Circulating Liver-Enriched miR-122 in COVID-19 Patients: A Longitudinal Real-Life Study
et al., International Journal of Molecular Sciences, doi:10.3390/ijms27052288, Feb 2026
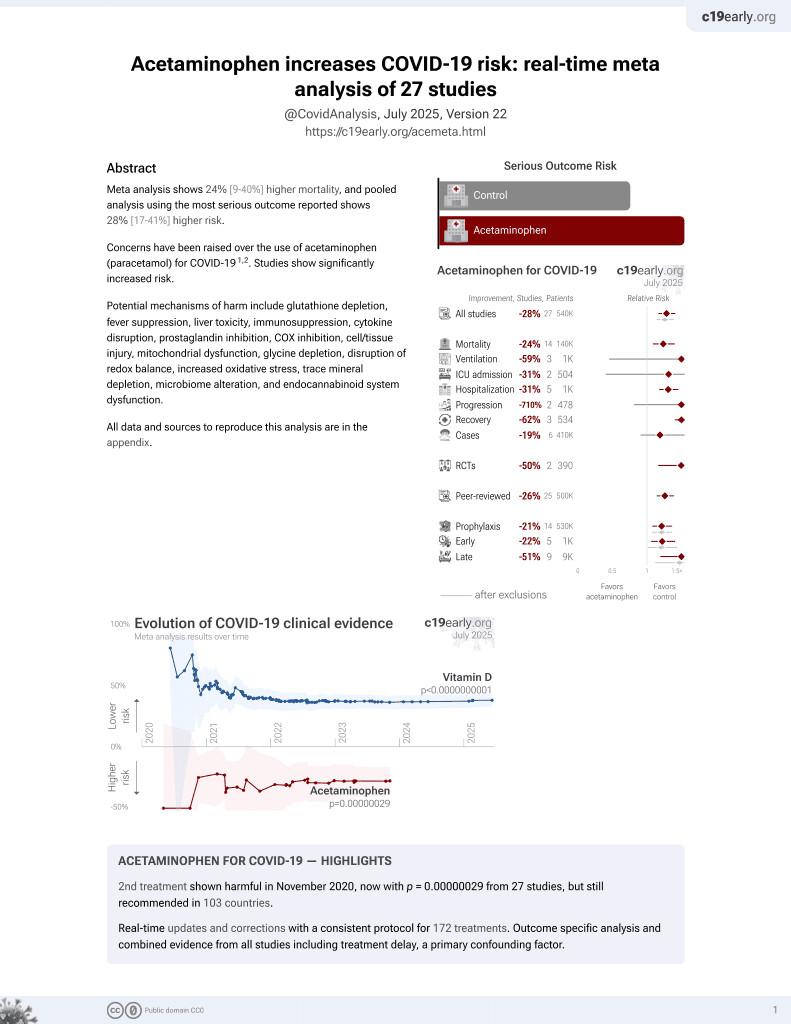
2nd treatment shown to increase risk in
November 2020, now with p = 0.00000029 from 27 studies, but still recommended in 103 countries.
6,600+ studies for
220+ treatments. c19early.org
|
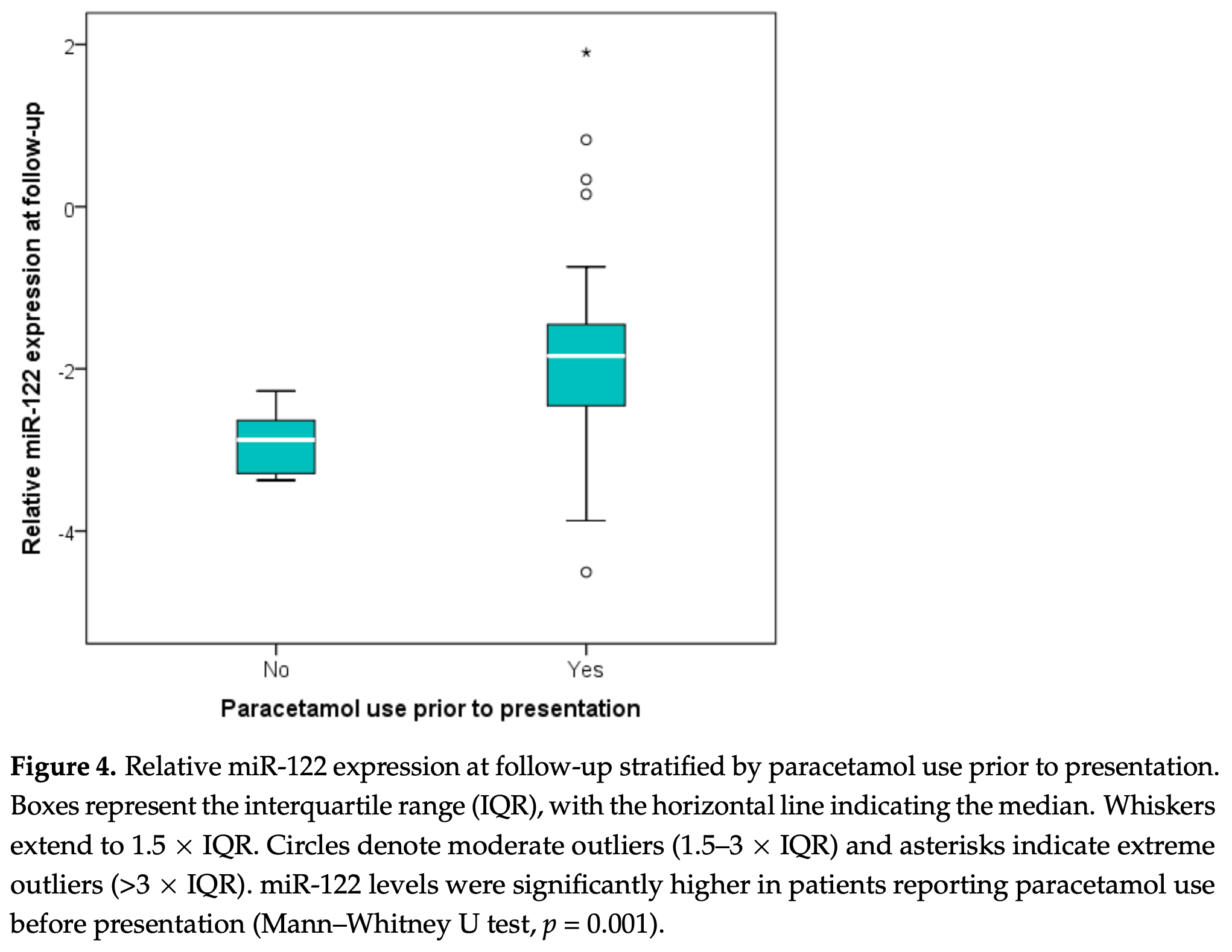
Prospective cohort study of 78 COVID-19 patients and 18 non-COVID-19 participants in Romania, reporting that prior therapeutic paracetamol use before hospital presentation was independently associated with higher miR-122 (a liver-specific biomarker of hepatocellular injury) expression at follow-up, suggesting cumulative hepatic stress in the context of COVID-19.
The most clinically relevant findings concern drug-associated effects on follow-up miR-122 levels. In bivariate analyses, both remdesivir administered for ≥5 days (p = 0.012) and prior therapeutic paracetamol use (p = 0.001) were associated with higher miR-122 at follow-up. However, in a multivariable model, only paracetamol retained independent significance (p = 0.011), leading the authors to conclude that therapeutic paracetamol may contribute to hepatocellular stress in COVID-19 patients.
Several features of the data structure warrant caution when interpreting this distinction. Remdesivir was administered to 96.5% of the longitudinal subgroup while 84.2% reported prior paracetamol use, meaning near-complete overlap between the two exposures. With only 2 patients not receiving remdesivir, the dataset provides almost no contrast for separating a remdesivir effect from a paracetamol effect. The potentially more informative remdesivir comparison - treatment for ≥5 days versus shorter courses (32 vs 25 patients) - did yield a significant bivariate result, but the paper does not report how this variable was parameterized in the multivariable model, nor does it provide the adjusted coefficient or p-value for remdesivir. Without the full regression output, it is unclear whether remdesivir was borderline significant after adjustment (suggesting a real but underpowered signal partially absorbed by the paracetamol variable) or clearly non-significant (suggesting confounding drove the bivariate finding). A cross-tabulation of paracetamol use by remdesivir duration group would also clarify the degree of collinearity between these two predictors on the parameterization that provides the most statistical leverage.
Paracetamol is also known as acetaminophen, Tylenol, Panadol, Calpol, Tempra, Calprofen, Doliprane, Efferalgan, Grippostad C, Dolo, Acamol, Fevadol, Crocin, and Perfalgan.
Study covers acetaminophen and remdesivir.
Mihai et al., 28 Feb 2026, prospective, Romania, peer-reviewed, 9 authors, study period March 2022 - July 2023.
Contact: catalin.tiliscan@umfcd.ro (corresponding author), nicoleta.mihai1@drd.umfcd.ro, oana-alexandra.ganea@drd.umfcd.ro, simona.ruta@umfcd.ro, victoria.arama@umfcd.ro, sorin.arama@umfcd.ro, andreea.arsene@umfcd.ro, iulia_iancu2005@yahoo.com, aida-isabela.adamescu@drd.umfcd.ro.
Abstract:
Article
Circulating Liver-Enriched miR-122 in COVID-19 Patients: ALongitudinal Real-Life Study
Nicoleta Mihai 1,4 , C ă t ă lin Tilis , can 2,4, *, Iulia Virginia Iancu 5 , Oana-Alexandra Ganea 1,4 , Aida-Isabela Adamescu 2,4, * , S , tefan Sorin Aram ă 2,4 , Andreea Letit ,ia Arsene 3 , Simona-Maria Rut , ă 1,5 and Victoria Aram ă 1,4
- 1 Faculty of Medicine, 'Carol Davila' University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania; nicoleta.mihai1@drd.umfcd.ro (N.M.);
oana-alexandra.ganea@drd.umfcd.ro (O.-A.G.); simona.ruta@umfcd.ro (S.-M.R.);
victoria.arama@umfcd.ro (V.A.)
- 2 Faculty of Dental Medicine, 'Carol Davila' University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania; sorin.arama@umfcd.ro
- 3 Faculty of Pharmacy, 'Carol Davila' University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania; andreea.arsene@umfcd.ro
- 4 'Prof. Dr. Matei Bals , ' National Institute for Infectious Diseases, 1 Calistrat Grozovici Street, 021105 Bucharest, Romania
- 5 '¸ Stefan S. Nicolau' Institute of Virology, 285 Mihai Bravu Road, 030304 Bucharest, Romania; iulia\_iancu2005@yahoo.com
* Correspondence: catalin.tiliscan@umfcd.ro (C.T.); aida-isabela.adamescu@drd.umfcd.ro (A.-I.A.)
Abstract
Abnormal liver function tests are frequently reported in patients with COVID-19. This study aimed to identify potential treatment-associated hepatocellular injury in COVID-19 patients by dynamically assessing circulating miR-122, a biomarker with high hepatic specificity and sensitivity. An exploratory approach was additionally used, given the limited evidence regarding factors influencing miR-122 expression in this setting. We performed a prospective cohort study including 96 adult participants enrolled at a tertiary hospital in Bucharest, Romania, between March 2022 and July 2023: 78 COVID-19 patients (57 with baseline and follow-up miR-122 assessment after 5 days of treatment and 21 with a single measurement) and 18 non-COVID-19 participants included for comparison. Plasma miR-122 levels were measured using quantitative polymerase chain reaction, normalized to U6 small nuclear RNA, and expressed as log10(2 -∆ Ct ). No associations were observed between miR-122 expression and remdesivir administered for standard treatment durations (3-5 days) or other COVID-19-specific therapies. However, a duration-dependent relationship with remdesivir cannot be excluded. Moreover, therapeutic paracetamol use prior to presentation was positively associated with miR-122 expression at follow-up and remained significant after adjustment. Additionally, bivariate analyses revealed inverse correlations between baseline miR-122 and inflammatory biomarkers, with multivariable analysis showing an independent positive association with lymphocyte count.
Keywords: miR-122; COVID-19; remdesivir; paracetamol; drug-induced liver injury; systemic inflammation
DOI record:
{
"DOI": "10.3390/ijms27052288",
"ISSN": [
"1422-0067"
],
"URL": "http://dx.doi.org/10.3390/ijms27052288",
"abstract": "<jats:p>Abnormal liver function tests are frequently reported in patients with COVID-19. This study aimed to identify potential treatment-associated hepatocellular injury in COVID-19 patients by dynamically assessing circulating miR-122, a biomarker with high hepatic specificity and sensitivity. An exploratory approach was additionally used, given the limited evidence regarding factors influencing miR-122 expression in this setting. We performed a prospective cohort study including 96 adult participants enrolled at a tertiary hospital in Bucharest, Romania, between March 2022 and July 2023: 78 COVID-19 patients (57 with baseline and follow-up miR-122 assessment after 5 days of treatment and 21 with a single measurement) and 18 non-COVID-19 participants included for comparison. Plasma miR-122 levels were measured using quantitative polymerase chain reaction, normalized to U6 small nuclear RNA, and expressed as log10(2−ΔCt). No associations were observed between miR-122 expression and remdesivir administered for standard treatment durations (3–5 days) or other COVID-19–specific therapies. However, a duration-dependent relationship with remdesivir cannot be excluded. Moreover, therapeutic paracetamol use prior to presentation was positively associated with miR-122 expression at follow-up and remained significant after adjustment. Additionally, bivariate analyses revealed inverse correlations between baseline miR-122 and inflammatory biomarkers, with multivariable analysis showing an independent positive association with lymphocyte count.</jats:p>",
"alternative-id": [
"ijms27052288"
],
"author": [
{
"ORCID": "https://orcid.org/0009-0005-3953-0767",
"affiliation": [
{
"name": "Faculty of Medicine, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
},
{
"name": "“Prof. Dr. Matei Balș” National Institute for Infectious Diseases, 1 Calistrat Grozovici Street, 021105 Bucharest, Romania"
}
],
"authenticated-orcid": false,
"family": "Mihai",
"given": "Nicoleta",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Faculty of Dental Medicine, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
},
{
"name": "“Prof. Dr. Matei Balș” National Institute for Infectious Diseases, 1 Calistrat Grozovici Street, 021105 Bucharest, Romania"
}
],
"family": "Tilișcan",
"given": "Cătălin",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-2703-6586",
"affiliation": [
{
"name": "“Ştefan S. Nicolau” Institute of Virology, 285 Mihai Bravu Road, 030304 Bucharest, Romania"
}
],
"authenticated-orcid": false,
"family": "Iancu",
"given": "Iulia Virginia",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Faculty of Medicine, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
},
{
"name": "“Prof. Dr. Matei Balș” National Institute for Infectious Diseases, 1 Calistrat Grozovici Street, 021105 Bucharest, Romania"
}
],
"family": "Ganea",
"given": "Oana-Alexandra",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0009-0000-8520-0375",
"affiliation": [
{
"name": "Faculty of Dental Medicine, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
},
{
"name": "“Prof. Dr. Matei Balș” National Institute for Infectious Diseases, 1 Calistrat Grozovici Street, 021105 Bucharest, Romania"
}
],
"authenticated-orcid": false,
"family": "Adamescu",
"given": "Aida-Isabela",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0003-0019-934X",
"affiliation": [
{
"name": "Faculty of Dental Medicine, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
},
{
"name": "“Prof. Dr. Matei Balș” National Institute for Infectious Diseases, 1 Calistrat Grozovici Street, 021105 Bucharest, Romania"
}
],
"authenticated-orcid": false,
"family": "Aramă",
"given": "Ștefan Sorin",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-9478-6176",
"affiliation": [
{
"name": "Faculty of Pharmacy, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
}
],
"authenticated-orcid": false,
"family": "Arsene",
"given": "Andreea Letiția",
"sequence": "additional"
},
{
"ORCID": "https://orcid.org/0000-0002-2492-6073",
"affiliation": [
{
"name": "Faculty of Medicine, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
},
{
"name": "“Ştefan S. Nicolau” Institute of Virology, 285 Mihai Bravu Road, 030304 Bucharest, Romania"
}
],
"authenticated-orcid": false,
"family": "Ruță",
"given": "Simona-Maria",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Faculty of Medicine, “Carol Davila” University of Medicine and Pharmacy, 37 Dionisie Lupu Street, 020021 Bucharest, Romania"
},
{
"name": "“Prof. Dr. Matei Balș” National Institute for Infectious Diseases, 1 Calistrat Grozovici Street, 021105 Bucharest, Romania"
}
],
"family": "Aramă",
"given": "Victoria",
"sequence": "additional"
}
],
"container-title": "International Journal of Molecular Sciences",
"container-title-short": "IJMS",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2026,
3,
2
]
],
"date-time": "2026-03-02T10:24:34Z",
"timestamp": 1772447074000
},
"deposited": {
"date-parts": [
[
2026,
3,
3
]
],
"date-time": "2026-03-03T12:51:31Z",
"timestamp": 1772542291000
},
"indexed": {
"date-parts": [
[
2026,
3,
3
]
],
"date-time": "2026-03-03T15:52:53Z",
"timestamp": 1772553173414,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issue": "5",
"issued": {
"date-parts": [
[
2026,
2,
28
]
]
},
"journal-issue": {
"issue": "5",
"published-online": {
"date-parts": [
[
2026,
3
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
2,
28
]
],
"date-time": "2026-02-28T00:00:00Z",
"timestamp": 1772236800000
}
}
],
"link": [
{
"URL": "https://www.mdpi.com/1422-0067/27/5/2288/pdf",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "1968",
"original-title": [],
"page": "2288",
"prefix": "10.3390",
"published": {
"date-parts": [
[
2026,
2,
28
]
]
},
"published-online": {
"date-parts": [
[
2026,
2,
28
]
]
},
"publisher": "MDPI AG",
"reference": [
{
"article-title": "MicroRNAs in Drug-induced Liver Injury",
"author": "Li",
"first-page": "162",
"journal-title": "J. Clin. Transl. Hepatol.",
"key": "ref_1",
"volume": "2",
"year": "2014"
},
{
"DOI": "10.1016/j.jhep.2014.10.004",
"article-title": "miR-122—A key factor and therapeutic target in liver disease",
"author": "Bandiera",
"doi-asserted-by": "crossref",
"first-page": "448",
"journal-title": "J. Hepatol.",
"key": "ref_2",
"volume": "62",
"year": "2015"
},
{
"DOI": "10.1371/journal.pone.0197637",
"doi-asserted-by": "crossref",
"key": "ref_3",
"unstructured": "Rahmel, T., Schäfer, S.T., Frey, U.H., Adamzik, M., and Peters, J. (2018). Increased circulating microRNA-122 is a biomarker for discrimination and risk stratification in patients defined by sepsis-3 criteria. PLoS ONE, 13."
},
{
"DOI": "10.1016/S0960-9822(02)00809-6",
"article-title": "Identification of tissue-specific microRNAs from mouse",
"author": "Rauhut",
"doi-asserted-by": "crossref",
"first-page": "735",
"journal-title": "Curr. Biol.",
"key": "ref_4",
"volume": "12",
"year": "2002"
},
{
"DOI": "10.4161/rna.1.2.1066",
"article-title": "miR-122, a mammalian liver-specific microRNA, is processed from hcr mRNA and may downregulate the high affinity cationic amino acid transporter CAT-1",
"author": "Chang",
"doi-asserted-by": "crossref",
"first-page": "106",
"journal-title": "RNA Biol.",
"key": "ref_5",
"volume": "1",
"year": "2004"
},
{
"DOI": "10.1093/cvr/cvab338",
"article-title": "Association of cardiometabolic microRNAs with COVID-19 severity and mortality",
"author": "Gutmann",
"doi-asserted-by": "crossref",
"first-page": "461",
"journal-title": "Cardiovasc. Res.",
"key": "ref_6",
"volume": "118",
"year": "2022"
},
{
"DOI": "10.20944/preprints202312.1840.v2",
"doi-asserted-by": "crossref",
"key": "ref_7",
"unstructured": "Franco, S., Mateu, L., Pluvinet, R., Sanchez-Herrero, J.F., Toledo, R., Sumoy, L., Massanella, M., and Martinez, M.A. (2024). Altered Plasma microRNA Signature in Hospitalized COVID-19 Patients Requiring Oxygen Support. Microorganisms, 12."
},
{
"DOI": "10.1016/j.jhep.2020.04.006",
"article-title": "COVID-19: Abnormal liver function tests",
"author": "Cai",
"doi-asserted-by": "crossref",
"first-page": "566",
"journal-title": "J. Hepatol.",
"key": "ref_8",
"volume": "73",
"year": "2020"
},
{
"DOI": "10.3390/microorganisms10102045",
"doi-asserted-by": "crossref",
"key": "ref_9",
"unstructured": "Mihai, N., Tiliscan, C., Visan, C.A., Stratan, L., Ganea, O., Arama, S.S., Lazar, M., and Arama, V. (2022). Evaluation of Drug-Induced Liver Injury in Hospitalized Patients with SARS-CoV-2 Infection. Microorganisms, 10."
},
{
"DOI": "10.3748/wjg.v29.i2.241",
"article-title": "Liver injury in COVID-19: Clinical features, potential mechanisms, risk factors and clinical treatments",
"author": "Zhao",
"doi-asserted-by": "crossref",
"first-page": "241",
"journal-title": "World J. Gastroenterol.",
"key": "ref_10",
"volume": "29",
"year": "2023"
},
{
"DOI": "10.20944/preprints202409.1173.v1",
"doi-asserted-by": "crossref",
"key": "ref_11",
"unstructured": "Mihai, N., Olariu, M.C., Ganea, O.-A., Adamescu, A.-I., Molagic, V., Aramă, Ș.S., Tilișcan, C., and Aramă, V. (2024). Risk of Hepatitis B Virus Reactivation in COVID-19 Patients Receiving Immunosuppressive Treatment: A Prospective Study. J. Clin. Med., 13."
},
{
"key": "ref_12",
"unstructured": "Food and Drug Administration (FDA), and Center for Drug Evaluation and Research (CDER) (2026, February 13). Summary Review: Remdesivir, Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2020/214787Orig1s000Sumr.pdf."
},
{
"DOI": "10.1038/s41598-025-05541-9",
"article-title": "The impact of remdesivir on renal and liver functions in severe COVID-19 patients with presence of viral load",
"author": "Waldauf",
"doi-asserted-by": "crossref",
"first-page": "20900",
"journal-title": "Sci. Rep.",
"key": "ref_13",
"volume": "15",
"year": "2025"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the treatment of COVID-19—Final report",
"author": "Beigel",
"doi-asserted-by": "crossref",
"first-page": "1813",
"journal-title": "N. Engl. J. Med.",
"key": "ref_14",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.3390/tropicalmed8020129",
"doi-asserted-by": "crossref",
"key": "ref_15",
"unstructured": "Almutairi, A.O., El-Readi, M.Z., Althubiti, M., Alhindi, Y.Z., Ayoub, N., Alzahrani, A.R., Al-Ghamdi, S.S., and Eid, S.Y. (2023). Liver injury in favipiravir-treated COVID-19 patients: Retrospective single-center cohort study. Trop. Med. Infect. Dis., 8."
},
{
"key": "ref_16",
"unstructured": "Food and Drug Administration (FDA) (2026, February 13). Center for Drug Evaluation and Research (CDER). Medical Review: Tocilizumab, Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2010/125276s000MedR.pdf."
},
{
"DOI": "10.1002/jmv.26429",
"article-title": "Late onset infectious complications and safety of tocilizumab in the management of COVID-19",
"author": "Pettit",
"doi-asserted-by": "crossref",
"first-page": "1459",
"journal-title": "J. Med. Virol.",
"key": "ref_17",
"volume": "93",
"year": "2021"
},
{
"key": "ref_18",
"unstructured": "National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) (2026, February 13). Low-Molecular-Weight Heparins, LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, Available online: https://www.ncbi.nlm.nih.gov/books/NBK548009/."
},
{
"DOI": "10.1056/EVIDoa2100043",
"article-title": "Phase 2/3 trial of molnupiravir for treatment of COVID-19 in nonhospitalized adults",
"author": "Caraco",
"doi-asserted-by": "crossref",
"first-page": "EVIDoa2100043",
"journal-title": "NEJM Evid.",
"key": "ref_19",
"volume": "1",
"year": "2022"
},
{
"DOI": "10.1053/j.gastro.2022.09.008",
"article-title": "Minimal risk of drug-induced liver injury with molnupiravir and ritonavir-boosted nirmatrelvir",
"author": "Wong",
"doi-asserted-by": "crossref",
"first-page": "151",
"journal-title": "Gastroenterology",
"key": "ref_20",
"volume": "164",
"year": "2023"
},
{
"key": "ref_21",
"unstructured": "National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) (2026, February 13). Corticosteroids, LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, Available online: https://www.ncbi.nlm.nih.gov/books/NBK548400/."
},
{
"DOI": "10.1016/j.taap.2012.01.009",
"article-title": "Omics analysis of low dose acetaminophen intake demonstrates novel response pathways in humans",
"author": "Jetten",
"doi-asserted-by": "crossref",
"first-page": "320",
"journal-title": "Toxicol. Appl. Pharmacol.",
"key": "ref_22",
"volume": "259",
"year": "2012"
},
{
"DOI": "10.1016/j.jare.2024.12.020",
"article-title": "Advances in statin adverse reactions and the potential mechanisms: A systematic review",
"author": "Zeng",
"doi-asserted-by": "crossref",
"first-page": "781",
"journal-title": "J. Adv. Res.",
"key": "ref_23",
"volume": "76",
"year": "2025"
},
{
"DOI": "10.1530/EJE-14-0867",
"article-title": "Elevated circulating microRNA-122 is associated with obesity and insulin resistance in young adults",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "291",
"journal-title": "Eur. J. Endocrinol.",
"key": "ref_24",
"volume": "172",
"year": "2015"
},
{
"DOI": "10.1002/hep.24538",
"article-title": "Circulating microRNAs as potential markers of human drug-induced liver injury",
"author": "Dear",
"doi-asserted-by": "crossref",
"first-page": "1767",
"journal-title": "Hepatology",
"key": "ref_25",
"volume": "54",
"year": "2011"
},
{
"DOI": "10.3390/microorganisms11112731",
"doi-asserted-by": "crossref",
"key": "ref_26",
"unstructured": "Manea, M., Apostol, D., and Constantinescu, I. (2023). The Connection between MiR-122 and Lymphocytes in Patients Receiving Treatment for Chronic Hepatitis B Virus Infection. Microorganisms, 11."
},
{
"DOI": "10.1186/s12876-019-1048-3",
"doi-asserted-by": "crossref",
"key": "ref_27",
"unstructured": "Shi, L., Zheng, X., Fan, Y., Yang, X., Li, A., and Qian, J. (2019). The contribution of miR-122 to the innate immunity by regulating toll-like receptor 4 in hepatoma cells. BMC Gastroenterol., 19."
},
{
"DOI": "10.1507/endocrj.EJ24-0317",
"article-title": "MicroRNA-122 protects against interferon-α-induced hepatic inflammatory response via the Janus kinase-signal transducer and activator of transcription pathway",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "53",
"journal-title": "Endocr. J.",
"key": "ref_28",
"volume": "72",
"year": "2025"
},
{
"DOI": "10.1038/s42003-024-06288-1",
"article-title": "Downregulation of HNF4A enables transcriptomic reprogramming during the hepatic acute-phase response",
"author": "Ehle",
"doi-asserted-by": "crossref",
"first-page": "589",
"journal-title": "Commun. Biol.",
"key": "ref_29",
"volume": "7",
"year": "2024"
},
{
"DOI": "10.1016/j.imlet.2020.06.015",
"article-title": "Upregulation of RelB in the miR-122 knockout mice contributes to increased levels of proinflammatory chemokines/cytokines in the liver and macrophages",
"author": "Hsu",
"doi-asserted-by": "crossref",
"first-page": "22",
"journal-title": "Immunol. Lett.",
"key": "ref_30",
"volume": "226",
"year": "2020"
},
{
"DOI": "10.1371/journal.pone.0251697",
"doi-asserted-by": "crossref",
"key": "ref_31",
"unstructured": "Zeinali, F., Aghaei Zarch, S.M., Jahan-Mihan, A., Kalantar, S.M., Mehrjardi, M.Y.V., Fallahzadeh, H., Hosseinzadeh, M., Rahmanian, M., and Mozaffari-Khosravi, H. (2021). Circulating microRNA-122, microRNA-126-3p and microRNA-146a are associated with inflammation in patients with pre-diabetes and type 2 diabetes mellitus: A case control study. PLoS ONE, 16."
},
{
"DOI": "10.1186/s12882-025-04619-w",
"doi-asserted-by": "crossref",
"key": "ref_32",
"unstructured": "Osman, E.A., Eltaweel, N.H., Madbouly, M., Mosbah, O., Gaber, M.G., and Khanany, F.M. (2025). Evaluation of serum miR-122-5p, miR-486-5p, miR-21-5p, and their interplay with inflammatory markers in diabetic nephropathy: A case control study. BMC Nephrol., 26."
},
{
"DOI": "10.21203/rs.3.rs-253459/v1",
"doi-asserted-by": "crossref",
"key": "ref_33",
"unstructured": "Farr, R.J., Rootes, C.L., Rowntree, L.C., Nguyen, T.H.O., Hensen, L., Kedzierski, L., Cheng, A.C., Kedzierska, K., Au, G.G., and Marsh, G.A. (2021). Altered microRNA expression in COVID-19 patients enables identification of SARS-CoV-2 infection. PLoS Pathog., 17."
},
{
"DOI": "10.3389/fimmu.2021.815651",
"doi-asserted-by": "crossref",
"key": "ref_34",
"unstructured": "Martínez-Fleta, P., Vera-Tomé, P., Jiménez-Fernández, M., Requena, S., Roy-Vallejo, E., Sanz-García, A., Lozano-Prieto, M., López-Sanz, C., Vara, A., and Lancho-Sánchez, Á. (2022). A Differential Signature of Circulating miRNAs and Cytokines Between COVID-19 and Community-Acquired Pneumonia Uncovers Novel Physiopathological Mechanisms of COVID-19. Front. Immunol., 12."
},
{
"DOI": "10.3390/ncrna7010018",
"doi-asserted-by": "crossref",
"key": "ref_35",
"unstructured": "Alam, T., and Lipovich, L. (2021). miRCOVID-19: Potential Targets of Human miRNAs in SARS-CoV-2 for RNA-Based Drug Discovery. Noncoding RNA, 7."
},
{
"DOI": "10.1080/15476286.2021.2010959",
"article-title": "Cellular miR-150-5p may have a crucial role to play in the biology of SARS-CoV-2 infection by regulating nsp10 gene",
"author": "Akula",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "RNA Biol.",
"key": "ref_36",
"volume": "19",
"year": "2022"
},
{
"DOI": "10.1016/j.xcrm.2021.100287",
"article-title": "Longitudinal proteomic analysis of severe COVID-19 reveals survival-associated signatures, tissue-specific cell death, and cell-cell interactions",
"author": "Filbin",
"doi-asserted-by": "crossref",
"first-page": "100287",
"journal-title": "Cell Rep. Med.",
"key": "ref_37",
"volume": "2",
"year": "2021"
},
{
"DOI": "10.7150/thno.19851",
"article-title": "Incremental Value of Circulating MiR-122-5p to Predict Outcome after Out of Hospital Cardiac Arrest",
"author": "Devaux",
"doi-asserted-by": "crossref",
"first-page": "2555",
"journal-title": "Theranostics",
"key": "ref_38",
"volume": "7",
"year": "2017"
},
{
"DOI": "10.4161/rna.18827",
"article-title": "Liver-specific microRNA-122: Biogenesis and function",
"author": "Jopling",
"doi-asserted-by": "crossref",
"first-page": "137",
"journal-title": "RNA Biol.",
"key": "ref_39",
"volume": "9",
"year": "2012"
},
{
"DOI": "10.1186/s12902-021-00829-z",
"doi-asserted-by": "crossref",
"key": "ref_40",
"unstructured": "Mohany, K.M., Al Rugaie, O., Al-Wutayd, O., and Al-Nafeesah, A. (2021). Investigation of the levels of circulating miR-29a, miR-122, sestrin 2 and inflammatory markers in obese children with/without type 2 diabetes: A case control study. BMC Endocr. Disord., 21."
},
{
"DOI": "10.3390/ncrna11040057",
"doi-asserted-by": "crossref",
"key": "ref_41",
"unstructured": "Garmaa, G., Bunduc, S., Kói, T., Hegyi, P., Csupor, D., Ganbat, D., Dembrovszky, F., Meznerics, F.A., Nasirzadeh, A., and Barbagallo, C. (2024). A Systematic Review and Meta-Analysis of microRNA Profiling Studies in Chronic Kidney Diseases. Non-Coding RNA, 10, Correction in Non-Coding RNA 2025, 11, 57. https://doi.org/10.3390/ncrna11040057."
},
{
"DOI": "10.1093/ndt/gfr485",
"article-title": "Circulating microRNA expression is reduced in chronic kidney disease",
"author": "Neal",
"doi-asserted-by": "crossref",
"first-page": "3794",
"journal-title": "Nephrol. Dial. Transplant.",
"key": "ref_42",
"volume": "26",
"year": "2011"
},
{
"DOI": "10.1111/bcp.13136",
"article-title": "The effect of renal dysfunction and haemodialysis on circulating liver specific miR-122",
"author": "Rivoli",
"doi-asserted-by": "crossref",
"first-page": "584",
"journal-title": "Br. J. Clin. Pharmacol.",
"key": "ref_43",
"volume": "83",
"year": "2017"
},
{
"article-title": "miR-122 inhibits viral replication and cell proliferation in hepatitis B virus-related hepatocellular carcinoma and targets NDRG3",
"author": "Fan",
"first-page": "1281",
"journal-title": "Oncol. Rep.",
"key": "ref_44",
"volume": "26",
"year": "2011"
},
{
"DOI": "10.1186/1471-2490-14-50",
"doi-asserted-by": "crossref",
"key": "ref_45",
"unstructured": "Feng, Y., Kang, Y., He, Y., Liu, J., Liang, B., Yang, P., and Yu, Z. (2014). microRNA-99a acts as a tumor suppressor and is down-regulated in bladder cancer. BMC Urol., 14."
}
],
"reference-count": 45,
"references-count": 45,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.mdpi.com/1422-0067/27/5/2288"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Circulating Liver-Enriched miR-122 in COVID-19 Patients: A Longitudinal Real-Life Study",
"type": "journal-article",
"update-policy": "https://doi.org/10.3390/mdpi_crossmark_policy",
"volume": "27"
}