Zinc pyrithione is a potent inhibitor of PLPro and cathepsin L enzymes with ex vivo inhibition of SARS-CoV-2 entry and replication
et al., Journal of Enzyme Inhibition and Medicinal Chemistry, doi:10.1080/14756366.2022.2108417, Aug 2022
Zinc for COVID-19
2nd treatment shown to reduce risk in
July 2020, now with p = 0.00000012 from 41 studies, recognized in 23 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Ex vivo study showing zinc pyrithione to be a potent inhibitor of SARS-CoV-2 entry and replication.
Kladnik et al., 9 Aug 2022, peer-reviewed, 9 authors.
Ex vivo studies are an important part of preclinical research, however results may be very different in vivo.
Zinc pyrithione is a potent inhibitor of PLPro and cathepsin L enzymes with ex vivo inhibition of SARS-CoV-2 entry and replication
Journal of Enzyme Inhibition and Medicinal Chemistry, doi:10.1080/14756366.2022.2108417
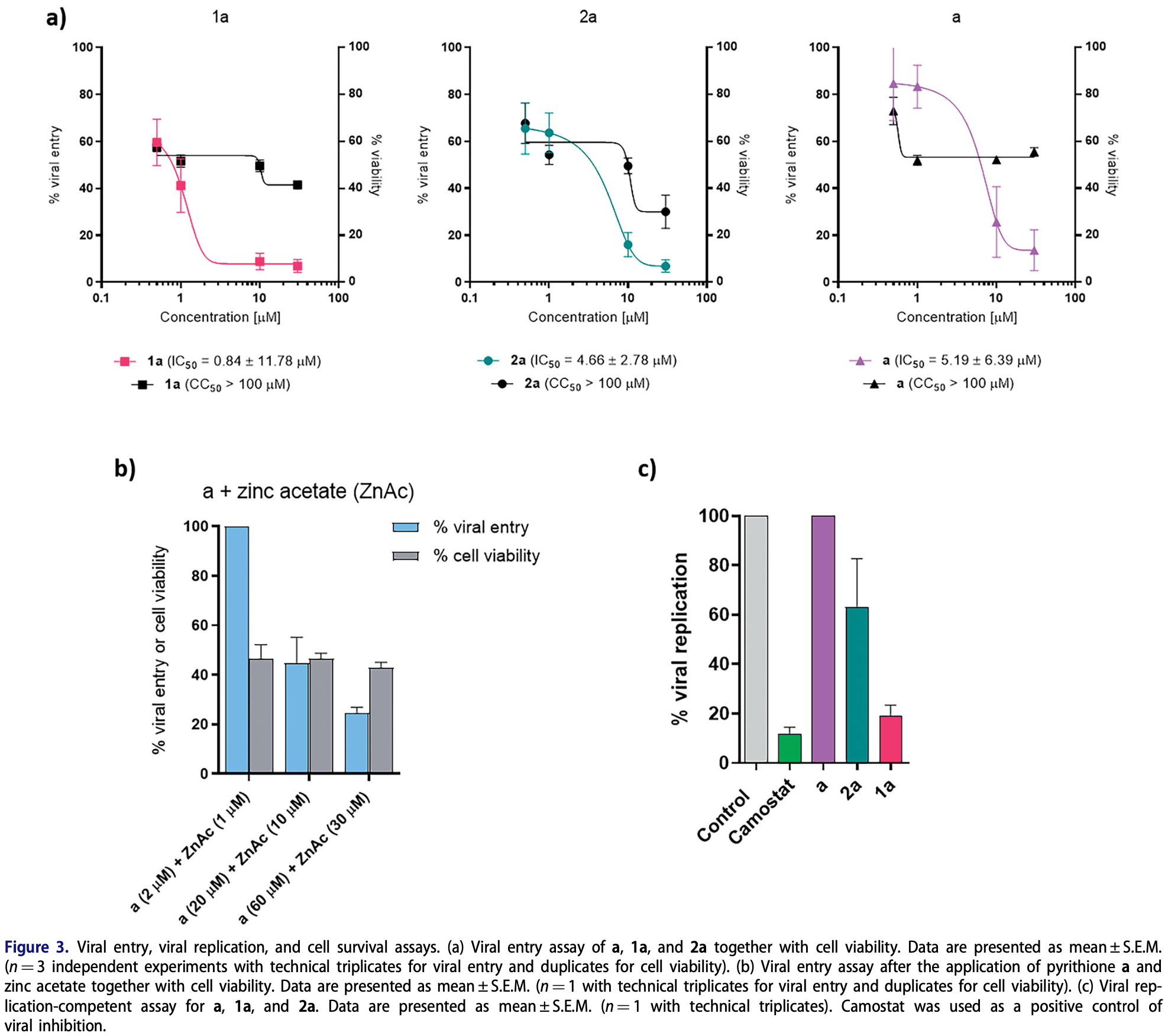
Zinc pyrithione (1a), together with its analogues 1b-h and ruthenium pyrithione complex 2a, were synthesised and evaluated for the stability in biologically relevant media and anti-SARS-CoV-2 activity. Zinc pyrithione revealed potent in vitro inhibition of cathepsin L (IC 50 ¼1.88 ± 0.49 mM) and PL Pro (IC 50 ¼0.50 ± 0.07 mM), enzymes involved in SARS-CoV-2 entry and replication, respectively, as well as antiviral entry and replication properties in an ex vivo system derived from primary human lung tissue. Zinc complexes 1b-h expressed comparable in vitro inhibition. On the contrary, ruthenium complex 2a and the ligand pyrithione a itself expressed poor inhibition in mentioned assays, indicating the importance of the selection of metal core and structure of metal complex for antiviral activity. Safe, effective, and preferably oral at-home therapeutics for COVID-19 are needed and as such zinc pyrithione, which is also commercially available, could be considered as a potential therapeutic agent against SARS-CoV-2.
Disclosure statement No potential conflict of interest was reported by the author(s).
Funding The
References
Asl, Nikfarjam, Zolbanin, Immunopharmacological perspective on zinc in SARS-CoV-2 infection, Int Immunopharmacol
B Aez-Santos, John, Mesecar, The SARS-coronavirus papain-like protease: structure, function and inhibition by designed antiviral compounds, Antiviral Res
Bafaro, Liu, Xu, Dempski, The emerging role of zinc transporters in cellular homeostasis and cancer, Signal Transduct Target Ther
Bakowski, Beutler, Wolff, Drug repurposing screens identify chemical entities for the development of COVID-19 interventions, Nat Commun
Barnett, Kretschmar, Hartman, Structural characterization of bis(N-oxopyridine-2-thionato)zinc(II), Inorg Chem
Barth, Rink, Wessels, Increase of the intracellular zinc concentration leads to an activation and internalisation of the epidermal growth factor receptor in A549 cells, Int J Mol Sci
Bolognesi, Harnessing polypharmacology with medicinal chemistry, ACS Med Chem Lett
Brewer, Marti, Brufsky, Potential interventions for SARS-CoV-2 infections: zinc showing promise, J Med Virol
Cavalcanti, Zampieri, Rosa, Hydroxychloroquine with or without azithromycin in mildto-moderate Covid-19, N Engl J Med
Chinni, El-Khoury, Perera, Zinc supplementation as an adjunct therapy for COVID-19: challenges and opportunities, Br J Clin Pharmacol
Daina, Michielin, Zoete, SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules, Sci Rep
Ddn, Cruz, Oliveira Ad, Antiviral and immunological activity of zinc and possible role in COVID-19, Br J Nutr
De Arruda, Rocha, Barrionuevo, The influence of Zn II coordination sphere and chemical structure over the reactivity of metallo-b-lactamase model compounds, Dalton Trans
De Paiva, Neto, Santos, What is holding back the development of antiviral metallodrugs? A literature overview and implications for SARS-CoV-2 therapeutics and future viral outbreaks, Dalton Trans
Doose, Ranke, Stock, Structure-activity relationships of pyrithiones-IPC-81 toxicity tests with the antifouling biocide zinc pyrithione and structural analogues, Green Chem
Equils, Lekaj, Wu, Intra-nasal zinc level relationship to COVID-19 anosmia and type 1 interferon response: a proposal, Laryngoscope Investig Otolaryngol
Frontera, Rahimian, Yaghi, Treatment with zinc is associated with reduced in-hospital mortality among COVID-19 patients: a multi-center cohort study, Res Sq
Gao, Gesenberg, Zheng, Oral formulations for preclinical studies: principle, design, and development considerations
Gawish, Starkl, Pimenov, ACE2 is the critical in vivo receptor for SARS-CoV-2 in a novel COVID-19 mouse model with TNF-and IFNc-driven immunopathology, Elife
Gomez-Orellana, Strategies to improve oral drug bioavailability, Expert Opin Drug Deliv
Grau-Exp Osito, Perea, Suppi, Evaluation of SARS-CoV-2 entry, inflammation and new therapeutics in human lung tissue cells, PLOS Pathog
Grifagni, Calderone, Giuntini, SARS-CoV-2 Mpro inhibition by a zinc ion: structural features and hints for drug design, Chem Commun
Han, Chang, Juo, Papain-like protease 2 (PLP2) from severe acute respiratory syndrome coronavirus (SARS-CoV): expression, purification, characterization, and inhibition, Biochemistry
Hecel, Ostrowska, Stokowa-Sołtys, Zinc(II)-the overlooked eminence grise of chloroquine's fight against COVID-19?, Pharmaceuticals
Hoang, Han, A possible application of hinokitiol as a natural zinc ionophore and anti-infective agent for the prevention and treatment of COVID-19 and viral infections, Med Hypotheses
Hoffmann, P€ Ohlmann S, How SARS-CoV-2 makes the cut, Nat Microbiol
Hsu, Kuo, Hsieh, Evaluation of metal-conjugated compounds as inhibitors of 3CL protease of SARS-CoV, FEBS Lett
Hu, Guo, Zhou, Characteristics of SARS-CoV-2 and COVID-19, Nat Rev Microbiol
Hu, Wang, Hu, A novel framework integrating AI model and enzymological experiments promotes identification of SARS-CoV-2 3CL protease inhibitors and activitybased probe, Brief Bioinform
Jankelson, Karam, Becker, QT prolongation, torsades de pointes, and sudden death with short courses of chloroquine or hydroxychloroquine as used in COVID-19: a systematic review, Heart Rhythm
Kladnik, Coverdale, Kljun, Organoruthenium complexes with benzo-fused pyrithiones overcome platinum resistance in ovarian cancer cells, Cancers
Kladnik, Dolinar, Kljun, Zinc pyrithione is a potent inhibitor of PLPro and cathepsin L enzymes with ex vivo inhibition of SARS-CoV-2 entry and replication, bioRxiv
Kladnik, Kljun, Burmeister, Towards identification of essential structural elements of organoruthenium(II)-pyrithionato complexes for anticancer activity, Che Eur J
Kladnik, Ristovski, Kljun, Structural isomerism and enhanced lipophilicity of pyrithione ligands of organoruthenium(II) complexes increase inhibition on AChE and BuChE, Int J Mol Sci
Kljun, Anko, Traven, Pyrithione-based ruthenium complexes as inhibitors of aldo-keto reductase 1C enzymes and anticancer agents, Dalton Trans
Komai, Goto, Suzuki, Zinc deficiency and taste dysfunction; contribution of carbonic anhydrase, a zinc-metalloenzyme, to normal taste sensation, BioFactors
Kre, Zel, Maret, The biological inorganic chemistry of zinc ions, Arch Biochem Biophys
Kuzikov, Costanzi, Reinshagen, Identification of inhibitors of SARS-CoV-2 3CL-Pro enzymatic activity using a small molecule in vitro repurposing screen, ACS Pharmacol Transl Sci
Li, Oliveira, Walker, The antimicrobial and immunomodulatory effects of ionophores for the treatment of human infection, J Inorg Biochem
Liu, Luo, Libby, Shi, Cathepsin L-selective inhibitors: a potentially promising treatment for COVID-19 patients, Pharmacol Ther
Lu, Zhao, Li, Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding, Lancet
Magda, Lecane, Wang, Synthesis and anticancer properties of water-soluble zinc ionophores, Cancer Res
Maio, Lafont, Sil, Fe-S cofactors in the SARS-CoV-2 RNA-dependent RNA polymerase are potential antiviral targets, Science
Mayor-Ibarguren, Busca-Arenzana, Robles-Marhuenda, A hypothesis for the possible role of zinc in the immunological pathways related to COVID-19 infection, Front Immunol
Mercuro, Yen, Shim, Risk of QT interval prolongation associated with use of hydroxychloroquine with or without concomitant azithromycin among hospitalized patients testing positive for coronavirus disease 2019 (COVID-19), JAMA Cardiol
Mirza, Ahmad, Abdullah, Identification of novel human USP2 inhibitor and its putative role in treatment of COVID-19 by inhibiting SARS-CoV-2 papain-like (PLpro) protease, J Bioinform Comput Biol
Moseley, Current evidence supporting the use of orally administered zinc in the treatment of COVID-19, OSF Preprints
Niu, Sun, Lu, Synthesis and crystal structure of dinuclear zinc(II) complex with 3-Me-N-oxopyridine-2-thiol, Chin J Struct Chem
Novinec, Pav Si C, Ci C B, A simple and efficient protocol for the production of recombinant cathepsin V and other cysteine cathepsins in soluble form in Escherichia coli, Protein Expr Purif
Osipiuk, Azizi, Dvorkin, Structure of papain-like protease from SARS-CoV-2 and its complexes with non-covalent inhibitors, Nat Commun
Palmier, Van Doren Sr, Rapid determination of enzyme kinetics from fluorescence: overcoming the inner filter effect, Anal Biochem
Panchariya, Khan, Kuila, Zinc 2þ ion inhibits SARS-CoV-2 main protease and viral replication in vitro, Chem Commun
Park, Cho, Lee, Understanding the mechanism of action of the anti-dandruff agent zinc pyrithione against Malassezia restricta, Sci Rep
Patel, Chinni, El-Khoury, A pilot double-blind safety and feasibility randomized controlled trial of highdose intravenous zinc in hospitalized COVID-19 patients, J Med Virol
Perera, Khoury, Chinni, Randomised controlled trial for high-dose intravenous zinc as adjunctive therapy in SARS-CoV-2 (COVID-19) positive critically ill patients: trial protocol, BMJ Open
Pormohammad, Monych, Turner, Zinc and SARS CoV 2: a molecular modeling study of Zn interactions with RNA dependent RNA polymerase and 3C like proteinase enzymes, Int J Mol Med
Ramajayam, Tan, Liang, Recent development of 3C and 3CL protease inhibitors for anti-coronavirus and antipicornavirus drug discovery, Biochem Soc Trans
Reeder, Kaplan, Xu, Zinc pyrithione inhibits yeast growth through copper influx and inactivation of ironsulfur proteins, Antimicrob Agents Chemother
Reeder, Xu, Youngquist, The antifungal mechanism of action of zinc pyrithione, Br J Dermatol
Rudzi Nska, Parodi, Soond, The role of cysteine cathepsins in cancer progression and drug resistance, Int J Mol Sci
Sacco, Ma, Lagarias, Structure and inhibition of the SARS-CoV-2 main protease reveal strategy for developing dual inhibitors against M pro and cathepsin L, Sci Adv
Scavo, Oliveri, Zinc ionophores: chemistry and biological applications, J Inorg Biochem
Shang, Ye, Shi, Structural basis of receptor recognition by SARS-CoV-2, Nature
Shin, Mukherjee, Grewe, Papain-like protease regulates SARS-CoV-2 viral spread and innate immunity, Nature
Skalny, Rink, Ajsuvakova, Zinc and respiratory tract infections: perspectives for COVID-19 (review), Int J Mol Med
Sulaiman, Aljuhani, Shaya, Evaluation of zinc sulfate as an adjunctive therapy in COVID-19 critically ill patients: a two center propensity-score matched study, Crit Care
Tan, Hu, Jadhav, Progress and challenges in targeting the SARS-CoV-2 papain-like protease, J Med Chem
Thomas, Patel, Bittel, Effect of high-dose zinc and ascorbic acid supplementation vs usual care on symptom length and reduction among ambulatory patients with SARS-CoV-2 infection: the COVID A to Z randomized clinical trial, JAMA Netw Open
V'kovski, Kratzel, Steiner, Coronavirus biology and replication: implications for SARS-CoV-2, Nat Rev Microbiol
Velthuis, Van Den Worml, Sims, Zn2þ inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture, PLoS Pathog
Vogel-Gonz Alez, Tall O-Parra, Herrera-Fern Andez, Low zinc levels at admission associates with poor clinical outcomes in SARS-CoV-2 infection, Nutrients
Wang, Chan, Wang, Orally administered bismuth drug together with N-acetyl cysteine as a broad-spectrum anti-coronavirus cocktail therapy, Chem Sci
Wen, Jung, Li, Drug delivery approaches in addressing clinical pharmacology-related issues: opportunities and challenges, AAPS J
Wessels, Rolles, Rink, The potential impact of zinc supplementation on COVID-19 pathogenesis, Front Immunol
West, Brown, Jasinski, Crystal structures of the cobalt(III), nickel(II), copper(II), and zinc(II) complexes of 2-thio-6-picoline N-oxide, J Chem Crystallogr
Xiong, Song, You, Syntheses and properties of some transition metal complexes with methyl substituted 1-hydroxy-2(1H)-pyridinethione and crystal structure of bis(1-hydroxy-4-methyl-2(1H)pyridinethionato-O,S 0 )zinc(II), Polyhedron
Zhao, Yang, Yang, Cathepsin L plays a key role in SARS-CoV-2 infection in humans and humanized mice and is a promising target for new drug development, Signal Transduct Target Ther
Zoghi, Khamirani, Dastgheib, An analysis of inhibition of the severe acute respiratory syndrome coronavirus 2 RNA-dependent RNA polymerase by zinc ion: an in silico approach, Future Virol
DOI record:
{
"DOI": "10.1080/14756366.2022.2108417",
"ISSN": [
"1475-6366",
"1475-6374"
],
"URL": "http://dx.doi.org/10.1080/14756366.2022.2108417",
"alternative-id": [
"10.1080/14756366.2022.2108417"
],
"assertion": [
{
"label": "Peer Review Statement",
"name": "peerreview_statement",
"order": 1,
"value": "The publishing and review policy for this title is described in its Aims & Scope."
},
{
"URL": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=ienz20",
"label": "Aim & Scope",
"name": "aims_and_scope_url",
"order": 2,
"value": "http://www.tandfonline.com/action/journalInformation?show=aimsScope&journalCode=ienz20"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Received",
"name": "received",
"order": 0,
"value": "2022-06-21"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Revised",
"name": "revised",
"order": 1,
"value": "2022-07-23"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Accepted",
"name": "accepted",
"order": 2,
"value": "2022-07-27"
},
{
"group": {
"label": "Publication History",
"name": "publication_history"
},
"label": "Published",
"name": "published",
"order": 3,
"value": "2022-08-09"
}
],
"author": [
{
"affiliation": [
{
"name": "Faculty of Chemistry and Chemical Technology, University of Ljubljana, Ljubljana, Slovenia"
}
],
"family": "Kladnik",
"given": "Jerneja",
"sequence": "first"
},
{
"affiliation": [
{
"name": "Faculty of Chemistry and Chemical Technology, University of Ljubljana, Ljubljana, Slovenia"
}
],
"family": "Dolinar",
"given": "Ana",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Faculty of Chemistry and Chemical Technology, University of Ljubljana, Ljubljana, Slovenia"
}
],
"family": "Kljun",
"given": "Jakob",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Infectious Diseases Department, Vall d’Hebron Research Institute (VHIR), Hospital Universitari Vall d’Hebron, Universitat Autònoma de Barcelona, VHIR Task Force COVID-19, Barcelona, Spain"
}
],
"family": "Perea",
"given": "David",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Infectious Diseases Department, Vall d’Hebron Research Institute (VHIR), Hospital Universitari Vall d’Hebron, Universitat Autònoma de Barcelona, VHIR Task Force COVID-19, Barcelona, Spain"
}
],
"family": "Grau-Expósito",
"given": "Judith",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Infectious Diseases Department, Vall d’Hebron Research Institute (VHIR), Hospital Universitari Vall d’Hebron, Universitat Autònoma de Barcelona, VHIR Task Force COVID-19, Barcelona, Spain"
}
],
"family": "Genescà",
"given": "Meritxell",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Faculty of Chemistry and Chemical Technology, University of Ljubljana, Ljubljana, Slovenia"
}
],
"family": "Novinec",
"given": "Marko",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Infectious Diseases Department, Vall d’Hebron Research Institute (VHIR), Hospital Universitari Vall d’Hebron, Universitat Autònoma de Barcelona, VHIR Task Force COVID-19, Barcelona, Spain"
}
],
"family": "Buzon",
"given": "Maria J.",
"sequence": "additional"
},
{
"affiliation": [
{
"name": "Faculty of Chemistry and Chemical Technology, University of Ljubljana, Ljubljana, Slovenia"
}
],
"family": "Turel",
"given": "Iztok",
"sequence": "additional"
}
],
"container-title": "Journal of Enzyme Inhibition and Medicinal Chemistry",
"container-title-short": "Journal of Enzyme Inhibition and Medicinal Chemistry",
"content-domain": {
"crossmark-restriction": true,
"domain": [
"www.tandfonline.com"
]
},
"created": {
"date-parts": [
[
2022,
8,
9
]
],
"date-time": "2022-08-09T12:22:09Z",
"timestamp": 1660047729000
},
"deposited": {
"date-parts": [
[
2022,
8,
9
]
],
"date-time": "2022-08-09T12:22:47Z",
"timestamp": 1660047767000
},
"funder": [
{
"DOI": "10.13039/501100004329",
"award": [
"P1-0175"
],
"doi-asserted-by": "publisher",
"name": "Slovenian Research Agency"
},
{
"award": [
"DGRIS 1_5"
],
"name": "Health Department of the Government of Catalonia"
},
{
"award": [
"202104FMTV3",
"202112FMTV3"
],
"name": "Fundació La Marató TV3"
},
{
"award": [
"CP17/00179"
],
"name": "Spanish Health Institute Carlos III"
}
],
"indexed": {
"date-parts": [
[
2022,
8,
10
]
],
"date-time": "2022-08-10T04:20:08Z",
"timestamp": 1660105208455
},
"is-referenced-by-count": 0,
"issue": "1",
"issued": {
"date-parts": [
[
2022,
8,
9
]
]
},
"journal-issue": {
"issue": "1",
"published-print": {
"date-parts": [
[
2022,
12,
31
]
]
}
},
"language": "en",
"license": [
{
"URL": "http://creativecommons.org/licenses/by/4.0/",
"content-version": "vor",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2022,
8,
9
]
],
"date-time": "2022-08-09T00:00:00Z",
"timestamp": 1660003200000
}
}
],
"link": [
{
"URL": "https://www.tandfonline.com/doi/pdf/10.1080/14756366.2022.2108417",
"content-type": "unspecified",
"content-version": "vor",
"intended-application": "similarity-checking"
}
],
"member": "301",
"original-title": [],
"page": "2158-2168",
"prefix": "10.1080",
"published": {
"date-parts": [
[
2022,
8,
9
]
]
},
"published-online": {
"date-parts": [
[
2022,
8,
9
]
]
},
"published-print": {
"date-parts": [
[
2022,
12,
31
]
]
},
"publisher": "Informa UK Limited",
"reference": [
{
"DOI": "10.1038/s41579-020-00459-7",
"doi-asserted-by": "publisher",
"key": "CIT0001"
},
{
"DOI": "10.1016/S0140-6736(20)30251-8",
"doi-asserted-by": "publisher",
"key": "CIT0002"
},
{
"DOI": "10.1038/s41586-020-2179-y",
"doi-asserted-by": "publisher",
"key": "CIT0003"
},
{
"DOI": "10.1038/s41564-021-00931-x",
"doi-asserted-by": "publisher",
"key": "CIT0004"
},
{
"DOI": "10.1038/s41579-020-00468-6",
"doi-asserted-by": "publisher",
"key": "CIT0005"
},
{
"key": "CIT0006",
"unstructured": "COVID-19 treatments: authorised. European Medicines Agency; 2021 [Internet] [cited 2021 Dec 17]. Available from: https://www.ema.europa.eu/en/human-regulatory/overview/public-health-threats/coronavirus-disease-covid-19/treatments-vaccines/covid-19-treatments"
},
{
"key": "CIT0007",
"unstructured": "Coronavirus (COVID-19). Drugs. FDA; 2021 [Internet] [cited 2021 Dec 17]. Available from: https://www.fda.gov/drugs/emergency-preparedness-drugs/coronavirus-covid-19-drugs"
},
{
"DOI": "10.1038/s41467-021-23328-0",
"author": "Bakowski MA",
"doi-asserted-by": "crossref",
"first-page": "3309",
"journal-title": "Nat Commun",
"key": "CIT0008",
"volume": "12",
"year": "2021"
},
{
"DOI": "10.1126/sciadv.abe0751",
"author": "Sacco MD",
"doi-asserted-by": "crossref",
"first-page": "eabe0751",
"journal-title": "Sci Adv",
"key": "CIT0009",
"volume": "6",
"year": "2022"
},
{
"author": "Bafaro E",
"first-page": "1",
"journal-title": "Signal Transduct Target Ther",
"key": "CIT0010",
"volume": "2",
"year": "2017"
},
{
"author": "Skalny AV",
"first-page": "17",
"journal-title": "Int J Mol Med",
"key": "CIT0011",
"volume": "46",
"year": "2020"
},
{
"DOI": "10.1111/bcp.14826",
"author": "Chinni V",
"doi-asserted-by": "crossref",
"first-page": "3737",
"journal-title": "Br J Clin Pharmacol",
"key": "CIT0012",
"volume": "87",
"year": "2021"
},
{
"DOI": "10.1017/S0007114521002099",
"author": "Marreiro DdN",
"doi-asserted-by": "crossref",
"first-page": "1172",
"journal-title": "Br J Nutr",
"key": "CIT0013",
"volume": "127",
"year": "2022"
},
{
"DOI": "10.31219/osf.io/z8wvq",
"doi-asserted-by": "crossref",
"key": "CIT0014",
"unstructured": "Moseley HNB. Current evidence supporting the use of orally administered zinc in the treatment of COVID-19. OSF Preprints; 2020."
},
{
"DOI": "10.1016/j.intimp.2021.107630",
"doi-asserted-by": "publisher",
"key": "CIT0015"
},
{
"DOI": "10.1002/biof.5520120111",
"doi-asserted-by": "publisher",
"key": "CIT0016"
},
{
"DOI": "10.1002/lio2.513",
"doi-asserted-by": "publisher",
"key": "CIT0017"
},
{
"DOI": "10.3389/fimmu.2020.01736",
"doi-asserted-by": "publisher",
"key": "CIT0018"
},
{
"DOI": "10.3389/fimmu.2020.01712",
"doi-asserted-by": "publisher",
"key": "CIT0019"
},
{
"DOI": "10.1002/jmv.26523",
"author": "Brewer J",
"doi-asserted-by": "crossref",
"first-page": "1201",
"journal-title": "J Med Virol",
"key": "CIT0020",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.2217/fvl-2020-0369",
"doi-asserted-by": "publisher",
"key": "CIT0021"
},
{
"DOI": "10.3892/ijmm.2020.4790",
"author": "Pormohammad A",
"doi-asserted-by": "crossref",
"first-page": "326",
"journal-title": "Int J Mol Med",
"key": "CIT0022",
"volume": "47",
"year": "2021"
},
{
"DOI": "10.1039/D1CC02956H",
"doi-asserted-by": "publisher",
"key": "CIT0023"
},
{
"DOI": "10.1039/D1CC03563K",
"author": "Panchariya L",
"doi-asserted-by": "crossref",
"first-page": "10083",
"journal-title": "Chem Commun",
"key": "CIT0024",
"volume": "57",
"year": "2021"
},
{
"DOI": "10.1371/journal.ppat.1001176",
"doi-asserted-by": "publisher",
"key": "CIT0025"
},
{
"DOI": "10.1042/BST0391371",
"doi-asserted-by": "publisher",
"key": "CIT0026"
},
{
"DOI": "10.1016/j.febslet.2004.08.015",
"doi-asserted-by": "publisher",
"key": "CIT0027"
},
{
"DOI": "10.1021/bi0504761",
"author": "Han YS",
"doi-asserted-by": "crossref",
"first-page": "10349",
"journal-title": "Biochemistry",
"key": "CIT0028",
"volume": "44",
"year": "2005"
},
{
"DOI": "10.1093/bib/bbab301",
"author": "Hu F",
"doi-asserted-by": "crossref",
"first-page": "bbab301",
"journal-title": "Brief Bioinform",
"key": "CIT0029",
"volume": "22",
"year": "2021"
},
{
"DOI": "10.1021/acsptsci.0c00216",
"doi-asserted-by": "publisher",
"key": "CIT0030"
},
{
"DOI": "10.7554/eLife.74623",
"author": "Gawish R",
"doi-asserted-by": "crossref",
"first-page": "e74623",
"journal-title": "Elife",
"key": "CIT0031",
"volume": "11",
"year": "2022"
},
{
"DOI": "10.1111/j.1365-2133.2011.10571.x",
"doi-asserted-by": "publisher",
"key": "CIT0032"
},
{
"DOI": "10.1128/AAC.00724-11",
"author": "Reeder NL",
"doi-asserted-by": "crossref",
"first-page": "5753",
"journal-title": "Antimicrob Agents Chemother",
"key": "CIT0033",
"volume": "55",
"year": "2011"
},
{
"DOI": "10.1038/s41598-018-35398-0",
"doi-asserted-by": "publisher",
"key": "CIT0034"
},
{
"DOI": "10.1126/science.abi5224",
"doi-asserted-by": "publisher",
"key": "CIT0035"
},
{
"DOI": "10.1039/C6DT00668J",
"author": "Kljun J",
"doi-asserted-by": "crossref",
"first-page": "11791",
"journal-title": "Dalton Trans",
"key": "CIT0036",
"volume": "45",
"year": "2016"
},
{
"DOI": "10.1002/chem.201903109",
"author": "Kladnik J",
"doi-asserted-by": "crossref",
"first-page": "14169",
"journal-title": "Che Eur J",
"key": "CIT0037",
"volume": "25",
"year": "2019"
},
{
"DOI": "10.3390/cancers13102493",
"doi-asserted-by": "publisher",
"key": "CIT0038"
},
{
"DOI": "10.3390/ijms21165628",
"doi-asserted-by": "publisher",
"key": "CIT0039"
},
{
"DOI": "10.1039/B314753C",
"doi-asserted-by": "publisher",
"key": "CIT0040"
},
{
"DOI": "10.1016/j.pep.2011.11.002",
"author": "Novinec M",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Protein Expr Purif",
"key": "CIT0041",
"volume": "82",
"year": "2012"
},
{
"DOI": "10.1038/s41586-020-2601-5",
"doi-asserted-by": "publisher",
"key": "CIT0042"
},
{
"DOI": "10.1016/j.ab.2007.07.008",
"doi-asserted-by": "publisher",
"key": "CIT0043"
},
{
"DOI": "10.1371/journal.ppat.1010171",
"author": "Grau-Expósito J",
"doi-asserted-by": "crossref",
"first-page": "e1010171",
"journal-title": "PLOS Pathog",
"key": "CIT0044",
"volume": "18",
"year": "2022"
},
{
"DOI": "10.1016/j.abb.2016.04.010",
"doi-asserted-by": "publisher",
"key": "CIT0045"
},
{
"DOI": "10.1158/0008-5472.CAN-08-0601",
"doi-asserted-by": "publisher",
"key": "CIT0046"
},
{
"DOI": "10.1021/ic50174a002",
"doi-asserted-by": "publisher",
"key": "CIT0047"
},
{
"author": "Niu DZ",
"first-page": "108",
"journal-title": "Chin J Struct Chem",
"key": "CIT0048",
"volume": "20",
"year": "2001"
},
{
"DOI": "10.1016/0277-5387(95)00296-8",
"doi-asserted-by": "publisher",
"key": "CIT0049"
},
{
"DOI": "10.1023/A:1022886116342",
"author": "West DX",
"doi-asserted-by": "crossref",
"first-page": "853",
"journal-title": "J Chem Crystallogr",
"key": "CIT0050",
"volume": "28",
"year": "1998"
},
{
"DOI": "10.1039/D0DT02478C",
"doi-asserted-by": "publisher",
"key": "CIT0051"
},
{
"DOI": "10.3390/nu13020562",
"doi-asserted-by": "publisher",
"key": "CIT0052"
},
{
"DOI": "10.1001/jamanetworkopen.2021.0369",
"doi-asserted-by": "publisher",
"key": "CIT0053"
},
{
"DOI": "10.1186/s13054-021-03785-1",
"author": "Al Sulaiman K",
"doi-asserted-by": "crossref",
"first-page": "363",
"journal-title": "Crit Care",
"key": "CIT0054",
"volume": "25",
"year": "2021"
},
{
"DOI": "10.1136/bmjopen-2020-040580",
"author": "Perera M",
"doi-asserted-by": "crossref",
"first-page": "e040580",
"journal-title": "BMJ Open",
"key": "CIT0055",
"volume": "10",
"year": "2020"
},
{
"DOI": "10.1002/jmv.26895",
"author": "Patel O",
"doi-asserted-by": "crossref",
"first-page": "3261",
"journal-title": "J Med Virol",
"key": "CIT0056",
"volume": "93",
"year": "2021"
},
{
"DOI": "10.1016/j.mehy.2020.110333",
"author": "Hoang BX",
"doi-asserted-by": "crossref",
"first-page": "110333",
"journal-title": "Med Hypotheses",
"key": "CIT0057",
"volume": "145",
"year": "2020"
},
{
"DOI": "10.3390/ph13090228",
"doi-asserted-by": "publisher",
"key": "CIT0058"
},
{
"DOI": "10.1016/j.jinorgbio.2021.111691",
"doi-asserted-by": "publisher",
"key": "CIT0059"
},
{
"DOI": "10.21203/rs.3.rs-94509/v1",
"doi-asserted-by": "crossref",
"key": "CIT0060",
"unstructured": "Frontera JA, Rahimian JO, Yaghi S, et al. Treatment with zinc is associated with reduced in-hospital mortality among COVID-19 patients: a multi-center cohort study. Res Sq 2020."
},
{
"DOI": "10.1056/NEJMoa2019014",
"doi-asserted-by": "publisher",
"key": "CIT0061"
},
{
"DOI": "10.1001/jamacardio.2020.1834",
"doi-asserted-by": "publisher",
"key": "CIT0062"
},
{
"DOI": "10.1016/j.hrthm.2020.05.008",
"doi-asserted-by": "publisher",
"key": "CIT0063"
},
{
"DOI": "10.3390/ijms22010326",
"author": "Barth LM",
"doi-asserted-by": "crossref",
"first-page": "326",
"journal-title": "Int J Mol Sci",
"key": "CIT0064",
"volume": "22",
"year": "2020"
},
{
"DOI": "10.1016/j.jinorgbio.2021.111661",
"doi-asserted-by": "publisher",
"key": "CIT0065"
},
{
"DOI": "10.1021/acs.jmedchem.2c00303",
"doi-asserted-by": "publisher",
"key": "CIT0066"
},
{
"DOI": "10.1038/s41392-020-00451-w",
"author": "Zhao MM",
"doi-asserted-by": "crossref",
"first-page": "1",
"journal-title": "Signal Transduct Target Ther",
"key": "CIT0067",
"volume": "6",
"year": "2021"
},
{
"DOI": "10.1016/j.pharmthera.2020.107587",
"doi-asserted-by": "publisher",
"key": "CIT0068"
},
{
"DOI": "10.1038/s41467-021-21060-3",
"doi-asserted-by": "publisher",
"key": "CIT0069"
},
{
"DOI": "10.1039/C8DT03905D",
"doi-asserted-by": "publisher",
"key": "CIT0070"
},
{
"DOI": "10.3390/ijms20143602",
"doi-asserted-by": "publisher",
"key": "CIT0071"
},
{
"author": "Mirza MU",
"first-page": "107376",
"journal-title": "J Bioinform Comput Biol",
"key": "CIT0072",
"volume": "89",
"year": "2020"
},
{
"DOI": "10.1039/D1SC04515F",
"doi-asserted-by": "publisher",
"key": "CIT0073"
},
{
"DOI": "10.1016/j.antiviral.2014.12.015",
"doi-asserted-by": "publisher",
"key": "CIT0074"
},
{
"DOI": "10.1021/acsmedchemlett.9b00039",
"doi-asserted-by": "publisher",
"key": "CIT0075"
},
{
"DOI": "10.1038/srep42717",
"doi-asserted-by": "publisher",
"key": "CIT0076"
},
{
"DOI": "10.1016/B978-0-12-802447-8.00017-0",
"doi-asserted-by": "publisher",
"key": "CIT0077"
},
{
"DOI": "10.1517/17425247.2.3.419",
"doi-asserted-by": "publisher",
"key": "CIT0078"
},
{
"DOI": "10.1208/s12248-015-9814-9",
"doi-asserted-by": "publisher",
"key": "CIT0079"
},
{
"DOI": "10.1101/2022.03.03.482819",
"doi-asserted-by": "crossref",
"key": "CIT0080",
"unstructured": "Kladnik J, Dolinar A, Kljun J, et al. Zinc pyrithione is a potent inhibitor of PLPro and cathepsin L enzymes with ex vivo inhibition of SARS-CoV-2 entry and replication. bioRxiv; 2022."
}
],
"reference-count": 80,
"references-count": 80,
"relation": {},
"resource": {
"primary": {
"URL": "https://www.tandfonline.com/doi/full/10.1080/14756366.2022.2108417"
}
},
"score": 1,
"short-title": [],
"source": "Crossref",
"subject": [
"Drug Discovery",
"Pharmacology",
"General Medicine"
],
"subtitle": [],
"title": "Zinc pyrithione is a potent inhibitor of PL<sup>Pro</sup> and cathepsin L enzymes with <i>ex vivo</i> inhibition of SARS-CoV-2 entry and replication",
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1080/tandf_crossmark_01",
"volume": "37"
}
