Androgen-deprivation therapies for prostate cancer and risk of infection by SARS-CoV-2: a population-based study (N = 4532)
et al., Annals of Oncology, doi:10.1016/j.annonc.2020.04.479, May 2020
7th treatment shown to reduce risk in
September 2020, now with p = 0.000000056 from 49 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
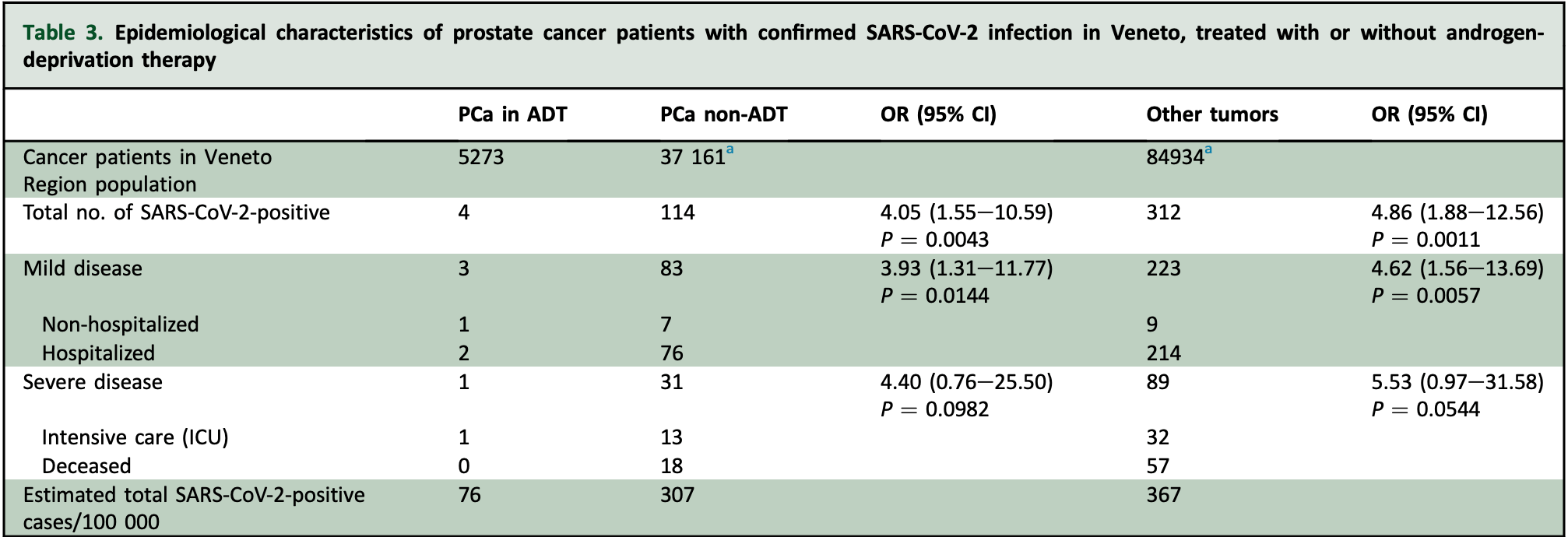
Retrospective 5,273 prostate cancer patients on androgen-deprivation therapy (ADT), and 37,161 not on ADT, showing lower risk of cases with treatment.
|
risk of death, 95.4% lower, RR 0.05, p = 0.15, treatment 0 of 5,273 (0.0%), control 18 of 37,161 (0.0%), NNT 2064, relative risk is not 0 because of continuity correction due to zero events (with reciprocal of the contrasting arm).
|
|
risk of severe case, 74.5% lower, RR 0.25, p = 0.01, treatment 1 of 5,273 (0.0%), control 31 of 37,161 (0.1%), NNT 1551, inverted to make RR<1 favor treatment, odds ratio converted to relative risk.
|
|
risk of case, 75.3% lower, RR 0.25, p = 0.004, treatment 4 of 5,273 (0.1%), control 114 of 37,161 (0.3%), NNT 433, inverted to make RR<1 favor treatment, odds ratio converted to relative risk.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Montopoli et al., 6 May 2020, retrospective, Italy, peer-reviewed, 12 authors.
Androgen-deprivation therapies for prostate cancer and risk of infection by SARS-CoV-2: a population-based study (N = 4532)
Annals of Oncology, doi:10.1016/j.annonc.2020.04.479
Background: Cell entry of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) depends on binding of the viral spike (S) proteins to angiotensin-converting enzyme 2 and on S protein priming by TMPRSS2. Inhibition of TMPRSS2 may work to block or decrease the severity of SARS-CoV-2 infections. Intriguingly, TMPRSS2 is an androgen-regulated gene that is up-regulated in prostate cancer where it supports tumor progression and is involved in a frequent genetic translocation with the ERG gene. First-or second-generation androgen-deprivation therapies (ADTs) decrease the levels of TMPRSS2. Here we put forward the hypothesis that ADTs may protect patients affected by prostate cancer from SARS-CoV-2 infections. Materials and methods: We extracted data regarding 9280 patients (4532 males) with laboratory-confirmed SARS-CoV-2 infection from 68 hospitals in Veneto, one of the Italian regions that was most affected by the coronavirus disease 2019 (COVID-19) pandemic. The parameters used for each COVID-19-positive patient were sex, hospitalization, admission to intensive care unit, death, tumor diagnosis, prostate cancer diagnosis, and ADT. Results: There were evaluable 9280 SARS-CoV-2-positive patients in Veneto on 1 April 2020. Overall, males developed more severe complications, were more frequently hospitalized, and had a worse clinical outcome than females. Considering only the Veneto male population (2.4 million men), 0.2% and 0.3% of non-cancer and cancer patients, respectively, tested positive for SARS-CoV-2. Comparing the total number of SARS-CoV-2-positive cases, prostate cancer patients receiving ADT had a significantly lower risk of SARS-CoV-2 infection compared with patients who did not receive ADT (OR 4.05; 95% CI 1.55e10.59). A greater difference was found comparing prostate cancer patients receiving ADT with patients with any other type of cancer (OR 4.86; 95% CI 1.88e12.56). Conclusion: Our data suggest that cancer patients have an increased risk of SARS-CoV-2 infections compared with noncancer patients. However, prostate cancer patients receiving ADT appear to be partially protected from SARS-CoV-2 infections.
References
Arora, Schenkein, Murali, Glucocorticoid receptor confers resistance to antiandrogens by bypassing androgen receptor blockade, Cell
Asangani, Dommeti, Wang, Therapeutic targeting of BET bromodomain proteins in castration-resistant prostate cancer, Nature
Barnes, Adrover, Baxter-Stoltzfus, Targeting potential drivers of COVID-19: neutrophil extracellular traps, J Exp Med
Bugge, Antalis, Wu, Type II transmembrane serine proteases, J Biol Chem
Cai, Sex difference and smoking predisposition in patients with COVID-19, Lancet Respir Med
Choi, Bertram, Glowacka, Park, Pohlmann, Type II transmembrane serine proteases in cancer and viral infections, Trends Mol Med
Drosten, Gunther, Preiser, Identification of a novel coronavirus in patients with severe acute respiratory syndrome, N Engl J Med
Faivre, Wilcox, Lin, Exploitation of castration-resistant prostate cancer transcription factor dependencies by the novel bet inhibitor ABBV-075, Mol Cancer Res
Ferlay, Colombet, Soerjomataram, Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018, Eur J Cancer
Glowacka, Bertram, Muller, Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response, J Virol
Gordon, Bhadel, Wunderlich, CDK9 regulates AR promoter selectivity and cell growth through serine 81 phosphorylation, Mol Endocrinol
Guan, Ni, Hu, Clinical characteristics of coronavirus disease 2019 in China, N Engl J Med
Hoffmann, Kleine-Weber, Schroeder, SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor, Cell
Huang, Wang, Li, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet
Ianculescu, Wu, Siegmund, Stallcup, Selective roles for cAMP response element-binding protein binding protein and p300 protein as coregulators for androgen-regulated gene expression in advanced prostate cancer cells, J Biol Chem
Iwata-Yoshikawa, Okamura, Shimizu, Hasegawa, Takeda et al., TMPRSS2 contributes to virus spread and immunopathology in the airways of murine models after coronavirus infection, J Virol
Klein, Flanagan, Sex differences in immune responses, Nat Rev Immunol
Ksiazek, Erdman, Goldsmith, A novel coronavirus associated with severe acute respiratory syndrome, N Engl J Med
Lazzerini, Putoto, COVID-19 in Italy: momentous decisions and many uncertainties, Lancet Glob Health
Liang, Guan, Chen, Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China, Lancet Oncol
Lucas, Heinlein, Kim, The androgen-regulated protease TMPRSS2 activates a proteolytic cascade involving components of the tumor microenvironment and promotes prostate cancer metastasis, Cancer Discov
Lucas, True, Hawley, The androgen-regulated type II serine protease TMPRSS2 is differentially expressed and mislocalized in prostate adenocarcinoma, J Pathol
Matsuyama, Nagata, Shirato, Kawase, Takeda et al., Efficient activation of the severe acute respiratory syndrome coronavirus spike protein by the transmembrane protease TMPRSS2, J Virol
Matsuyama, Nao, Shirato, Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells, Proc Natl Acad Sci U S A
Miettinen, Nurminen, Comparative analysis of two rates, Stat Med
Mikkonen, Pihlajamaa, Sahu, Zhang, Janne, Androgen receptor and androgen-dependent gene expression in lung, Mol Cell Endocrinol
Remuzzi, Remuzzi, COVID-19 and Italy: what next?, Lancet
Shaw, Whitaker, Corcoran, The early effects of rapid androgen deprivation on human prostate cancer, Eur Urol
Shen, Mao, Wu, Tanaka, Zhang, TMPRSS2: a potential target for treatment of influenza virus and coronavirus infections, Biochimie
Sheskin, Handbook of Parametric and Nonparametric Statistical Procedures
Shin, Seong, Type II transmembrane serine proteases as potential target for anti-influenza drug discovery, Expert Opin Drug Discov
Shinde, Albino, Zoma, Transcriptional reprogramming and inhibition of tumor-propagating stem-like cells by EC-8042 in ERG-positive prostate cancer, Eur Urol Oncol
Shulla, Heald-Sargent, Subramanya, Zhao, Perlman et al., A transmembrane serine protease is linked to the severe acute respiratory syndrome coronavirus receptor and activates virus entry, J Virol
Tarnow, Engels, Arendt, TMPRSS2 is a host factor that is essential for pneumotropism and pathogenicity of H7N9 influenza A virus in mice, J Virol
Tomlins, Rhodes, Perner, Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer, Science
Xu, Wang, Xiao, Nuclear receptor ERRalpha and transcription factor ERG form a reciprocal loop in the regulation of TMPRSS2:ERG fusion gene in prostate cancer, Oncogene
Zaki, Van Boheemen, Bestebroer, Osterhaus, Fouchier, Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia, N Engl J Med
Zhou, Yang, Wang, A pneumonia outbreak associated with a new coronavirus of probable bat origin, Nature
Zhu, Zhang, Wang, A novel coronavirus from patients with pneumonia in China, 2019, N Engl J Med
DOI record:
{
"DOI": "10.1016/j.annonc.2020.04.479",
"ISSN": [
"0923-7534"
],
"URL": "http://dx.doi.org/10.1016/j.annonc.2020.04.479",
"alternative-id": [
"S0923753420397970"
],
"author": [
{
"affiliation": [],
"family": "Montopoli",
"given": "M.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Zumerle",
"given": "S.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Vettor",
"given": "R.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Rugge",
"given": "M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Zorzi",
"given": "M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Catapano",
"given": "C.V.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Carbone",
"given": "G.M.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Cavalli",
"given": "A.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Pagano",
"given": "F.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ragazzi",
"given": "E.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Prayer-Galetti",
"given": "T.",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Alimonti",
"given": "A.",
"sequence": "additional"
}
],
"container-title": "Annals of Oncology",
"container-title-short": "Annals of Oncology",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2020,
5,
6
]
],
"date-time": "2020-05-06T23:12:25Z",
"timestamp": 1588806745000
},
"deposited": {
"date-parts": [
[
2021,
8,
2
]
],
"date-time": "2021-08-02T18:35:23Z",
"timestamp": 1627929323000
},
"funder": [
{
"DOI": "10.13039/501100004361",
"doi-asserted-by": "publisher",
"name": "Krebsliga Schweiz"
},
{
"DOI": "10.13039/501100005010",
"doi-asserted-by": "publisher",
"name": "Associazione Italiana per la Ricerca sul Cancro"
},
{
"DOI": "10.13039/501100001711",
"doi-asserted-by": "publisher",
"name": "Schweizerischer Nationalfonds zur Förderung der Wissenschaftlichen Forschung"
},
{
"DOI": "10.13039/501100000781",
"doi-asserted-by": "publisher",
"name": "European Research Council"
},
{
"DOI": "10.13039/100000892",
"doi-asserted-by": "publisher",
"name": "Prostate Cancer Foundation"
}
],
"indexed": {
"date-parts": [
[
2024,
4,
9
]
],
"date-time": "2024-04-09T17:18:12Z",
"timestamp": 1712683092241
},
"is-referenced-by-count": 426,
"issue": "8",
"issued": {
"date-parts": [
[
2020,
8
]
]
},
"journal-issue": {
"issue": "8",
"published-print": {
"date-parts": [
[
2020,
8
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2020,
8,
1
]
],
"date-time": "2020-08-01T00:00:00Z",
"timestamp": 1596240000000
}
},
{
"URL": "http://www.elsevier.com/open-access/userlicense/1.0/",
"content-version": "vor",
"delay-in-days": 365,
"start": {
"date-parts": [
[
2021,
8,
1
]
],
"date-time": "2021-08-01T00:00:00Z",
"timestamp": 1627776000000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0923753420397970?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0923753420397970?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "1040-1045",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2020,
8
]
]
},
"published-print": {
"date-parts": [
[
2020,
8
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1016/S0140-6736(20)30183-5",
"article-title": "Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China",
"author": "Huang",
"doi-asserted-by": "crossref",
"first-page": "497",
"journal-title": "Lancet",
"key": "10.1016/j.annonc.2020.04.479_bib1",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1038/s41586-020-2012-7",
"article-title": "A pneumonia outbreak associated with a new coronavirus of probable bat origin",
"author": "Zhou",
"doi-asserted-by": "crossref",
"first-page": "270",
"journal-title": "Nature",
"key": "10.1016/j.annonc.2020.04.479_bib2",
"volume": "579",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2001017",
"article-title": "A novel coronavirus from patients with pneumonia in China, 2019",
"author": "Zhu",
"doi-asserted-by": "crossref",
"first-page": "727",
"journal-title": "N Engl J Med",
"key": "10.1016/j.annonc.2020.04.479_bib3",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2002032",
"article-title": "Clinical characteristics of coronavirus disease 2019 in China",
"author": "Guan",
"doi-asserted-by": "crossref",
"first-page": "1708",
"journal-title": "N Engl J Med",
"key": "10.1016/j.annonc.2020.04.479_bib4",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1074/jbc.R109.021006",
"article-title": "Type II transmembrane serine proteases",
"author": "Bugge",
"doi-asserted-by": "crossref",
"first-page": "23177",
"journal-title": "J Biol Chem",
"key": "10.1016/j.annonc.2020.04.479_bib6",
"volume": "284",
"year": "2009"
},
{
"DOI": "10.1016/j.molmed.2009.05.003",
"article-title": "Type II transmembrane serine proteases in cancer and viral infections",
"author": "Choi",
"doi-asserted-by": "crossref",
"first-page": "303",
"journal-title": "Trends Mol Med",
"key": "10.1016/j.annonc.2020.04.479_bib7",
"volume": "15",
"year": "2009"
},
{
"DOI": "10.1080/17460441.2017.1372417",
"article-title": "Type II transmembrane serine proteases as potential target for anti-influenza drug discovery",
"author": "Shin",
"doi-asserted-by": "crossref",
"first-page": "1139",
"journal-title": "Expert Opin Drug Discov",
"key": "10.1016/j.annonc.2020.04.479_bib8",
"volume": "12",
"year": "2017"
},
{
"DOI": "10.1128/JVI.02232-10",
"article-title": "Evidence that TMPRSS2 activates the severe acute respiratory syndrome coronavirus spike protein for membrane fusion and reduces viral control by the humoral immune response",
"author": "Glowacka",
"doi-asserted-by": "crossref",
"first-page": "4122",
"journal-title": "J Virol",
"key": "10.1016/j.annonc.2020.04.479_bib9",
"volume": "85",
"year": "2011"
},
{
"DOI": "10.1128/JVI.01815-18",
"article-title": "TMPRSS2 contributes to virus spread and immunopathology in the airways of murine models after coronavirus infection",
"author": "Iwata-Yoshikawa",
"doi-asserted-by": "crossref",
"first-page": "e01815",
"journal-title": "J Virol",
"key": "10.1016/j.annonc.2020.04.479_bib10",
"volume": "93",
"year": "2019"
},
{
"DOI": "10.1128/JVI.01542-10",
"article-title": "Efficient activation of the severe acute respiratory syndrome coronavirus spike protein by the transmembrane protease TMPRSS2",
"author": "Matsuyama",
"doi-asserted-by": "crossref",
"first-page": "12658",
"journal-title": "J Virol",
"key": "10.1016/j.annonc.2020.04.479_bib11",
"volume": "84",
"year": "2010"
},
{
"DOI": "10.1016/j.biochi.2017.07.016",
"article-title": "TMPRSS2: a potential target for treatment of influenza virus and coronavirus infections",
"author": "Shen",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Biochimie",
"key": "10.1016/j.annonc.2020.04.479_bib12",
"volume": "142",
"year": "2017"
},
{
"DOI": "10.1128/JVI.02062-10",
"article-title": "A transmembrane serine protease is linked to the severe acute respiratory syndrome coronavirus receptor and activates virus entry",
"author": "Shulla",
"doi-asserted-by": "crossref",
"first-page": "873",
"journal-title": "J Virol",
"key": "10.1016/j.annonc.2020.04.479_bib13",
"volume": "85",
"year": "2011"
},
{
"DOI": "10.1128/JVI.03799-13",
"article-title": "TMPRSS2 is a host factor that is essential for pneumotropism and pathogenicity of H7N9 influenza A virus in mice",
"author": "Tarnow",
"doi-asserted-by": "crossref",
"first-page": "4744",
"journal-title": "J Virol",
"key": "10.1016/j.annonc.2020.04.479_bib14",
"volume": "88",
"year": "2014"
},
{
"DOI": "10.1016/j.cell.2020.02.052",
"article-title": "SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor",
"author": "Hoffmann",
"doi-asserted-by": "crossref",
"first-page": "271",
"journal-title": "Cell",
"key": "10.1016/j.annonc.2020.04.479_bib15",
"volume": "181",
"year": "2020"
},
{
"DOI": "10.1073/pnas.2002589117",
"article-title": "Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells",
"author": "Matsuyama",
"doi-asserted-by": "crossref",
"first-page": "7001",
"journal-title": "Proc Natl Acad Sci U S A",
"key": "10.1016/j.annonc.2020.04.479_bib16",
"volume": "117",
"year": "2020"
},
{
"DOI": "10.1158/2159-8290.CD-13-1010",
"article-title": "The androgen-regulated protease TMPRSS2 activates a proteolytic cascade involving components of the tumor microenvironment and promotes prostate cancer metastasis",
"author": "Lucas",
"doi-asserted-by": "crossref",
"first-page": "1310",
"journal-title": "Cancer Discov",
"key": "10.1016/j.annonc.2020.04.479_bib17",
"volume": "4",
"year": "2014"
},
{
"DOI": "10.1002/path.2330",
"article-title": "The androgen-regulated type II serine protease TMPRSS2 is differentially expressed and mislocalized in prostate adenocarcinoma",
"author": "Lucas",
"doi-asserted-by": "crossref",
"first-page": "118",
"journal-title": "J Pathol",
"key": "10.1016/j.annonc.2020.04.479_bib18",
"volume": "215",
"year": "2008"
},
{
"DOI": "10.1016/j.mce.2009.12.022",
"article-title": "Androgen receptor and androgen-dependent gene expression in lung",
"author": "Mikkonen",
"doi-asserted-by": "crossref",
"first-page": "14",
"journal-title": "Mol Cell Endocrinol",
"key": "10.1016/j.annonc.2020.04.479_bib19",
"volume": "317",
"year": "2010"
},
{
"DOI": "10.1002/sim.4780040211",
"article-title": "Comparative analysis of two rates",
"author": "Miettinen",
"doi-asserted-by": "crossref",
"first-page": "213",
"journal-title": "Stat Med",
"key": "10.1016/j.annonc.2020.04.479_bib20",
"volume": "4",
"year": "1985"
},
{
"author": "Sheskin",
"key": "10.1016/j.annonc.2020.04.479_bib21",
"series-title": "Handbook of Parametric and Nonparametric Statistical Procedures",
"year": "2004"
},
{
"DOI": "10.1016/S2213-2600(20)30117-X",
"article-title": "Sex difference and smoking predisposition in patients with COVID-19",
"author": "Cai",
"doi-asserted-by": "crossref",
"first-page": "e20",
"journal-title": "Lancet Respir Med",
"key": "10.1016/j.annonc.2020.04.479_bib22",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/j.ejca.2018.07.005",
"article-title": "Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018",
"author": "Ferlay",
"doi-asserted-by": "crossref",
"first-page": "356",
"journal-title": "Eur J Cancer",
"key": "10.1016/j.annonc.2020.04.479_bib23",
"volume": "103",
"year": "2018"
},
{
"DOI": "10.1016/S1470-2045(20)30096-6",
"article-title": "Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China",
"author": "Liang",
"doi-asserted-by": "crossref",
"first-page": "335",
"journal-title": "Lancet Oncol",
"key": "10.1016/j.annonc.2020.04.479_bib24",
"volume": "21",
"year": "2020"
},
{
"DOI": "10.1016/S2214-109X(20)30110-8",
"article-title": "COVID-19 in Italy: momentous decisions and many uncertainties",
"author": "Lazzerini",
"doi-asserted-by": "crossref",
"first-page": "e641",
"journal-title": "Lancet Glob Health",
"key": "10.1016/j.annonc.2020.04.479_bib25",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30627-9",
"article-title": "COVID-19 and Italy: what next?",
"author": "Remuzzi",
"doi-asserted-by": "crossref",
"first-page": "1225",
"journal-title": "Lancet",
"key": "10.1016/j.annonc.2020.04.479_bib26",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa030747",
"article-title": "Identification of a novel coronavirus in patients with severe acute respiratory syndrome",
"author": "Drosten",
"doi-asserted-by": "crossref",
"first-page": "1967",
"journal-title": "N Engl J Med",
"key": "10.1016/j.annonc.2020.04.479_bib27",
"volume": "348",
"year": "2003"
},
{
"DOI": "10.1056/NEJMoa030781",
"article-title": "A novel coronavirus associated with severe acute respiratory syndrome",
"author": "Ksiazek",
"doi-asserted-by": "crossref",
"first-page": "1953",
"journal-title": "N Engl J Med",
"key": "10.1016/j.annonc.2020.04.479_bib28",
"volume": "348",
"year": "2003"
},
{
"DOI": "10.1056/NEJMoa1211721",
"article-title": "Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia",
"author": "Zaki",
"doi-asserted-by": "crossref",
"first-page": "1814",
"journal-title": "N Engl J Med",
"key": "10.1016/j.annonc.2020.04.479_bib29",
"volume": "367",
"year": "2012"
},
{
"DOI": "10.1126/science.1117679",
"article-title": "Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer",
"author": "Tomlins",
"doi-asserted-by": "crossref",
"first-page": "644",
"journal-title": "Science",
"key": "10.1016/j.annonc.2020.04.479_bib30",
"volume": "310",
"year": "2005"
},
{
"DOI": "10.1016/j.eururo.2015.10.042",
"article-title": "The early effects of rapid androgen deprivation on human prostate cancer",
"author": "Shaw",
"doi-asserted-by": "crossref",
"first-page": "214",
"journal-title": "Eur Urol",
"key": "10.1016/j.annonc.2020.04.479_bib31",
"volume": "70",
"year": "2016"
},
{
"DOI": "10.1038/nature13229",
"article-title": "Therapeutic targeting of BET bromodomain proteins in castration-resistant prostate cancer",
"author": "Asangani",
"doi-asserted-by": "crossref",
"first-page": "278",
"journal-title": "Nature",
"key": "10.1016/j.annonc.2020.04.479_bib32",
"volume": "510",
"year": "2014"
},
{
"DOI": "10.1158/1541-7786.MCR-16-0221",
"article-title": "Exploitation of castration-resistant prostate cancer transcription factor dependencies by the novel bet inhibitor ABBV-075",
"author": "Faivre",
"doi-asserted-by": "crossref",
"first-page": "35",
"journal-title": "Mol Cancer Res",
"key": "10.1016/j.annonc.2020.04.479_bib33",
"volume": "15",
"year": "2017"
},
{
"DOI": "10.1074/jbc.M111.300194",
"article-title": "Selective roles for cAMP response element-binding protein binding protein and p300 protein as coregulators for androgen-regulated gene expression in advanced prostate cancer cells",
"author": "Ianculescu",
"doi-asserted-by": "crossref",
"first-page": "4000",
"journal-title": "J Biol Chem",
"key": "10.1016/j.annonc.2020.04.479_bib34",
"volume": "287",
"year": "2012"
},
{
"DOI": "10.1210/me.2010-0238",
"article-title": "CDK9 regulates AR promoter selectivity and cell growth through serine 81 phosphorylation",
"author": "Gordon",
"doi-asserted-by": "crossref",
"first-page": "2267",
"journal-title": "Mol Endocrinol",
"key": "10.1016/j.annonc.2020.04.479_bib35",
"volume": "24",
"year": "2010"
},
{
"DOI": "10.1016/j.euo.2018.08.024",
"article-title": "Transcriptional reprogramming and inhibition of tumor-propagating stem-like cells by EC-8042 in ERG-positive prostate cancer",
"author": "Shinde",
"doi-asserted-by": "crossref",
"first-page": "415",
"journal-title": "Eur Urol Oncol",
"key": "10.1016/j.annonc.2020.04.479_bib36",
"volume": "2",
"year": "2019"
},
{
"DOI": "10.1038/nri.2016.90",
"article-title": "Sex differences in immune responses",
"author": "Klein",
"doi-asserted-by": "crossref",
"first-page": "626",
"journal-title": "Nat Rev Immunol",
"key": "10.1016/j.annonc.2020.04.479_bib37",
"volume": "16",
"year": "2016"
},
{
"DOI": "10.1084/jem.20200652",
"article-title": "Targeting potential drivers of COVID-19: neutrophil extracellular traps",
"author": "Barnes",
"doi-asserted-by": "crossref",
"first-page": "e20200652",
"journal-title": "J Exp Med",
"key": "10.1016/j.annonc.2020.04.479_bib38",
"volume": "217",
"year": "2020"
},
{
"DOI": "10.1016/j.cell.2013.11.012",
"article-title": "Glucocorticoid receptor confers resistance to antiandrogens by bypassing androgen receptor blockade",
"author": "Arora",
"doi-asserted-by": "crossref",
"first-page": "1309",
"journal-title": "Cell",
"key": "10.1016/j.annonc.2020.04.479_bib39",
"volume": "155",
"year": "2013"
},
{
"DOI": "10.1038/s41388-018-0409-7",
"article-title": "Nuclear receptor ERRalpha and transcription factor ERG form a reciprocal loop in the regulation of TMPRSS2:ERG fusion gene in prostate cancer",
"author": "Xu",
"doi-asserted-by": "crossref",
"first-page": "6259",
"journal-title": "Oncogene",
"key": "10.1016/j.annonc.2020.04.479_bib40",
"volume": "37",
"year": "2018"
}
],
"reference-count": 39,
"references-count": 39,
"relation": {
"has-review": [
{
"asserted-by": "object",
"id": "10.3410/f.737916363.793574732",
"id-type": "doi"
}
]
},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S0923753420397970"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Oncology",
"Hematology"
],
"subtitle": [],
"title": "Androgen-deprivation therapies for prostate cancer and risk of infection by SARS-CoV-2: a population-based study (N = 4532)",
"type": "journal-article",
"volume": "31"
}
