Assessing Efficacy of Antiviral Therapy for COVID-19 Patients: A Case Study on Remdesivir with Bayesian Synthesis Design and Multistate Analysis
et al., Journal of the Formosan Medical Association, doi:10.1016/j.jfma.2021.04.026, May 2021
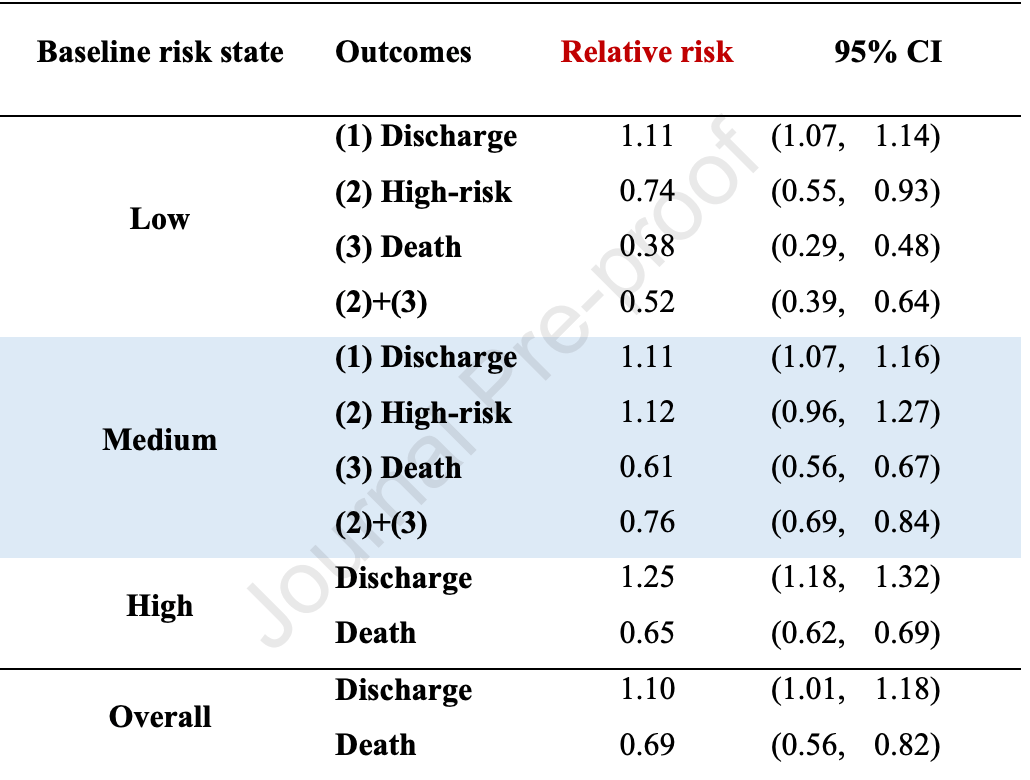
Bayesian synthesis design and multistate analysis of remdesivir results showing 31% [18-44%] lower risk of death and 10% [1-18%] higher recovery.
Currently there are 81 remdesivir studies and meta-analysis shows:
| Outcome | Improvement |
|---|---|
| Mortality | 1% higher [-8‑11%] |
| Ventilation | 11% higher [-11‑38%] |
| ICU admission | 119% higher [33‑259%] |
| Hospitalization | 21% higher [-4‑52%] |
Liao et al., 4 May 2021, peer-reviewed, 7 authors.
Assessing efficacy of antiviral therapy for COVID-19 patients: A case study on remdesivir with bayesian synthesis design and multistate analysis
Journal of the Formosan Medical Association, doi:10.1016/j.jfma.2021.04.026
Bayesian synthesis sequential design remdesivir. A multistate model was developed to model the dynamics of hospitalized COVID-19 patients from three transient states of low, medium-, and high-risk until the two outcomes of recovery and death. The outcome measures for clinical efficacy comprised high-risk state, death, and discharge. Results: The efficacy of remdesivir in reducing the risk of death and enhancing the odds of recovery were estimated as 31% (95% CI, 18e44%) and 10% (95% CI, 1e18%), respectively. Remdesivir therapy for patients with low-risk state showed the efficacy in reducing subsequent progression to high-risk state and death by 26% (relative rate (RR), 0.74; 95% CI, 0.55e0.93) and 62% (RR, 0.38; 95% CI, 0.29e0.48), respectively. Less but still statistically significant efficacy in mortality reduction was noted for the medium-and high-risk patients. Remdesivir treated patients had a significantly shorter period of hospitalization (9.9 days) compared with standard care group (12.9 days).
Conclusion: The clinical efficacy of remdesvir therapy in reducing mortality and accelerating discharge has been proved by the Bayesian synthesis design and multistate analysis.
Role of funder/sponsor The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Declaration of competing interest The authors have no conflicts of interest relevant to this article.
Appendix A. Supplementary data Supplementary data to this article can be found online at https://doi.org/10.1016/j.jfma.2021.04.026.
References
Abd-Elsalam, Esmail, Khalaf, Abdo, Medhat et al., Hydroxychloroquine in the treatment of COVID-19: a multicenter randomized controlled study, Am J Trop Med Hyg
Abdelrahman, Liu, Jiang, Li, Sun et al., Evaluation of the current therapeutic approaches for COVID-19: a systematic review and a meta-analysis, Front Pharmacol
Baden, Elsahly, Essink, Kotloff, Frey et al., Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine, N Engl J Med
Beigel, Tomashek, Dodd, Mehta, Zingman et al., Remdesivir for the treatment of covid-19 -final report, N Engl J Med
Beigel, Tomashek, Dodd, Mehta, Zingman et al., Remdesivir for the treatment of covid-19dpreliminary report, N Engl J Med
Cao, Wang, Wen, Liu, Wang et al., A trial of lopinavir-ritonavir in adults hospitalized with severe covid-19, N Engl J Med
Cavalcanti, Zampieri, Rosa, Azevedo, Veiga et al., Hydroxychloroquine with or without azithromycin in mild-to-moderate covid-19, N Engl J Med
Chen, Lin, Chen, Tseng, Wong et al., A multicenter, randomized, open-label, controlled trial to evaluate the efficacy and tolerability of hydroxychloroquine and a retrospective study in adult patients with mild to moderate coronavirus disease 2019 (COVID-19), PloS One
Cohen, Kupferschmidt, A very, very bad look" for remdesivir, Science
Dagan, Barda, Kepten, Miron, Perchik et al., BNT162b2 mRNA covid-19 vaccine in a nationwide mass vaccination setting, N Engl J Med
Dong, Du, Gardner, An interactive web-based dashboard to track COVID-19 in real time, Lancet Infect Dis
Dror, Eisenbach, Taiber, Morozov, Mizrachi et al., Vaccine hesitancy: the next challenge in the fight against COVID-19, Eur J Epidemiol
Dyer, Covid-19: remdesivir has little or no impact on survival, WHO trial shows, BMJ
Ferguson, Cummings, Cauchemez, Fraser, Riley et al., Strategies for containing an emerging influenza pandemic in Southeast Asia, Nature
Gandhi, Lynch, Rio, Mild or moderate covid-19, N Engl J Med
Gao, Yan, Huang, Liu, Zhao et al., Structure of the RNA-dependent RNA polymerase from COVID-19 virus, Science
Gordon, Tchesnokov, Feng, Porter, Go ¨tte M, The antiviral compound remdesivir potently inhibits RNAdependent RNA polymerase from Middle East respiratory syndrome coronavirus, J Biol Chem
Grein, Ohmagari, Shin, Diaz, Asperges et al., Compassionate use of remdesivir for patients with severe covid-19, N Engl J Med
Huang, Wang, Li, Ren, Zhao et al., Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China, Lancet
Jen, Chang, Lin, Hsu, Yen et al., Evaluating clinical efficacy of antiviral therapy for COVID-19: a surrogate endpoint Approach, Infect Dis Ther
Jorgensen, Kebriaei, Dresser, Remdesivir: review of pharmacology, pre-clinical data, and emerging clinical experience for COVID-19, Pharmacotherapy
Kalil, Patterson, Mehta, Tomashek, Wolfe et al., Baricitinib plus remdesivir for hospitalized adults with covid-19, N Engl J Med
Lamontagne, Agoritsas, Siemieniuk, Rochwerg, Bartoszko et al., A living WHO guideline on drugs to prevent covid-19, BMJ
Li, Guan, Wu, Wang, Zhou et al., Early transmission dynamics in wuhan, China, of novel coronavirusinfected pneumonia, N Engl J Med
Liu, Zhou, Chen, Ye, Liu et al., Efficacy and safety of antiviral treatment for COVID-19 from evidence in studies of SARS-CoV-2 and other acute viral infections: a systematic review and meta-analysis, CMAJ (Can Med Assoc J)
Mccreary, Angus, Efficacy of remdesivir in COVID-19, J Am Med Assoc
Mitja, Corbacho-Monne, Ubals, Tebe, ˜afiel et al., Hydroxychloroquine for early treatment of adults with mild covid-19: a randomized-controlled trial, Clin Infect Dis
Moscona, Neuraminidase inhibitors for influenza, N Engl J Med
Nachega, Sam-Agudu, Masekela, Van Der Zalm, Nsanzimana et al., Addressing challenges to rolling out COVID-19 vaccines in African countries, Lancet Glob Heal
Norrie, Remdesivir for COVID-19: challenges of underpowered studies, Lancet
Omrani, Pathan, Thomas, Harris, Coyle et al., Randomized double-blinded placebocontrolled trial of hydroxychloroquine with or without azithromycin for virologic cure of non-severe Covid-19, EClini-calMedicine
Peters, Chawla, Turnbull, Transforming ORs into ICUs, N Engl J Med
Phua, Weng, Ling, Egi, Lim et al., Intensive care management of coronavirus disease 2019 (COVID-19): challenges and recommendations, Lancet Respir Med
Polack, Thomas, Kitchin, Absalon, Gurtman et al., Safety and efficacy of the BNT162b2 mRNA covid-19 vaccine, N Engl J Med
Ranney, Griffeth, Jha, Critical supply shortages -the need for ventilators and personal protective equipment during the covid-19 pandemic, N Engl J Med
Recovery Collaborative Group, Horby, Mafham, Linsell, Bell et al., Effect of hydroxychloroquine in hospitalized patients with covid-19, N Engl J Med
Rubin, Chan-Tack, Farley, Sherwat, FDA approval of remdesivir -a step in the right direction, N Engl J Med
Sabino, Buss, Carvalho, Prete, Crispim et al., Resurgence of COVID-19 in Manaus, Brazil, despite high seroprevalence, Lancet
Sadoff, Legars, Shukarev, Heerwegh, Truyers et al., Interim results of a phase 1-2a trial of Ad26.COV2.S covid-19 vaccine, N Engl J Med
Schaffer Deroo, Pudalov, Ful, Planning for a COVID-19 vaccination program, J Am Med Assoc
Siemieniuk, Rochwerg, Agoritsas, Lamontagne, Leo et al., A living WHO guideline on drugs for covid-19, BMJ
Truog, Mitchell, Daley, The toughest triage -allocating ventilators in a pandemic, N Engl J Med
Vergara, Sarmiento, Lagman, Building public trust: a response to COVID-19 vaccine hesitancy predicament, J Public Health
Voysey, Clemens, Madhi, Weckx, Folegatti et al., Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK, Lancet
Wang, Zhang, Du, Du, Zhao et al., Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial, Lancet
Wu, Yu, Chen, Risk assessment of multistate progression of breast tumor with state-dependent genetic and environmental covariates, Risk Anal
DOI record:
{
"DOI": "10.1016/j.jfma.2021.04.026",
"ISSN": [
"0929-6646"
],
"URL": "http://dx.doi.org/10.1016/j.jfma.2021.04.026",
"alternative-id": [
"S0929664621001832"
],
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Assessing efficacy of antiviral therapy for COVID-19 patients: A case study on remdesivir with bayesian synthesis design and multistate analysis"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Journal of the Formosan Medical Association"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.jfma.2021.04.026"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2021 Formosan Medical Association. Published by Elsevier Taiwan LLC."
}
],
"author": [
{
"ORCID": "http://orcid.org/0000-0002-7339-2768",
"affiliation": [],
"authenticated-orcid": false,
"family": "Liao",
"given": "Sih-Han",
"sequence": "first"
},
{
"affiliation": [],
"family": "Hung",
"given": "Chien-Ching",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chen",
"given": "Chiung-Nien",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yen",
"given": "Jui-Yi",
"sequence": "additional"
},
{
"ORCID": "http://orcid.org/0000-0001-8586-0277",
"affiliation": [],
"authenticated-orcid": false,
"family": "Hsu",
"given": "Chen-Yang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yen",
"given": "Amy Ming-Fang",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Chen",
"given": "Chi-Ling",
"sequence": "additional"
}
],
"container-title": "Journal of the Formosan Medical Association",
"container-title-short": "Journal of the Formosan Medical Association",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"clinicalkey.jp",
"clinicalkey.com",
"clinicalkey.es",
"clinicalkey.com.au",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2021,
5,
4
]
],
"date-time": "2021-05-04T21:36:40Z",
"timestamp": 1620164200000
},
"deposited": {
"date-parts": [
[
2021,
8,
30
]
],
"date-time": "2021-08-30T23:59:32Z",
"timestamp": 1630367972000
},
"funder": [
{
"DOI": "10.13039/501100004663",
"award": [
"108-2118-M-038-001-MY3",
"109-2327-B-002 -009"
],
"doi-asserted-by": "publisher",
"name": "Ministry of Science and Technology, Taiwan"
}
],
"indexed": {
"date-parts": [
[
2024,
1,
13
]
],
"date-time": "2024-01-13T02:59:28Z",
"timestamp": 1705114768241
},
"is-referenced-by-count": 5,
"issued": {
"date-parts": [
[
2021,
6
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
6,
1
]
],
"date-time": "2021-06-01T00:00:00Z",
"timestamp": 1622505600000
}
},
{
"URL": "http://creativecommons.org/licenses/by-nc-nd/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
4,
29
]
],
"date-time": "2021-04-29T00:00:00Z",
"timestamp": 1619654400000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0929664621001832?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0929664621001832?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "S77-S85",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
6
]
]
},
"published-print": {
"date-parts": [
[
2021,
6
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"DOI": "10.1056/NEJMoa2001316",
"article-title": "Early transmission dynamics in wuhan, China, of novel coronavirus-infected pneumonia",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "1199",
"issue": "13",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib1",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30183-5",
"article-title": "Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China",
"author": "Huang",
"doi-asserted-by": "crossref",
"first-page": "497",
"issue": "10223",
"journal-title": "Lancet",
"key": "10.1016/j.jfma.2021.04.026_bib2",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1016/S1473-3099(20)30120-1",
"article-title": "An interactive web-based dashboard to track COVID-19 in real time",
"author": "Dong",
"doi-asserted-by": "crossref",
"first-page": "533",
"issue": "5",
"journal-title": "Lancet Infect Dis",
"key": "10.1016/j.jfma.2021.04.026_bib3",
"volume": "20",
"year": "2020"
},
{
"DOI": "10.1056/NEJMc2010853",
"article-title": "Transforming ORs into ICUs",
"author": "Peters",
"doi-asserted-by": "crossref",
"first-page": "e52",
"issue": "19",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib4",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1016/S2213-2600(20)30161-2",
"article-title": "Intensive care management of coronavirus disease 2019 (COVID-19): challenges and recommendations",
"author": "Phua",
"doi-asserted-by": "crossref",
"first-page": "506",
"issue": "5",
"journal-title": "Lancet Respir Med",
"key": "10.1016/j.jfma.2021.04.026_bib5",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1056/NEJMp2006141",
"article-title": "Critical supply shortages - the need for ventilators and personal protective equipment during the covid-19 pandemic",
"author": "Ranney",
"doi-asserted-by": "crossref",
"first-page": "e41",
"issue": "18",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib6",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1056/NEJMp2005689",
"article-title": "The toughest triage - allocating ventilators in a pandemic",
"author": "Truog",
"doi-asserted-by": "crossref",
"first-page": "1973",
"issue": "21",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib7",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2034577",
"article-title": "Safety and efficacy of the BNT162b2 mRNA covid-19 vaccine",
"author": "Polack",
"doi-asserted-by": "crossref",
"first-page": "2603",
"issue": "27",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib8",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2035389",
"article-title": "Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine",
"author": "Baden",
"doi-asserted-by": "crossref",
"first-page": "403",
"issue": "5",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib9",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2034201",
"article-title": "Interim results of a phase 1-2a trial of Ad26.COV2.S covid-19 vaccine",
"author": "Sadoff",
"doi-asserted-by": "crossref",
"first-page": "1824",
"issue": "19",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib10",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)32661-1",
"article-title": "Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK",
"author": "Voysey",
"doi-asserted-by": "crossref",
"first-page": "99",
"issue": "10269",
"journal-title": "Lancet",
"key": "10.1016/j.jfma.2021.04.026_bib11",
"volume": "397",
"year": "2021"
},
{
"DOI": "10.1056/NEJMoa2101765",
"article-title": "BNT162b2 mRNA covid-19 vaccine in a nationwide mass vaccination setting",
"author": "Dagan",
"doi-asserted-by": "crossref",
"first-page": "1412",
"issue": "15",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib12",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1503/cmaj.200647",
"article-title": "Efficacy and safety of antiviral treatment for COVID-19 from evidence in studies of SARS-CoV-2 and other acute viral infections: a systematic review and meta-analysis",
"author": "Liu",
"doi-asserted-by": "crossref",
"first-page": "E734",
"issue": "27",
"journal-title": "CMAJ (Can Med Assoc J)",
"key": "10.1016/j.jfma.2021.04.026_bib13",
"volume": "192",
"year": "2020"
},
{
"DOI": "10.3389/fphar.2021.607408",
"article-title": "Evaluation of the current therapeutic approaches for COVID-19: a systematic review and a meta-analysis",
"author": "Abdelrahman",
"doi-asserted-by": "crossref",
"first-page": "607408",
"journal-title": "Front Pharmacol",
"key": "10.1016/j.jfma.2021.04.026_bib14",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1056/NEJMcp2009249",
"article-title": "Mild or moderate covid-19",
"author": "Gandhi",
"doi-asserted-by": "crossref",
"first-page": "1757",
"issue": "18",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib15",
"volume": "383",
"year": "2020"
},
{
"author": "World Health Organization",
"key": "10.1016/j.jfma.2021.04.026_bib16",
"series-title": "Therapeutics and COVID-19: living guideline, 20 November 2020",
"year": "2020"
},
{
"first-page": "2020",
"key": "10.1016/j.jfma.2021.04.026_bib17",
"series-title": "COVID-19 Treatment guidelines",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2007016",
"article-title": "Compassionate use of remdesivir for patients with severe covid-19",
"author": "Grein",
"doi-asserted-by": "crossref",
"first-page": "2327",
"issue": "24",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib18",
"volume": "382",
"year": "2020"
},
{
"article-title": "Remdesivir for the treatment of covid-19—preliminary report",
"author": "Beigel",
"first-page": "993",
"issue": "10",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib19",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2007764",
"article-title": "Remdesivir for the treatment of covid-19 - final report",
"author": "Beigel",
"doi-asserted-by": "crossref",
"first-page": "1813",
"issue": "19",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib20",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2031994",
"article-title": "Baricitinib plus remdesivir for hospitalized adults with covid-19",
"author": "Kalil",
"doi-asserted-by": "crossref",
"first-page": "795",
"issue": "9",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib21",
"volume": "384",
"year": "2021"
},
{
"DOI": "10.1016/S0140-6736(20)31022-9",
"article-title": "Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial",
"author": "Wang",
"doi-asserted-by": "crossref",
"first-page": "1569",
"issue": "10236",
"journal-title": "Lancet",
"key": "10.1016/j.jfma.2021.04.026_bib22",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.4269/ajtmh.20-0873",
"article-title": "Hydroxychloroquine in the treatment of COVID-19: a multicenter randomized controlled study",
"author": "Abd-Elsalam",
"doi-asserted-by": "crossref",
"first-page": "1635",
"issue": "4",
"journal-title": "Am J Trop Med Hyg",
"key": "10.1016/j.jfma.2021.04.026_bib23",
"volume": "103",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2001282",
"article-title": "A trial of lopinavir-ritonavir in adults hospitalized with severe covid-19",
"author": "Cao",
"doi-asserted-by": "crossref",
"first-page": "1787",
"issue": "19",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib24",
"volume": "382",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2019014",
"article-title": "Hydroxychloroquine with or without azithromycin in mild-to-moderate covid-19",
"author": "Cavalcanti",
"doi-asserted-by": "crossref",
"first-page": "2041",
"issue": "21",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib25",
"volume": "383",
"year": "2020"
},
{
"article-title": "A multicenter, randomized, open-label, controlled trial to evaluate the efficacy and tolerability of hydroxychloroquine and a retrospective study in adult patients with mild to moderate coronavirus disease 2019 (COVID-19)",
"author": "Chen",
"issue": "12",
"journal-title": "PloS One",
"key": "10.1016/j.jfma.2021.04.026_bib26",
"volume": "15",
"year": "2020"
},
{
"DOI": "10.1056/NEJMoa2022926",
"article-title": "Effect of hydroxychloroquine in hospitalized patients with covid-19",
"author": "Horby",
"doi-asserted-by": "crossref",
"first-page": "2030",
"issue": "21",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib27",
"volume": "383",
"year": "2020"
},
{
"article-title": "Hydroxychloroquine for early treatment of adults with mild covid-19: a randomized-controlled trial",
"author": "Mitjà",
"journal-title": "Clin Infect Dis",
"key": "10.1016/j.jfma.2021.04.026_bib28",
"year": "2020"
},
{
"DOI": "10.1016/j.eclinm.2020.100645",
"article-title": "Randomized double-blinded placebo-controlled trial of hydroxychloroquine with or without azithromycin for virologic cure of non-severe Covid-19",
"author": "Omrani",
"doi-asserted-by": "crossref",
"first-page": "100645",
"journal-title": "EClinicalMedicine",
"key": "10.1016/j.jfma.2021.04.026_bib29",
"volume": "29",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)32013-4",
"article-title": "Lopinavir-ritonavir in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial",
"doi-asserted-by": "crossref",
"first-page": "1345",
"issue": "10259",
"journal-title": "Lancet",
"key": "10.1016/j.jfma.2021.04.026_bib30",
"volume": "396",
"year": "2020"
},
{
"DOI": "10.1136/bmj.n526",
"article-title": "A living WHO guideline on drugs to prevent covid-19",
"author": "Lamontagne",
"doi-asserted-by": "crossref",
"first-page": "n526",
"journal-title": "BMJ",
"key": "10.1016/j.jfma.2021.04.026_bib31",
"volume": "372",
"year": "2021"
},
{
"article-title": "A living WHO guideline on drugs for covid-19",
"author": "Siemieniuk",
"first-page": "m3379",
"journal-title": "BMJ",
"key": "10.1016/j.jfma.2021.04.026_bib32",
"volume": "370",
"year": "2020"
},
{
"DOI": "10.1056/NEJMp2032369",
"article-title": "FDA approval of remdesivir - a step in the right direction",
"author": "Rubin",
"doi-asserted-by": "crossref",
"first-page": "2598",
"issue": "27",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib33",
"volume": "383",
"year": "2020"
},
{
"DOI": "10.1126/science.abb7498",
"article-title": "Structure of the RNA-dependent RNA polymerase from COVID-19 virus",
"author": "Gao",
"doi-asserted-by": "crossref",
"first-page": "779",
"issue": "6492",
"journal-title": "Science",
"key": "10.1016/j.jfma.2021.04.026_bib34",
"volume": "368",
"year": "2020"
},
{
"DOI": "10.1074/jbc.AC120.013056",
"article-title": "The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus",
"author": "Gordon",
"doi-asserted-by": "crossref",
"first-page": "4773",
"issue": "15",
"journal-title": "J Biol Chem",
"key": "10.1016/j.jfma.2021.04.026_bib35",
"volume": "295",
"year": "2020"
},
{
"DOI": "10.1002/phar.2429",
"article-title": "Remdesivir: review of pharmacology, pre-clinical data, and emerging clinical experience for COVID-19",
"author": "Jorgensen",
"doi-asserted-by": "crossref",
"first-page": "659",
"issue": "7",
"journal-title": "Pharmacotherapy",
"key": "10.1016/j.jfma.2021.04.026_bib36",
"volume": "40",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)31023-0",
"article-title": "Remdesivir for COVID-19: challenges of underpowered studies",
"author": "Norrie",
"doi-asserted-by": "crossref",
"first-page": "1525",
"issue": "10236",
"journal-title": "Lancet",
"key": "10.1016/j.jfma.2021.04.026_bib37",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.16337",
"article-title": "Efficacy of remdesivir in COVID-19",
"author": "McCreary",
"doi-asserted-by": "crossref",
"first-page": "1041",
"issue": "11",
"journal-title": "J Am Med Assoc",
"key": "10.1016/j.jfma.2021.04.026_bib38",
"volume": "324",
"year": "2020"
},
{
"DOI": "10.1126/science.370.6517.642",
"article-title": "“A very, very bad look” for remdesivir",
"author": "Cohen",
"doi-asserted-by": "crossref",
"first-page": "642",
"issue": "6517",
"journal-title": "Science",
"key": "10.1016/j.jfma.2021.04.026_bib39",
"volume": "370",
"year": "2020"
},
{
"article-title": "Covid-19: remdesivir has little or no impact on survival, WHO trial shows",
"author": "Dyer",
"journal-title": "BMJ",
"key": "10.1016/j.jfma.2021.04.026_bib40",
"volume": "371",
"year": "2020"
},
{
"author": "World Health Organization",
"key": "10.1016/j.jfma.2021.04.026_bib41",
"series-title": "WHO R&D Blueprint novel coronavirus: COVID-19 therapeutic trial synopsis",
"year": "2020"
},
{
"article-title": "Evaluating clinical efficacy of antiviral therapy for COVID-19: a surrogate endpoint Approach",
"author": "Jen",
"first-page": "1",
"journal-title": "Infect Dis Ther",
"key": "10.1016/j.jfma.2021.04.026_bib42",
"year": "2021"
},
{
"DOI": "10.1111/risa.12116",
"article-title": "Risk assessment of multistate progression of breast tumor with state-dependent genetic and environmental covariates",
"author": "Wu",
"doi-asserted-by": "crossref",
"first-page": "367",
"issue": "2",
"journal-title": "Risk Anal",
"key": "10.1016/j.jfma.2021.04.026_bib43",
"volume": "34",
"year": "2014"
},
{
"DOI": "10.1016/S2214-109X(21)00097-8",
"article-title": "Addressing challenges to rolling out COVID-19 vaccines in African countries",
"author": "Nachega",
"doi-asserted-by": "crossref",
"first-page": "e746",
"issue": "6",
"journal-title": "Lancet Glob Heal",
"key": "10.1016/j.jfma.2021.04.026_bib44",
"volume": "9",
"year": "2021"
},
{
"DOI": "10.1093/pubmed/fdaa282",
"article-title": "Building public trust: a response to COVID-19 vaccine hesitancy predicament",
"author": "Vergara",
"doi-asserted-by": "crossref",
"journal-title": "J Public Health",
"key": "10.1016/j.jfma.2021.04.026_bib45",
"year": "2021"
},
{
"DOI": "10.1001/jama.2020.8711",
"article-title": "Planning for a COVID-19 vaccination program",
"author": "Schaffer DeRoo",
"doi-asserted-by": "crossref",
"first-page": "2458",
"issue": "24",
"journal-title": "J Am Med Assoc",
"key": "10.1016/j.jfma.2021.04.026_bib46",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1007/s10654-020-00671-y",
"article-title": "Vaccine hesitancy: the next challenge in the fight against COVID-19",
"author": "Dror",
"doi-asserted-by": "crossref",
"first-page": "775",
"issue": "8",
"journal-title": "Eur J Epidemiol",
"key": "10.1016/j.jfma.2021.04.026_bib47",
"volume": "35",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(21)00183-5",
"article-title": "Resurgence of COVID-19 in Manaus, Brazil, despite high seroprevalence",
"author": "Sabino",
"doi-asserted-by": "crossref",
"first-page": "452",
"issue": "10273",
"journal-title": "Lancet",
"key": "10.1016/j.jfma.2021.04.026_bib48",
"volume": "397",
"year": "2021"
},
{
"DOI": "10.1038/nature04017",
"article-title": "Strategies for containing an emerging influenza pandemic in Southeast Asia",
"author": "Ferguson",
"doi-asserted-by": "crossref",
"first-page": "209",
"issue": "7056",
"journal-title": "Nature",
"key": "10.1016/j.jfma.2021.04.026_bib49",
"volume": "437",
"year": "2005"
},
{
"DOI": "10.1056/NEJMra050740",
"article-title": "Neuraminidase inhibitors for influenza",
"author": "Moscona",
"doi-asserted-by": "crossref",
"first-page": "1363",
"issue": "13",
"journal-title": "N Engl J Med",
"key": "10.1016/j.jfma.2021.04.026_bib50",
"volume": "353",
"year": "2005"
}
],
"reference-count": 50,
"references-count": 50,
"relation": {},
"resource": {
"primary": {
"URL": "https://linkinghub.elsevier.com/retrieve/pii/S0929664621001832"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [],
"subtitle": [],
"title": "Assessing efficacy of antiviral therapy for COVID-19 patients: A case study on remdesivir with bayesian synthesis design and multistate analysis",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "120"
}