DOI": "10.3390/ph15081013",
"article-title": "Efficacy of Kan jang® in patients with mild COVID-19: interim analysis of a randomized, quadruple-blind, placebo-controlled trial",
"author": "Ratiani",
"doi-asserted-by": "publisher",
"first-page": "1013",
"journal-title": "Pharmaceuticals",
"key": "B24",
"volume": "15",
"year": "2022"
},
{
"author": "Rowland",
"key": "B25",
"volume-title": "Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications",
"year": "2011"
},
{
"DOI": "10.1021/acs.jnatprod.0c01324",
"article-title": "Anti-SARS-CoV-2 activity of Andrographis paniculata extract and its major component andrographolide in human lung epithelial cells and cytotoxicity evaluation in major organ cell representatives",
"author": "Sa-Ngiamsuntorn",
"doi-asserted-by": "publisher",
"first-page": "1261",
"journal-title": "J. Nat. Prod.",
"key": "B26",
"volume": "84",
"year": "2021"
},
{
"DOI": "10.4103/2229-3485.71856",
"article-title": "Sample size estimation in clinical trial",
"author": "Sakpal",
"doi-asserted-by": "crossref",
"first-page": "67",
"journal-title": "Perspect. Clin. Res.",
"key": "B27",
"volume": "1",
"year": "2010"
},
{
"DOI": "10.3389/fphar.2022.952660",
"article-title": "A comprehensive review on disposition kinetics and dosage of oral administration of Andrographis paniculata, an alternative herbal medicine, in co-treatment of coronavirus disease",
"author": "Songvut",
"doi-asserted-by": "publisher",
"first-page": "952660",
"journal-title": "Front. Pharmacol.",
"key": "B28",
"volume": "13",
"year": "2022"
},
{
"DOI": "10.1038/s41598-023-28612-1",
"article-title": "A validated LC–MS/MS method for clinical pharmacokinetics and presumptive phase II metabolic pathways following oral administration of Andrographis paniculata extract",
"author": "Songvut",
"doi-asserted-by": "publisher",
"first-page": "2534",
"journal-title": "Sci. Rep.",
"key": "B29",
"volume": "13",
"year": ""
},
{
"DOI": "10.3389/fphar.2023.1230401",
"article-title": "Comparative pharmacokinetics and safety evaluation of high dosage regimens of Andrographis paniculata aqueous extract after single and multiple oral administration in healthy participants",
"author": "Songvut",
"doi-asserted-by": "publisher",
"first-page": "1230401",
"journal-title": "Front. Pharmacol.",
"key": "B30",
"volume": "14",
"year": ""
},
{
"DOI": "10.1080/13880209.2024.2444446",
"article-title": "Non-linear oral bioavailability and clinical pharmacokinetics of high-dose Andrographis paniculata ethanolic extract: relevant dosage implications for COVID-19 treatment",
"author": "Songvut",
"doi-asserted-by": "publisher",
"first-page": "42",
"journal-title": "Pharm. Biol.",
"key": "B31",
"volume": "63",
"year": "2025"
},
{
"DOI": "10.1186/s12879-024-10059-y",
"article-title": "Post-viral symptoms and conditions are more frequent in COVID-19 than influenza, but not more persistent",
"author": "Tesch",
"doi-asserted-by": "publisher",
"first-page": "1126",
"journal-title": "BMC Infect. Dis.",
"key": "B32",
"volume": "24",
"year": "2024"
},
{
"DOI": "10.1038/s41564-020-0761-6",
"article-title": "Analytical sensitivity and efficiency comparisons of SARS-CoV-2 RT–qPCR primer–probe sets",
"author": "Vogels",
"doi-asserted-by": "publisher",
"first-page": "1299",
"journal-title": "Nat. Microbiol.",
"key": "B33",
"volume": "5",
"year": "2020"
},
{
"DOI": "10.1038/aps.2009.205",
"article-title": "Immunomodulatory activity of andrographolide on macrophage activation and specific antibody response",
"author": "Wang",
"doi-asserted-by": "publisher",
"first-page": "191",
"journal-title": "Acta Pharmacol. Sin.",
"key": "B34",
"volume": "31",
"year": "2010"
},
{
"key": "B35",
"unstructured": "Clinical management of COVID-19: Interim guidance, 27 may 2020. WHO/2019-nCoV/clinical/2020\n \n 5\n 2020"
},
{
"DOI": "10.1093/cid/ciaa1280",
"article-title": "Viral dynamics and immune correlates of coronavirus disease 2019 (COVID-19) severity",
"author": "Young",
"doi-asserted-by": "publisher",
"first-page": "e2932",
"journal-title": "Clin. Infect. Dis.",
"key": "B36",
"volume": "73",
"year": "2021"
},
{
"DOI": "10.1002/ptr.7141",
"article-title": "Efficacy and safety of Xiyanping injection in the treatment of COVID-19: a multicenter, prospective, open-label and randomized controlled trial",
"author": "Zhang",
"doi-asserted-by": "publisher",
"first-page": "4401",
"journal-title": "Phytother. Res.",
"key": "B37",
"volume": "35",
"year": "2021"
}
],
"reference-count": 37,
"references-count": 37,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.frontiersin.org/articles/10.3389/fphar.2026.1781740/full"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Exploratory pharmacokinetic-pharmacodynamic characterization and safety of standardized Andrographis paniculata aqueous extract capsules in patients with mild COVID-19",
"type": "journal-article",
"update-policy": "https://doi.org/10.3389/crossmark-policy",
"volume": "17"
}