Randomized controlled trial of the effect of regular paracetamol on influenza infection
et al., Respirology, doi:10.1111/resp.12685, ACTRN12611000497909, Dec 2015
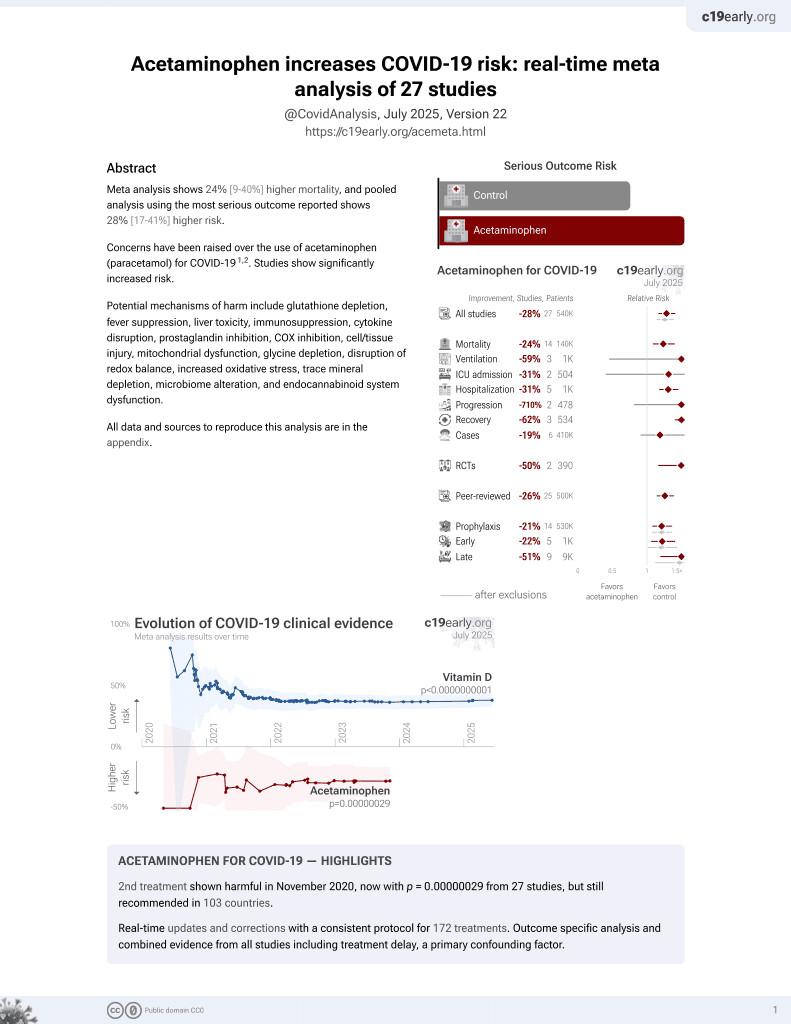
2nd treatment shown to increase risk in
November 2020, now with p = 0.00000029 from 27 studies, but still recommended in 103 countries.
6,600+ studies for
220+ treatments. c19early.org
|
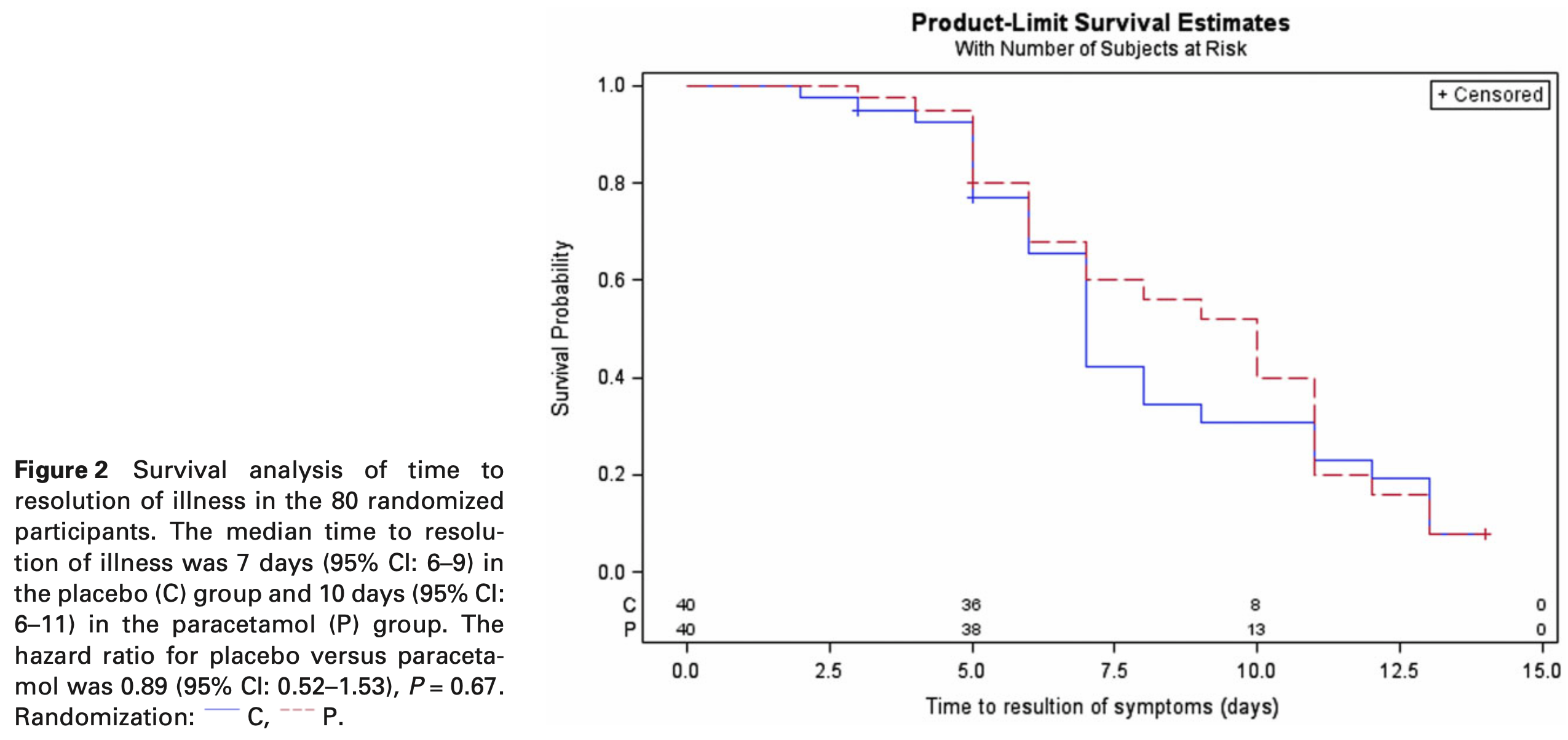
RCT 80 outpatients with influenza-like illness showing no significant differences with acetaminophen versus placebo. There was only 24 and 22 PCR-confirmed patients. While not significant with the small sample, risk was higher with treatment for recovery time (10 vs. 7 days), positive viral culture @ 5 days (2 vs. 0), detectable viral load @ 5 days (71% vs. 51%), and viral load AUC (4.65 vs. 4.4). Authors incorrectly state "paracetamol had no effect".
Acetaminophen is also known as paracetamol, Tylenol, Panadol, Calpol, Tempra, Calprofen, Doliprane, Efferalgan, Grippostad C, Dolo, Acamol, Fevadol, Crocin, and Perfalgan.
Jefferies et al., 6 Dec 2015, Double Blind Randomized Controlled Trial, placebo-controlled, USA, peer-reviewed, 12 authors, trial ACTRN12611000497909.
Contact: irene.braithwaite@mrinz.ac.nz.
Abstract:
Editor's Choice
ORIGINAL ARTICLE
Randomized controlled trial of the effect of regular paracetamol on influenza infection
SARAH JEFFERIES, 1 IRENE BRAITHWAITE, 1 STEVEN WALKER, 1 MARK WEATHERALL, 2,3 LANCE JENNINGS, 4 MICHELLE LUCK, 4 KEVIN BARRETT, 4 ROBERT SIEBERS, 2 TIMOTHY BLACKMORE, 3 RICHARD BEASLEY 1,3 AND KYLE PERRIN 1,2,3 On behalf of the Pi Study Group*
1 Medical Research Institute of New Zealand, 2 Department of Medicine, University of Otago Wellington, 3 Capital & Coast District Health Board, Wellington, and 4 Canterbury Health Laboratories, Canterbury District Health Board, Christchurch, New Zealand
ABSTRACT
Background and objective: Anti-pyretic treatment is recommended in the management of influenza infection. In animal models anti-pyretic treatment increases mortality from influenza.We investigated the effects of paracetamol on viral and clinical outcomes in adults with influenza infection.
Methods: This is a randomized, double-blind, placebo-controlled trial of adults aged 18-65 years with influenza-like illness and positive influenza rapid antigen test. Treatments were 1 g paracetamol four times a day, or matching placebo, for 5 days. Pernasal swabs were taken for influenza quantitative RT-PCR at Baseline and Days 1, 2 and 5. Temperature and symptom scores were recorded for 5-14 days or time of resolution respectively. The primary outcome variable was area under the curve (AUC) for quantitative PCR log10 viral load from Baseline to Day 5.
Correspondence: Irene Braithwaite, Medical Research Institute of New Zealand, Private Bag 7902, Newtown, Wellington 6242, New Zealand. Email: irene.braithwaite@mrinz.ac.nz
Disclosure statement: RB has been a member of the GlaxoSmithKline (GSK) New Zealand advisory board and has received research grants, payments for lectures and/or attended meetings from GSK, a manufacturer of paracetamol. LJ has received unrestricted research funding from F. Hoffman-La Roche, and honoraria and travel assistance from F. Hoffman-La Roche, GlaxoSmithKline and Sanofi Pasteur for participating on advisory groups or in scientific meetings.
*Pi Study Group: Medical Research Institute of New Zealand: Irene Braithwaite, Richard Beasley, James Fingleton, Sarah Jefferies, Mark Holliday, Claire Munro, Mitesh Patel, Kyle Perrin, Janine Pilcher, Alison Pritchard and Steve Walker; Capital & Coast District Health Board: Jonathon Barrett, Tim Blackmore, Marina Dzhelali, Leeanne Olsen, Serena Rooker and Helen White; University of Otago, Wellington: Rob Siebers and Mark Weatherall; Canterbury Health Laboratories: Kevin Barratt, Lance Jennings and Michelle Luck.
Received 8 April 2015; invited to revise 21 May and 3 July 2015; revised 26 May and 14 July 2015; accepted 31 August 2015 (Associate Editor: Marcos Restrepo).
This is an open access article under the terms of the Creative Commons Attribution-NonCommercial License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited and is not used for commercial purposes.
Article first published online: 6 December 2015
SUMMARY AT A GLANCE
Despite recommendations to administer paracetamol for symptom relief in influenza and influenzalike-illnesses, this study has found that regular administration of paracetamol has no effect on viral or clinical outcomes in this setting.
Results: A total of 80 participants were randomized: no one was lost to follow up, and one withdrew after 4 days.There were 22 and 24..
DOI record:
{
"DOI": "10.1111/resp.12685",
"ISSN": [
"1323-7799",
"1440-1843"
],
"URL": "http://dx.doi.org/10.1111/resp.12685",
"abstract": "<jats:title>Abstract</jats:title><jats:sec><jats:title>Background and objective</jats:title><jats:p>Anti‐pyretic treatment is recommended in the management of influenza infection. In animal models anti‐pyretic treatment increases mortality from influenza. We investigated the effects of paracetamol on viral and clinical outcomes in adults with influenza infection.</jats:p></jats:sec><jats:sec><jats:title>Methods</jats:title><jats:p>This is a randomized, double‐blind, placebo‐controlled trial of adults aged 18–65 years with influenza‐like illness and positive influenza rapid antigen test. Treatments were 1 g paracetamol four times a day, or matching placebo, for 5 days. Pernasal swabs were taken for influenza quantitative <jats:styled-content style=\"fixed-case\">RT‐PCR</jats:styled-content> at <jats:styled-content style=\"fixed-case\">B</jats:styled-content>aseline and <jats:styled-content style=\"fixed-case\">D</jats:styled-content>ays 1, 2 and 5. Temperature and symptom scores were recorded for 5–14 days or time of resolution respectively. The primary outcome variable was area under the curve (<jats:styled-content style=\"fixed-case\">AUC</jats:styled-content>) for quantitative <jats:styled-content style=\"fixed-case\">PCR</jats:styled-content> log<jats:sub>10</jats:sub> viral load from <jats:styled-content style=\"fixed-case\">B</jats:styled-content>aseline to <jats:styled-content style=\"fixed-case\">D</jats:styled-content>ay 5.</jats:p></jats:sec><jats:sec><jats:title>Results</jats:title><jats:p>A total of 80 participants were randomized: no one was lost to follow up, and one withdrew after 4 days. There were 22 and 24 participants who were influenza <jats:styled-content style=\"fixed-case\">PCR</jats:styled-content>‐positive in placebo and in paracetamol groups respectively. Mean (<jats:styled-content style=\"fixed-case\">SD</jats:styled-content>) <jats:styled-content style=\"fixed-case\">AUC PCR</jats:styled-content> log<jats:sub>10</jats:sub> viral load was 4.40 (0.91) in placebo and 4.64 (0.88) in paracetamol; difference was −0.24, 95% <jats:styled-content style=\"fixed-case\">CI</jats:styled-content>: −0.78 to 0.29, <jats:italic>P</jats:italic> = 0.36. In all participants there were no differences in symptom scores, temperature, time to resolution of illness and health status, with no interaction between randomized treatment and whether influenza was detected by <jats:styled-content style=\"fixed-case\">PCR</jats:styled-content>.</jats:p></jats:sec><jats:sec><jats:title>Conclusion</jats:title><jats:p>Regular paracetamol had no effect on viral shedding, temperature or clinical symptoms in patients with <jats:styled-content style=\"fixed-case\">PCR</jats:styled-content>‐confirmed influenza. There remains an insufficient evidence base for paracetamol use in influenza infection.</jats:p><jats:p>Clinical trial registration: <jats:bold><jats:styled-content style=\"fixed-case\">ACTRN</jats:styled-content>12611000497909 at the <jats:styled-content style=\"fixed-case\">A</jats:styled-content>ustralian <jats:styled-content style=\"fixed-case\">N</jats:styled-content>ew <jats:styled-content style=\"fixed-case\">Z</jats:styled-content>ealand <jats:styled-content style=\"fixed-case\">C</jats:styled-content>linical <jats:styled-content style=\"fixed-case\">T</jats:styled-content>rials <jats:styled-content style=\"fixed-case\">R</jats:styled-content>egistry.</jats:bold></jats:p></jats:sec>",
"alternative-id": [
"10.1111/resp.12685"
],
"author": [
{
"affiliation": [
{
"name": "Medical Research Institute of New Zealand New Zealand Wellington"
}
],
"family": "Jefferies",
"given": "Sarah",
"sequence": "first"
},
{
"ORCID": "https://orcid.org/0000-0001-5327-3027",
"affiliation": [
{
"name": "Medical Research Institute of New Zealand New Zealand Wellington"
}
],
"authenticated-orcid": false,
"family": "Braithwaite",
"given": "Irene",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Research Institute of New Zealand New Zealand Wellington"
}
],
"family": "Walker",
"given": "Steven",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine University of Otago Wellington New Zealand Wellington"
},
{
"name": "Capital & Coast District Health Board New Zealand Wellington"
}
],
"family": "Weatherall",
"given": "Mark",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Canterbury Health Laboratories Canterbury District Health Board Christchurch New Zealand"
}
],
"family": "Jennings",
"given": "Lance",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Canterbury Health Laboratories Canterbury District Health Board Christchurch New Zealand"
}
],
"family": "Luck",
"given": "Michelle",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Canterbury Health Laboratories Canterbury District Health Board Christchurch New Zealand"
}
],
"family": "Barrett",
"given": "Kevin",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Department of Medicine University of Otago Wellington New Zealand Wellington"
}
],
"family": "Siebers",
"given": "Robert",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Capital & Coast District Health Board New Zealand Wellington"
}
],
"family": "Blackmore",
"given": "Timothy",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Research Institute of New Zealand New Zealand Wellington"
},
{
"name": "Capital & Coast District Health Board New Zealand Wellington"
}
],
"family": "Beasley",
"given": "Richard",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Medical Research Institute of New Zealand New Zealand Wellington"
},
{
"name": "Department of Medicine University of Otago Wellington New Zealand Wellington"
},
{
"name": "Capital & Coast District Health Board New Zealand Wellington"
}
],
"family": "Perrin",
"given": "Kyle",
"sequence": "additional"
},
{
"affiliation": [],
"name": "On behalf of the Pi Study Group",
"sequence": "additional"
}
],
"container-title": "Respirology",
"container-title-short": "Respirology",
"content-domain": {
"crossmark-restriction": false,
"domain": []
},
"created": {
"date-parts": [
[
2015,
12,
7
]
],
"date-time": "2015-12-07T00:57:59Z",
"timestamp": 1449449879000
},
"deposited": {
"date-parts": [
[
2023,
9,
7
]
],
"date-time": "2023-09-07T01:46:54Z",
"timestamp": 1694051214000
},
"funder": [
{
"DOI": "10.13039/501100001505",
"doi-asserted-by": "crossref",
"id": [
{
"asserted-by": "crossref",
"id": "10.13039/501100001505",
"id-type": "DOI"
}
],
"name": "Health Research Council of New Zealand"
}
],
"indexed": {
"date-parts": [
[
2026,
2,
11
]
],
"date-time": "2026-02-11T03:55:13Z",
"timestamp": 1770782113916,
"version": "3.50.0"
},
"is-referenced-by-count": 18,
"issue": "2",
"issued": {
"date-parts": [
[
2015,
12,
6
]
]
},
"journal-issue": {
"issue": "2",
"published-print": {
"date-parts": [
[
2016,
2
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by-nc/3.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2015,
12,
6
]
],
"date-time": "2015-12-06T00:00:00Z",
"timestamp": 1449360000000
}
}
],
"link": [
{
"URL": "https://api.wiley.com/onlinelibrary/tdm/v1/articles/10.1111%2Fresp.12685",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.wiley.com/onlinelibrary/tdm/v1/articles/10.1111%2Fresp.12685",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1111/resp.12685",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/full-xml/10.1111/resp.12685",
"content-type": "application/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://onlinelibrary.wiley.com/doi/pdf/10.1111/resp.12685",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "311",
"original-title": [],
"page": "370-377",
"prefix": "10.1111",
"published": {
"date-parts": [
[
2015,
12,
6
]
]
},
"published-online": {
"date-parts": [
[
2015,
12,
6
]
]
},
"published-print": {
"date-parts": [
[
2016,
2
]
]
},
"publisher": "Wiley",
"reference": [
{
"DOI": "10.1016/j.vaccine.2007.03.046",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_2_1"
},
{
"DOI": "10.2105/AJPH.87.12.1944",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_3_1"
},
{
"DOI": "10.1016/S0140-6736(09)61131-4",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_4_1"
},
{
"DOI": "10.1136/thx.2006.073080",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_5_1"
},
{
"DOI": "10.1016/S0891-5520(05)70282-8",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_6_1"
},
{
"DOI": "10.1093/infdis/155.5.991",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_7_1"
},
{
"DOI": "10.1016/0361‐9230(80)90285‐3",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_8_1"
},
{
"DOI": "10.1016/0165-2427(85)90004-2",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_9_1"
},
{
"DOI": "10.1258/jrsm.2010.090441",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_10_1"
},
{
"DOI": "10.1016/S0022‐3476(89)80461‐5",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_11_1"
},
{
"DOI": "10.1016/S0140‐6736(97)02255‐1",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_12_1"
},
{
"DOI": "10.1093/infdis/162.6.1277",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_13_1"
},
{
"DOI": "10.1093/clinids/13.3.462",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_14_1"
},
{
"DOI": "10.1128/JVI.52.3.928-931.1984",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_15_1"
},
{
"DOI": "10.4049/jimmunol.133.4.2037",
"article-title": "The effect of febrile temperatures on biologic actions of interferons: abrogation of suppression of delayed‐type hypersensitivity and antibody production",
"author": "Ron Y",
"doi-asserted-by": "crossref",
"first-page": "2037",
"journal-title": "J. Immunol.",
"key": "e_1_2_7_16_1",
"volume": "133",
"year": "1984"
},
{
"DOI": "10.1016/0162‐3109(92)90068‐N",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_17_1"
},
{
"DOI": "10.1016/0006‐2952(89)90163‐9",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_18_1"
},
{
"DOI": "10.1002/(SICI)1099‐081X(199608)17:6<501::AID‐BDD968>3.0.CO;2‐1",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_19_1"
},
{
"DOI": "10.1016/S0140‐6736(09)61208‐3",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_20_1"
},
{
"DOI": "10.1186/1743‐422X‐3‐58",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_21_1"
},
{
"DOI": "10.1016/0168‐702(84)90056‐X",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_22_1"
},
{
"DOI": "10.1128/JVI.41.2.353-359.1982",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_23_1"
},
{
"key": "e_1_2_7_24_1",
"unstructured": "REMEL.X/pect™ FLU A&B Data Sheet IFU24600 revised August2003."
},
{
"DOI": "10.1016/j.jviromet.2013.04.005",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_25_1"
},
{
"DOI": "10.1086/600383",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_26_1"
},
{
"DOI": "10.1001/jama.1996.03530280047035",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_27_1"
},
{
"DOI": "10.1016/S0140‐6736(00)02288‐1",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_28_1"
},
{
"DOI": "10.1086/598513",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_29_1"
},
{
"DOI": "10.1002/14651858.CD008965.pub3",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_30_1"
},
{
"article-title": "A double‐blind comparison of fenoprofen calcium, acetaminophen, and placebo in the palliative treatment of common nonbacterial upper respiratory infections",
"author": "Ryan PB",
"first-page": "17",
"journal-title": "Curr. Ther. Res. Clin. Exp.",
"key": "e_1_2_7_31_1",
"volume": "41",
"year": "1987"
},
{
"DOI": "10.1016/j.clinthera.2005.06.002",
"doi-asserted-by": "publisher",
"key": "e_1_2_7_32_1"
}
],
"reference-count": 31,
"references-count": 31,
"relation": {},
"resource": {
"primary": {
"URL": "https://onlinelibrary.wiley.com/doi/10.1111/resp.12685"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Randomized controlled trial of the effect of regular paracetamol on influenza infection",
"type": "journal-article",
"volume": "21"
}