Anti-COVID-19 activity and simple HPLC method for concurrent detection of repurposed drugs in novel binary mixtures
et al., AMB Express, doi:10.1186/s13568-026-02030-8, Apr 2026
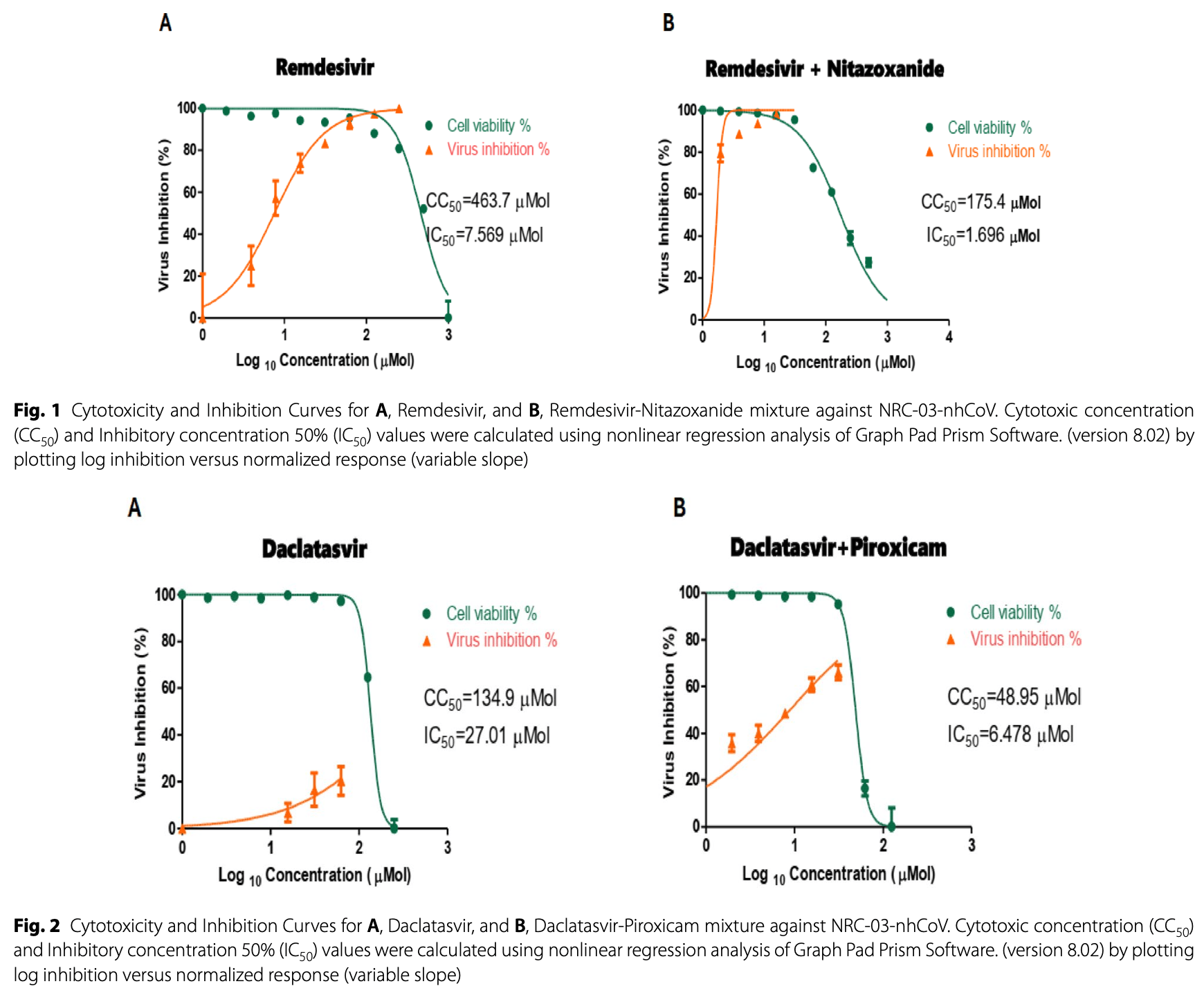
In vitro study showing enhanced anti-SARS-CoV-2 activity of remdesivir-nitazoxanide and daclatasvir-piroxicam mixtures in Vero-E6 cells.
Abd Elsalam et al., 9 Apr 2026, Egypt, peer-reviewed, 9 authors.
Contact: sarah.shabayek@pharm.suez.edu.eg.
In vitro studies are an important part of preclinical research, however results may be very different in vivo.
Abstract: ## RESEARCH
Open Access
Anti-COVID-19 activity and simple HPLC method for concurrent detection of repurposed drugs in novel binary mixtures
Mohamed A. Abd elsalam 1 , Omnia Kutkat 2 , Yassmin Moatasim 2 , Ahmed Mostafa 2 , Mohamed Gaballah 2 , Alaa-eldin Elgendy 1 , Mohamed A Ali 2 , Ghada Hadad 1 and Sarah Shabayek 3*
Abstract
The discovery of a new antiviral drug specifically targeting SARS-CoV-2 would require considerable time and effort. Repositioning drugs that are already used for other viral infections is a valuable approach. Remdesivir is the first antiviral treatment approved by the Food and Drug Administration (FDA) for COVID-19. Nitazoxanide is a broadspectrum anti-infective agent that has been suggested for repurposing in COVID-19 treatment. Recent research has demonstrated the synergistic anti-SARS-CoV-2 antiviral activity of Remdesivir when combined with Nitazoxanide. Moreover, Daclatasvir is an FDA-approved antiviral for managing chronic HCV infection, and its potential use in treating COVID-19 has been reported. Piroxicam is an FDA-approved anti-inflammatory with some clinical evidence supporting its application for COVID-19. There is no evidence for exploiting the anti-SARS-CoV-2 activity of Daclatasvir when combined with Piroxicam. We were able to demonstrate the enhanced anti-SARS-CoV-2 activity of the Remdesivir-Nitazoxanide and Daclatasvir-Piroxicam binary mixtures in Vero-E6 cells. In addition, we were able to develop a simple HPLC methodology for the concurrent determination of the two analytes of each binary mixture (Remdesivir-Nitazoxanide mixture and Daclatasvir-Piroxicam mixture) in both pure form and in human plasma using non-complicated, widely available analytical instruments. The proposed method is simple, accurate, quick, and sensitive. No drug interactions were observed in the tested binary mixtures.
Keywords COVID-19, Antiviral, HPLC, Remdesivir, Nitazoxanide, Daclatasvir, Piroxicam
DOI record:
{
"DOI": "10.1186/s13568-026-02030-8",
"ISSN": [
"2191-0855"
],
"URL": "http://dx.doi.org/10.1186/s13568-026-02030-8",
"alternative-id": [
"2030"
],
"article-number": "47",
"assertion": [
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Received",
"name": "received",
"order": 1,
"value": "5 December 2025"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "13 February 2026"
},
{
"group": {
"label": "Article History",
"name": "ArticleHistory"
},
"label": "First Online",
"name": "first_online",
"order": 3,
"value": "9 April 2026"
},
{
"group": {
"label": "Declarations",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 1
},
{
"group": {
"label": "Ethics approval and consent to participate",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 2,
"value": "All experiments were performed in accordance with relevant guidelines and regulations. This study was approved by the scientific research ethics committee of the Faculty of Pharmacy, Suez Canal University, Ismailia, Egypt (Reference number 202010M2)."
},
{
"group": {
"label": "Competing interests",
"name": "EthicsHeading"
},
"name": "Ethics",
"order": 3,
"value": "The authors declare no competing interests."
}
],
"author": [
{
"affiliation": [],
"family": "Abd elsalam",
"given": "Mohamed A.",
"sequence": "first"
},
{
"affiliation": [],
"family": "Kutkat",
"given": "Omnia",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Moatasim",
"given": "Yassmin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Mostafa",
"given": "Ahmed",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gaballah",
"given": "Mohamed",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Elgendy",
"given": "Alaa-eldin",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Ali",
"given": "Mohamed A",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Hadad",
"given": "Ghada",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Shabayek",
"given": "Sarah",
"sequence": "additional"
}
],
"container-title": "AMB Express",
"container-title-short": "AMB Expr",
"content-domain": {
"crossmark-restriction": false,
"domain": [
"link.springer.com"
]
},
"created": {
"date-parts": [
[
2026,
4,
9
]
],
"date-time": "2026-04-09T15:55:15Z",
"timestamp": 1775750115000
},
"deposited": {
"date-parts": [
[
2026,
4,
16
]
],
"date-time": "2026-04-16T04:35:13Z",
"timestamp": 1776314113000
},
"funder": [
{
"DOI": "10.13039/501100024743",
"doi-asserted-by": "crossref",
"id": [
{
"asserted-by": "crossref",
"id": "10.13039/501100024743",
"id-type": "DOI"
}
],
"name": "Suez Canal University"
}
],
"indexed": {
"date-parts": [
[
2026,
4,
16
]
],
"date-time": "2026-04-16T05:00:01Z",
"timestamp": 1776315601829,
"version": "3.50.1"
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2026,
4,
9
]
]
},
"journal-issue": {
"issue": "1",
"published-online": {
"date-parts": [
[
2026,
12
]
]
}
},
"language": "en",
"license": [
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2026,
4,
9
]
],
"date-time": "2026-04-09T00:00:00Z",
"timestamp": 1775692800000
}
},
{
"URL": "https://creativecommons.org/licenses/by/4.0",
"content-version": "vor",
"delay-in-days": 7,
"start": {
"date-parts": [
[
2026,
4,
16
]
],
"date-time": "2026-04-16T00:00:00Z",
"timestamp": 1776297600000
}
}
],
"link": [
{
"URL": "https://link.springer.com/article/10.1186/s13568-026-02030-8",
"content-type": "text/html",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1186/s13568-026-02030-8.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://link.springer.com/content/pdf/10.1186/s13568-026-02030-8.pdf",
"content-type": "application/pdf",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "297",
"original-title": [],
"prefix": "10.1186",
"published": {
"date-parts": [
[
2026,
4,
9
]
]
},
"published-online": {
"date-parts": [
[
2026,
4,
9
]
]
},
"publisher": "Springer Science and Business Media LLC",
"reference": [
{
"DOI": "10.1186/s13065-024-01364-3",
"author": "AB Ahmed",
"doi-asserted-by": "publisher",
"first-page": "11",
"issue": "1",
"journal-title": "BMC Chem",
"key": "2030_CR1",
"unstructured": "Ahmed AB, Abdelrahman MM, Edrees FH (2025a) Eco-sustainable chromatographic method for the determination of favipiravir and nitazoxanide for COVID-19: application to human plasma. BMC Chem 19(1):11. https://doi.org/10.1186/s13065-024-01364-3",
"volume": "19",
"year": "2025"
},
{
"DOI": "10.1186/s13065-024-01364-3",
"author": "AB Ahmed",
"doi-asserted-by": "publisher",
"first-page": "11",
"issue": "1",
"journal-title": "BMC Chem",
"key": "2030_CR2",
"unstructured": "Ahmed AB, Abdelrahman MM, Edrees FH (2025b) Eco-sustainable chromatographic method for the determination of favipiravir and nitazoxanide for COVID-19: application to human plasma. BMC Chem 19(1):11. https://doi.org/10.1186/s13065-024-01364-3",
"volume": "19",
"year": "2025"
},
{
"DOI": "10.1002/bio.4099",
"author": "A Almahri",
"doi-asserted-by": "publisher",
"first-page": "1544",
"issue": "6",
"journal-title": "Luminescence",
"key": "2030_CR3",
"unstructured": "Almahri A, Abdel-Lateef MA (2021) Applying different spectroscopic techniques for the selective determination of daclatasvir using merbromin as a probe: applications on pharmaceutical analysis. Luminescence 36(6):1544–1552. https://doi.org/10.1002/bio.4099",
"volume": "36",
"year": "2021"
},
{
"DOI": "10.1093/jac/dkaa152",
"author": "V Avataneo",
"doi-asserted-by": "publisher",
"first-page": "1772",
"issue": "7",
"journal-title": "J Antimicrob Chemother",
"key": "2030_CR4",
"unstructured": "Avataneo V, de Nicolò A, Cusato J, Antonucci M, Manca A, Palermiti A, Waitt C, Walimbwa S, Lamorde M, di Perri G, D’Avolio A (2020) Development and validation of a UHPLC-MS/MS method for quantification of the prodrug remdesivir and its metabolite GS-441524: a tool for clinical pharmacokinetics of SARS-CoV-2/COVID-19 and Ebola virus disease. J Antimicrob Chemother 75(7):1772–1777. https://doi.org/10.1093/jac/dkaa152",
"volume": "75",
"year": "2020"
},
{
"DOI": "10.1016/bs.podrm.2022.11.003",
"author": "AH Bakheit",
"doi-asserted-by": "publisher",
"first-page": "71",
"journal-title": "Profiles Drug Subst Excip Relat Methodol",
"key": "2030_CR5",
"unstructured": "Bakheit AH, Darwish H, Darwish IA, Al-Ghusn AI (2023) Remdesivir. Profiles Drug Subst Excip Relat Methodol 48:71–108. https://doi.org/10.1016/bs.podrm.2022.11.003",
"volume": "48",
"year": "2023"
},
{
"DOI": "10.3390/vaccines11030515",
"doi-asserted-by": "publisher",
"key": "2030_CR6",
"unstructured": "Bansode S, Singh PK, Tellis M, Chugh A, Deshmukh N, Gupta M, Verma S, Giri A, Kulkarni M, Joshi R, Chaudhary D (2023) A comprehensive molecular and clinical investigation of approved Anti-HCV drugs repurposing against SARS-CoV-2 infection: a glaring gap between benchside and bedside medicine. Vaccines (Basel) 11(3). https://doi.org/10.3390/vaccines11030515"
},
{
"DOI": "10.1007/s10096-020-04138-6",
"author": "S Beyerstedt",
"doi-asserted-by": "publisher",
"first-page": "905",
"issue": "5",
"journal-title": "Eur J Clin Microbiol Infect Dis",
"key": "2030_CR7",
"unstructured": "Beyerstedt S, Casaro EB, Rangel É, B (2021) COVID-19: angiotensin-converting enzyme 2 (ACE2) expression and tissue susceptibility to SARS-CoV-2 infection. Eur J Clin Microbiol Infect Dis 40(5):905–919. https://doi.org/10.1007/s10096-020-04138-6",
"volume": "40",
"year": "2021"
},
{
"DOI": "10.1016/j.ymthe.2020.12.016",
"author": "T Bobrowski",
"doi-asserted-by": "publisher",
"first-page": "873",
"issue": "2",
"journal-title": "Mol Ther",
"key": "2030_CR8",
"unstructured": "Bobrowski T, Chen L, Eastman RT, Itkin Z, Shinn P, Chen CZ, Guo H, Zheng W, Michael S, Simeonov A, Hall MD, Zakharov AV, Muratov EN (2021) Synergistic and antagonistic drug combinations against SARS-CoV-2. Mol Ther 29(2):873–885. https://doi.org/10.1016/j.ymthe.2020.12.016",
"volume": "29",
"year": "2021"
},
{
"DOI": "10.3897/pharmacia.71.e120463",
"author": "S Bozhanov",
"doi-asserted-by": "publisher",
"first-page": "1",
"journal-title": "Pharmacia",
"key": "2030_CR9",
"unstructured": "Bozhanov S, Smerikarova M, Maslarska V (2024) Simultaneous HPLC determination of remdesivir and dexamethasone in the presence of metformin, sitagliptin, and glimepiride in a synthetic mixture and spiked human plasma. Pharmacia 71:1–10",
"volume": "71",
"year": "2024"
},
{
"DOI": "10.1016/j.jpba.2015.12.042",
"author": "AM Calvo",
"doi-asserted-by": "publisher",
"first-page": "212",
"journal-title": "J Pharm Biomed Anal",
"key": "2030_CR10",
"unstructured": "Calvo AM, Santos GM, Dionísio TJ, Marques MP, Brozoski DT, Lanchote VL, Fernandes MH, Faria FA, Santos CF (2016) Quantification of piroxicam and 5’-hydroxypiroxicam in human plasma and saliva using liquid chromatography-tandem mass spectrometry following oral administration. J Pharm Biomed Anal 120:212–220. https://doi.org/10.1016/j.jpba.2015.12.042",
"volume": "120",
"year": "2016"
},
{
"DOI": "10.1093/chromsci/bmae021",
"doi-asserted-by": "publisher",
"key": "2030_CR11",
"unstructured": "Chauhan V, Grover P, Bhardwaj M, Kumar S, Nagarajan K (2025) Development and validation of fast and sensitive RP-HPLC stability-indicating method for quantification of piroxicam in bulk drug. J Chromatogr Sci 63(1). https://doi.org/10.1093/chromsci/bmae021"
},
{
"DOI": "10.1002/j.1875-9114.1982.tb03178.x",
"author": "SL Dahl",
"doi-asserted-by": "publisher",
"first-page": "80",
"issue": "2",
"journal-title": "Pharmacotherapy",
"key": "2030_CR12",
"unstructured": "Dahl SL, Ward JR (1982) Pharmacology, clinical efficacy, and adverse effects of piroxicam, a new nonsteroidal anti-inflammatory agent. Pharmacotherapy 2(2):80–90. https://doi.org/10.1002/j.1875-9114.1982.tb03178.x",
"volume": "2",
"year": "1982"
},
{
"DOI": "10.1007/s11224-022-01975-3",
"author": "P De",
"doi-asserted-by": "publisher",
"first-page": "1741",
"issue": "5",
"journal-title": "Struct Chem",
"key": "2030_CR13",
"unstructured": "De P, Kumar V, Kar S, Roy K, Leszczynski J (2022) Repurposing FDA approved drugs as possible anti-SARS-CoV-2 medications using ligand-based computational approaches: sum of ranking difference-based model selection. Struct Chem 33(5):1741–1753. https://doi.org/10.1007/s11224-022-01975-3",
"volume": "33",
"year": "2022"
},
{
"DOI": "10.1086/653080",
"author": "S Einav",
"doi-asserted-by": "publisher",
"first-page": "65",
"issue": "1",
"journal-title": "J Infect Dis",
"key": "2030_CR14",
"unstructured": "Einav S, Sobol HD, Gehrig E, Glenn JS (2010) The hepatitis C virus (HCV) NS4B RNA binding inhibitor clemizole is highly synergistic with HCV protease inhibitors. J Infect Dis 202(1):65–74. https://doi.org/10.1086/653080",
"volume": "202",
"year": "2010"
},
{
"DOI": "10.1038/s41598-024-71413-3",
"author": "HI El-Shorbagy",
"doi-asserted-by": "publisher",
"first-page": "21758",
"issue": "1",
"journal-title": "Sci Rep",
"key": "2030_CR15",
"unstructured": "El-Shorbagy HI, Mohamed MA, El-Gindy A, Hadad GM, Belal F (2024) UPLC-PDA factorial design assisted method for simultaneous determination of oseltamivir, dexamethasone, and remdesivir in human plasma. Sci Rep 14(1):21758. https://doi.org/10.1038/s41598-024-71413-3",
"volume": "14",
"year": "2024"
},
{
"DOI": "10.1111/jvh.70032",
"author": "T Elbaz",
"doi-asserted-by": "publisher",
"first-page": "e70032",
"issue": "7",
"journal-title": "J Viral Hepat",
"key": "2030_CR16",
"unstructured": "Elbaz T, Al-Naamani K, Abosheaishaa H, Alswat K, El-Kassas M (2025) Leading role of Sofosbuvir/Daclatasvir in achieving Hepatitis C elimination in Egypt. J Viral Hepat 32(7):e70032. https://doi.org/10.1111/jvh.70032",
"volume": "32",
"year": "2025"
},
{
"DOI": "10.1093/chromsci/bmab100",
"doi-asserted-by": "publisher",
"key": "2030_CR17",
"unstructured": "Fayed AS, Hegazy MA, Kamel EB, Eissa MS (2021) HPLC-UV and TLC-densitometry methods for simultaneous determination of Sofosbuvir and Daclatasvir: Application to Darvoni® Tablet. Journal of Chromatographic Science 60(6), 606–612. https://doi.org/10.1093/chromsci/bmab100"
},
{
"DOI": "10.1093/chromsci/bmab100",
"author": "AS Fayed",
"doi-asserted-by": "publisher",
"first-page": "606",
"issue": "6",
"journal-title": "J Chromatogr Sci",
"key": "2030_CR18",
"unstructured": "Fayed AS, Hegazy MA, Kamel EB, Eissa MS (2022) HPLC-UV and TLC-densitometry methods for simultaneous determination of Sofosbuvir and Daclatasvir: application to Darvoni® Tablet. J Chromatogr Sci 60(6):606–612. https://doi.org/10.1093/chromsci/bmab100",
"volume": "60",
"year": "2022"
},
{
"DOI": "10.1007/s12325-016-0407-5",
"author": "T Garimella",
"doi-asserted-by": "publisher",
"first-page": "1867",
"issue": "11",
"journal-title": "Adv Ther",
"key": "2030_CR19",
"unstructured": "Garimella T, You X, Wang R, Huang SP, Kandoussi H, Bifano M, Bertz R, Eley T (2016) A review of daclatasvir drug-drug interactions. Adv Ther 33(11):1867–1884. https://doi.org/10.1007/s12325-016-0407-5",
"volume": "33",
"year": "2016"
},
{
"DOI": "10.1111/nep.13222",
"author": "A Goel",
"doi-asserted-by": "publisher",
"first-page": "316",
"issue": "3",
"journal-title": "Nephrol (Carlton)",
"key": "2030_CR20",
"unstructured": "Goel A, Bhadauria DS, Kaul A, Verma P, Mehrotra M, Gupta A, Sharma RK, Rai P, Aggarwal R (2019) Daclatasvir and reduced-dose sofosbuvir: an effective and pangenotypic treatment for hepatitis C in patients with estimated glomerular filtration rate < 30 mL/min. Nephrol (Carlton) 24(3):316–321. https://doi.org/10.1111/nep.13222",
"volume": "24",
"year": "2019"
},
{
"key": "2030_CR21",
"unstructured": "Guideline IJIG (2022a) Switzerland Validation of analytical procedures Q2 (R2). 1"
},
{
"key": "2030_CR22",
"unstructured": "Guideline I (2022b) Switzerland Bioanalytical method validation and study sample analysis M10"
},
{
"DOI": "10.1016/j.jpba.2021.113935",
"author": "K Habler",
"doi-asserted-by": "publisher",
"first-page": "113935",
"journal-title": "J Pharm Biomed Anal",
"key": "2030_CR23",
"unstructured": "Habler K, Brügel M, Teupser D, Liebchen U, Scharf C, Schönermarck U, Vogeser M, Paal M (2021) Simultaneous quantification of seven repurposed COVID-19 drugs remdesivir (plus metabolite GS-441524), chloroquine, hydroxychloroquine, lopinavir, ritonavir, favipiravir and azithromycin by a two-dimensional isotope dilution LC-MS/MS method in human serum. J Pharm Biomed Anal 196:113935. https://doi.org/10.1016/j.jpba.2021.113935",
"volume": "196",
"year": "2021"
},
{
"DOI": "10.1186/s12913-022-08272-z",
"author": "N Krishnamurthy",
"doi-asserted-by": "publisher",
"first-page": "970",
"issue": "1",
"journal-title": "BMC Health Serv Res",
"key": "2030_CR24",
"unstructured": "Krishnamurthy N, Grimshaw AA, Axson SA, Choe SH, Miller JE (2022) Drug repurposing: a systematic review on root causes, barriers and facilitators. BMC Health Serv Res 22(1):970. https://doi.org/10.1186/s12913-022-08272-z",
"volume": "22",
"year": "2022"
},
{
"DOI": "10.1038/s41598-022-17082-6",
"author": "O Kutkat",
"doi-asserted-by": "publisher",
"first-page": "12920",
"issue": "1",
"journal-title": "Sci Rep",
"key": "2030_CR25",
"unstructured": "Kutkat O, Moatasim Y, Al-Karmalawy AA, Abulkhair HS, Gomaa MR, El-Taweel AN, Abo Shama NM, GabAllah M, Mahmoud DB, Kayali G, Ali MA, Kandeil A, Mostafa A (2022) Robust antiviral activity of commonly prescribed antidepressants against emerging coronaviruses: in vitro and in silico drug repurposing studies. Sci Rep 12(1):12920. https://doi.org/10.1038/s41598-022-17082-6",
"volume": "12",
"year": "2022"
},
{
"DOI": "10.1016/j.ejphar.2020.173748",
"author": "AS Lokhande",
"doi-asserted-by": "publisher",
"first-page": "173748",
"journal-title": "Eur J Pharmacol",
"key": "2030_CR26",
"unstructured": "Lokhande AS, Devarajan PV (2021) A review on possible mechanistic insights of Nitazoxanide for repurposing in COVID-19. Eur J Pharmacol 891:173748. https://doi.org/10.1016/j.ejphar.2020.173748",
"volume": "891",
"year": "2021"
},
{
"DOI": "10.3390/v16121856",
"doi-asserted-by": "publisher",
"key": "2030_CR27",
"unstructured": "Mattos M, Sacramento CQ, Ferreira AC, Fintelman-Rodrigues N, Pereira-Dutra FS, de Freitas CS, Gesto JSM, Temerozo JR, Silva A, Moreira MTG, Silva RSC, Silveira GPE, Pinto DP, Pereira HM, Fonseca LB, Ferreira A, Blanco M, Viola C, Bou-Habib JPB, Bozza DC, P.T., and, Souza TML (2024) Newly proposed dose of daclatasvir to prevent lethal SARS-CoV-2 infection in human transgenic ACE-2 mice. Viruses 16(12). https://doi.org/10.3390/v16121856"
},
{
"DOI": "10.1002/cpmc.105",
"author": "EJ Mendoza",
"doi-asserted-by": "publisher",
"first-page": "ecpmc105",
"issue": "1",
"journal-title": "Curr Protoc Microbiol",
"key": "2030_CR28",
"unstructured": "Mendoza EJ, Manguiat K, Wood H, Drebot M (2020) Two detailed plaque assay protocols for the quantification of infectious SARS-CoV-2. Curr Protoc Microbiol 57(1):ecpmc105. https://doi.org/10.1002/cpmc.105",
"volume": "57",
"year": "2020"
},
{
"DOI": "10.1016/j.pharmthera.2021.107930",
"author": "YL Ng",
"doi-asserted-by": "publisher",
"first-page": "107930",
"journal-title": "Pharmacol Ther",
"key": "2030_CR29",
"unstructured": "Ng YL, Salim CK, Chu JJH (2021) Drug repurposing for COVID-19: approaches, challenges and promising candidates. Pharmacol Ther 228:107930. https://doi.org/10.1016/j.pharmthera.2021.107930",
"volume": "228",
"year": "2021"
},
{
"DOI": "10.1016/j.jchromb.2021.122641",
"author": "R Nguyen",
"doi-asserted-by": "publisher",
"first-page": "122641",
"journal-title": "J Chromatogr B Analyt Technol Biomed Life Sci",
"key": "2030_CR30",
"unstructured": "Nguyen R, Goodell JC, Shankarappa PS, Zimmerman S, Yin T, Peer CJ, Figg WD (2021) Development and validation of a simple, selective, and sensitive LC-MS/MS assay for the quantification of remdesivir in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 1171:122641. https://doi.org/10.1016/j.jchromb.2021.122641",
"volume": "1171",
"year": "2021"
},
{
"DOI": "10.1016/s1473-3099(22)00433-9",
"author": "N Perico",
"doi-asserted-by": "publisher",
"first-page": "e22",
"issue": "1",
"journal-title": "Lancet Infect Dis",
"key": "2030_CR31",
"unstructured": "Perico N, Cortinovis M, Suter F, Remuzzi G (2023) Home as the new frontier for the treatment of COVID-19: the case for anti-inflammatory agents. Lancet Infect Dis 23(1):e22–e33. https://doi.org/10.1016/s1473-3099(22)00433-9",
"volume": "23",
"year": "2023"
},
{
"DOI": "10.1038/s41598-023-29517-9",
"author": "SR Radoshitzky",
"doi-asserted-by": "publisher",
"first-page": "3131",
"issue": "1",
"journal-title": "Sci Rep",
"key": "2030_CR32",
"unstructured": "Radoshitzky SR, Iversen P, Lu X, Zou J, Kaptein SJF, Stuthman KS, Van Tongeren SA, Steffens J, Gong R, Truong H, Sapre AA, Yang H, Xie X, Chia JJ, Song ZJ, Leventhal SM, Chan J, Shornikov A, Zhang X, Cowfer D, Yu H, Warren T, Cihlar T, Porter DP, Neyts J, Shi PY, Wells J, Bilello JP, Feng JY (2023) Expanded profiling of Remdesivir as a broad-spectrum antiviral and low potential for interaction with other medications in vitro. Sci Rep 13(1):3131. https://doi.org/10.1038/s41598-023-29517-9",
"volume": "13",
"year": "2023"
},
{
"DOI": "10.1016/j.jbior.2021.100818",
"author": "E Ricciotti",
"doi-asserted-by": "publisher",
"first-page": "100818",
"journal-title": "Adv Biol Regul",
"key": "2030_CR33",
"unstructured": "Ricciotti E, Laudanski K, FitzGerald GA (2021) Nonsteroidal anti-inflammatory drugs and glucocorticoids in COVID-19. Adv Biol Regul 81:100818. https://doi.org/10.1016/j.jbior.2021.100818",
"volume": "81",
"year": "2021"
},
{
"DOI": "10.1016/j.jiph.2016.04.001",
"author": "JF Rossignol",
"doi-asserted-by": "publisher",
"first-page": "227",
"issue": "3",
"journal-title": "J Infect Public Health",
"key": "2030_CR34",
"unstructured": "Rossignol JF (2016) Nitazoxanide, a new drug candidate for the treatment of Middle East respiratory syndrome coronavirus. J Infect Public Health 9(3):227–230. https://doi.org/10.1016/j.jiph.2016.04.001",
"volume": "9",
"year": "2016"
},
{
"DOI": "10.1093/jac/dkab072",
"author": "CQ Sacramento",
"doi-asserted-by": "publisher",
"first-page": "1874",
"issue": "7",
"journal-title": "J Antimicrob Chemother",
"key": "2030_CR35",
"unstructured": "Sacramento CQ, Fintelman-Rodrigues N, Temerozo JR, Da Silva APD, Dias S, da Silva CDS, Ferreira AC, Mattos M, Pão CRR, de Freitas CS, Soares VC, Hoelz LVB, Fernandes TVA, Branco FSC, Bastos MM, Boechat N, Saraiva FB, Ferreira MA, Jockusch S, Wang X, Tao C, Chien M, Xie W, Patel D, Garzia A, Tuschl T, Russo JJ, Rajoli RKR, Pedrosa CSG, Vitória G, Souza LRQ, Goto-Silva L, Guimarães MZ, Rehen SK, Owen A, Bozza FA, Bou-Habib DC, Ju J, Bozza PT, Souza TML (2021) In vitro antiviral activity of the anti-HCV drugs daclatasvir and sofosbuvir against SARS-CoV-2, the aetiological agent of COVID-19. J Antimicrob Chemother 76(7):1874–1885. https://doi.org/10.1093/jac/dkab072",
"volume": "76",
"year": "2021"
},
{
"DOI": "10.1007/s00580-016-2284-3",
"author": "SA Saganuwan",
"doi-asserted-by": "publisher",
"first-page": "941",
"issue": "5",
"journal-title": "Comp Clin Pathol",
"key": "2030_CR36",
"unstructured": "Saganuwan SA (2016) Physicochemical and structure-activity properties of piroxicam—a mini review. Comp Clin Pathol 25(5):941–945. https://doi.org/10.1007/s00580-016-2284-3",
"volume": "25",
"year": "2016"
},
{
"DOI": "10.1016/j.nmni.2021.100895",
"author": "M Shabani",
"doi-asserted-by": "publisher",
"first-page": "100895",
"journal-title": "New Microbes New Infect",
"key": "2030_CR37",
"unstructured": "Shabani M, Sadegh Ehdaei B, Fathi F, Dowran R (2021) A mini-review on sofosbuvir and daclatasvir treatment in coronavirus disease 2019. New Microbes New Infect 42:100895. https://doi.org/10.1016/j.nmni.2021.100895",
"volume": "42",
"year": "2021"
},
{
"DOI": "10.1016/s2213-2600(22)00412-x",
"author": "PL Shah",
"doi-asserted-by": "publisher",
"first-page": "415",
"issue": "5",
"journal-title": "Lancet Respir Med",
"key": "2030_CR38",
"unstructured": "Shah PL, Orton CM, Grinsztejn B, Donaldson GC, Crabtree Ramírez B, Tonkin J, Santos BR, Cardoso SW, Ritchie AI, Conway F, Riberio MPD, Wiseman DJ, Tana A, Vijayakumar B, Caneja C, Leaper C, Mann B, Samson A, Bhavsar PK, Boffito M, Johnson MR, Pozniak A, Pelly M (2023) Favipiravir in patients hospitalised with COVID-19 (PIONEER trial): a multicentre, open-label, phase 3, randomised controlled trial of early intervention versus standard care. Lancet Respir Med 11(5):415–424. https://doi.org/10.1016/s2213-2600(22)00412-x",
"volume": "11",
"year": "2023"
},
{
"DOI": "10.4103/0250-474x.89763",
"author": "S Sharma",
"doi-asserted-by": "publisher",
"first-page": "84",
"issue": "1",
"journal-title": "Indian J Pharm Sci",
"key": "2030_CR40",
"unstructured": "Sharma S, Bhandari A, Choudhary VR, Rajpurohit H, Khandelwal P (2011) RP-HPLC method for simultaneous estimation of nitazoxanide and ofloxacin in tablets. Indian J Pharm Sci 73(1):84–88. https://doi.org/10.4103/0250-474x.89763",
"volume": "73",
"year": "2011"
},
{
"DOI": "10.3390/v13020202",
"doi-asserted-by": "publisher",
"key": "2030_CR39",
"unstructured": "Sharma A, Ahmad Farouk I, Lal SK (2021) COVID-19: A review on the novel coronavirus disease evolution, transmission, detection, control and prevention. Viruses 13(2). https://doi.org/10.3390/v13020202"
},
{
"DOI": "10.1021/acs.jmedchem.6b01594",
"author": "D Siegel",
"doi-asserted-by": "publisher",
"first-page": "1648",
"issue": "5",
"journal-title": "J Med Chem",
"key": "2030_CR41",
"unstructured": "Siegel D, Hui HC, Doerffler E, Clarke MO, Chun K, Zhang L, Neville S, Carra E, Lew W, Ross B, Wang Q, Wolfe L, Jordan R, Soloveva V, Knox J, Perry J, Perron M, Stray KM, Barauskas O, Feng JY, Xu Y, Lee G, Rheingold AL, Ray AS, Bannister R, Strickley R, Swaminathan S, Lee WA, Bavari S, Cihlar T, Lo MK, Warren TK, Mackman RL (2017) Discovery and synthesis of a phosphoramidate prodrug of a Pyrrolo[2,1-f][triazin-4-amino] Adenine C-Nucleoside (GS-5734) for the treatment of ebola and emerging viruses. J Med Chem 60(5):1648–1661. https://doi.org/10.1021/acs.jmedchem.6b01594",
"volume": "60",
"year": "2017"
},
{
"DOI": "10.1177/1060028015610342",
"author": "MA Smith",
"doi-asserted-by": "publisher",
"first-page": "39",
"issue": "1",
"journal-title": "Ann Pharmacother",
"key": "2030_CR42",
"unstructured": "Smith MA, Regal RE, Mohammad RA (2016) Daclatasvir: a NS5A replication complex inhibitor for Hepatitis C infection. Ann Pharmacother 50(1):39–46. https://doi.org/10.1177/1060028015610342",
"volume": "50",
"year": "2016"
},
{
"DOI": "10.1002/ccr3.8162",
"author": "DD Stewart",
"doi-asserted-by": "publisher",
"first-page": "e8162",
"issue": "11",
"journal-title": "Clin Case Rep",
"key": "2030_CR43",
"unstructured": "Stewart DD (2023) Can Nitazoxanide and/or other anti-viral medications be a solution to long COVID? Case report with a brief literature review. Clin Case Rep 11(11):e8162. https://doi.org/10.1002/ccr3.8162",
"volume": "11",
"year": "2023"
},
{
"DOI": "10.5414/cpp40213",
"author": "A Stockis",
"doi-asserted-by": "publisher",
"first-page": "213",
"issue": "5",
"journal-title": "Int J Clin Pharmacol Ther",
"key": "2030_CR44",
"unstructured": "Stockis A, Allemon AM, De Bruyn S, Gengler C (2002) Nitazoxanide pharmacokinetics and tolerability in man using single ascending oral doses. Int J Clin Pharmacol Ther 40(5):213–220. https://doi.org/10.5414/cpp40213",
"volume": "40",
"year": "2002"
},
{
"DOI": "10.1002/jssc.201300104",
"author": "N Sultana",
"doi-asserted-by": "publisher",
"first-page": "3176",
"issue": "19",
"journal-title": "J Sep Sci",
"key": "2030_CR45",
"unstructured": "Sultana N, Arayne MS, Ali SN (2013) Liquid chromatographic method for the simultaneous determination of captopril, piroxicam, and amlodipine in bulk drug, pharmaceutical formulation, and human serum by programming the detector. J Sep Sci 36(19):3176–3183. https://doi.org/10.1002/jssc.201300104",
"volume": "36",
"year": "2013"
},
{
"DOI": "10.3390/chemosensors10020047",
"doi-asserted-by": "crossref",
"key": "2030_CR46",
"unstructured": "Varodi C, Coros M, Pogăcean F, Ciorîţă A, Turza A, Pruneanu S (2022) Nitrogen-doped graphene-based sensor for electrochemical detection of piroxicam, a NSAID Drug for COVID-19 Patients. 10(2):47"
},
{
"DOI": "10.1016/j.scitotenv.2022.155317",
"author": "D Wojcieszyńska",
"doi-asserted-by": "publisher",
"first-page": "155317",
"journal-title": "Sci Total Environ",
"key": "2030_CR47",
"unstructured": "Wojcieszyńska D, Guzik H, Guzik U (2022) Non-steroidal anti-inflammatory drugs in the era of the Covid-19 pandemic in the context of the human and the environment. Sci Total Environ 834:155317. https://doi.org/10.1016/j.scitotenv.2022.155317",
"volume": "834",
"year": "2022"
},
{
"DOI": "10.1016/j.ab.2021.114118",
"author": "D Xiao",
"doi-asserted-by": "publisher",
"first-page": "114118",
"journal-title": "Anal Biochem",
"key": "2030_CR48",
"unstructured": "Xiao D, Ling J, Tarnowski KH, Humeniuk T, German R, Mathias P, Chu A, Chen J, Y.S., and, van Ingen E (2021) Validation of LC-MS/MS methods for determination of remdesivir and its metabolites GS-441524 and GS-704277 in acidified human plasma and their application in COVID-19 related clinical studies. Anal Biochem 617:114118. https://doi.org/10.1016/j.ab.2021.114118",
"volume": "617",
"year": "2021"
},
{
"DOI": "10.1038/s41598-025-07401-y",
"author": "AA Youssef",
"doi-asserted-by": "publisher",
"first-page": "22393",
"issue": "1",
"journal-title": "Sci Rep",
"key": "2030_CR49",
"unstructured": "Youssef AA, Magdy N, Hussein LA, El-Kosasy AM (2025) Sensitive RP-HPLC method with fluorimetric detection for concurrent quantification of emtricitabine, Daclatasvir and Ledipasvir in human urine. Sci Rep 15(1):22393. https://doi.org/10.1038/s41598-025-07401-y",
"volume": "15",
"year": "2025"
},
{
"DOI": "10.1016/s0021-9673(99)00515-4",
"author": "M Yritia",
"doi-asserted-by": "publisher",
"first-page": "199",
"issue": "1–2",
"journal-title": "J Chromatogr A",
"key": "2030_CR50",
"unstructured": "Yritia M, Parra P, Fernández JM, Barbanoj JM (1999) Piroxicam quantitation in human plasma by high-performance liquid chromatography with on- and off-line solid-phase extraction. J Chromatogr A 846(1–2):199–205. https://doi.org/10.1016/s0021-9673(99)00515-4",
"volume": "846",
"year": "1999"
}
],
"reference-count": 50,
"references-count": 50,
"relation": {},
"resource": {
"primary": {
"URL": "https://link.springer.com/10.1186/s13568-026-02030-8"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Anti-COVID-19 activity and simple HPLC method for concurrent detection of repurposed drugs in novel binary mixtures",
"type": "journal-article",
"update-policy": "https://doi.org/10.1007/springer_crossmark_policy",
"volume": "16"
}