Fluvoxamine reduces COVID-19 risk: real-time meta-analysis of 21 studies (Version 39)
, May 2026
30th treatment shown to reduce risk in
November 2021, now with p = 0.00014 from 21 studies, recognized in 2 countries.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
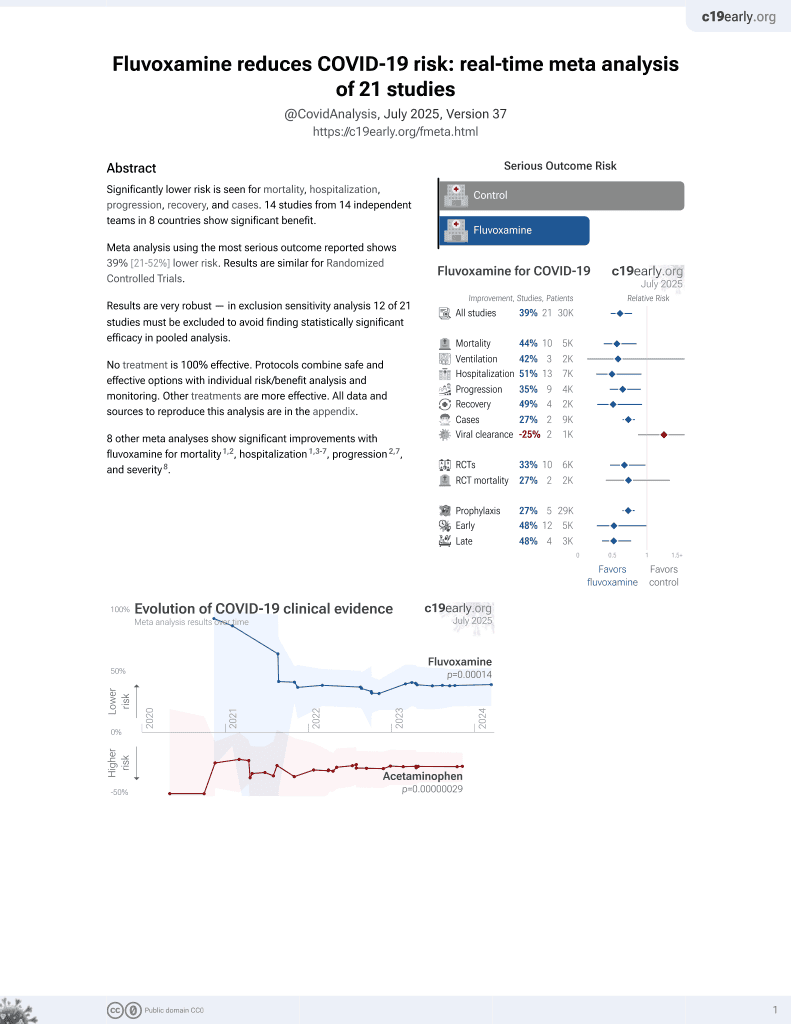
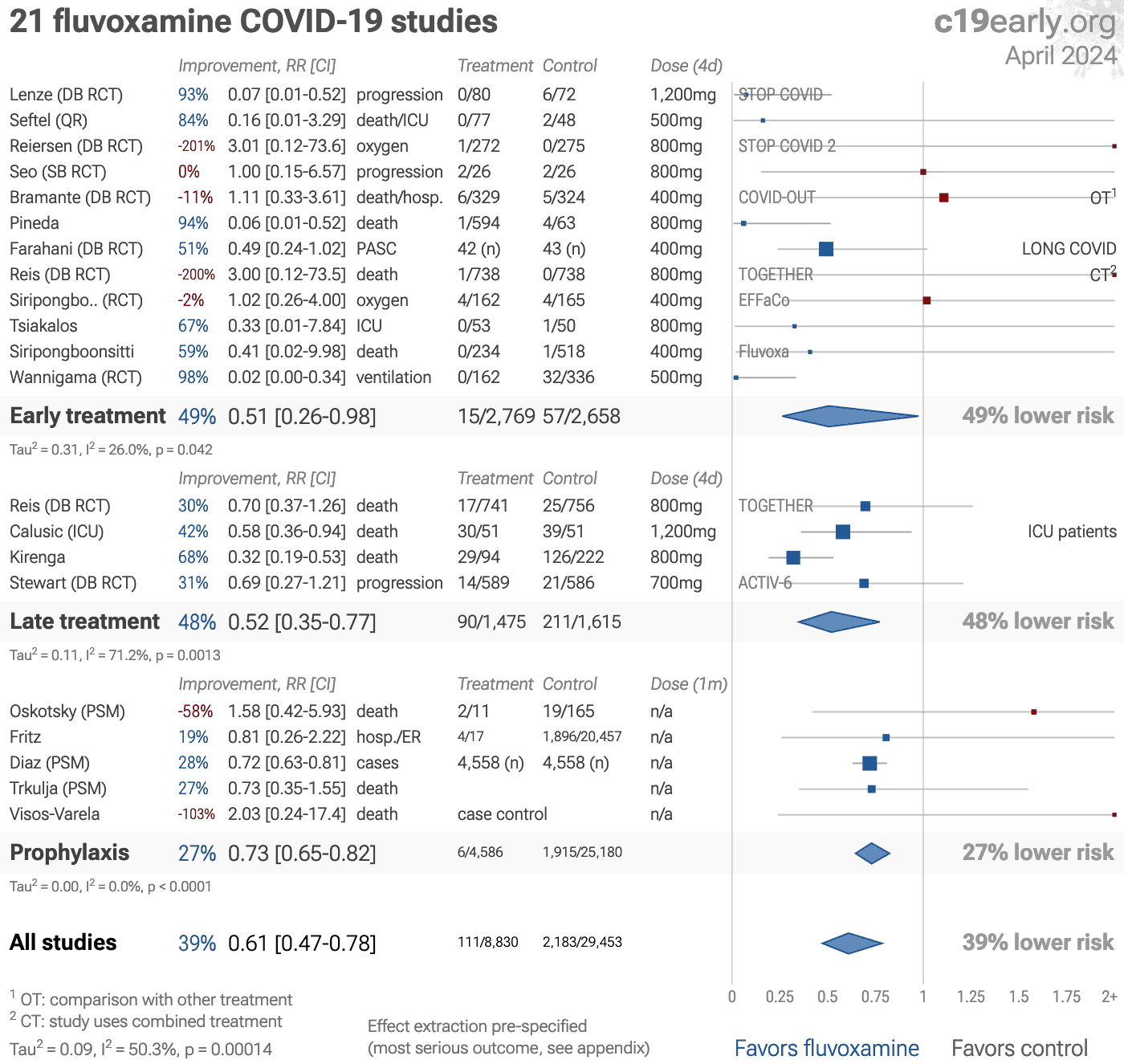
Significantly lower risk is seen for mortality, hospitalization, progression, recovery, and cases. 14 studies from 14 independent teams in 8 countries show significant benefit.
Meta-analysis using the most serious outcome reported shows 39% [22‑53%] lower risk. Results are similar for Randomized Controlled Trials.
Results are very robust—in worst case exclusion sensitivity analysis 12 of 21 studies must be excluded before statistical significance is lost.
Control Fluvoxamine
No treatment is 100% effective. Protocols combine safe and effective options with individual risk/benefit analysis and monitoring. Other treatments are more effective. All data and sources to reproduce this analysis are in the appendix.
9 other meta-analyses show significant improvements with fluvoxamine for mortality1-3, hospitalization1,4-8, progression2,8, and severity9.
9 meta-analyses show significant improvements with fluvoxamine for mortality1-3,
hospitalization1,4-8 ,
progression2,8, and
severity9.
1.
Deng et al., Efficacy and safety of selective serotonin reuptake inhibitors in COVID-19 management: A systematic review and meta-analysis, Clinical Microbiology and Infection, doi:10.1016/j.cmi.2023.01.010.
2.
Prasanth et al., A systematic review and meta-analysis, investigating dose and time of fluvoxamine treatment efficacy for COVID-19 clinical deterioration, death, and Long-COVID complications, Scientific Reports, doi:10.1038/s41598-024-64260-9.
3.
Fico et al., Psychotropic drug repurposing for COVID-19: A Systematic Review and Meta-Analysis, European Neuropsychopharmacology, doi:10.1016/j.euroneuro.2022.10.004.
4.
Lee et al., Fluvoxamine for Outpatient Management of COVID-19 to Prevent Hospitalization: A Systematic Review and Meta-analysis, JAMA Network Open, doi:10.1001/jamanetworkopen.2022.6269.
5.
Lu et al., Effect of fluvoxamine on outcomes of nonhospitalized patients with COVID-19: A systematic review and meta-analysis, Journal of Infection and Public Health, doi:10.1016/j.jiph.2022.10.010.
6.
Marcec et al., A meta-analysis regarding fluvoxamine and hospitalization risk of COVID-19 patients: TOGETHER making a difference, Journal of Infection, doi:10.1016/j.jinf.2022.11.011.
7.
Deng (B) et al., Evaluating fluvoxamine for the outpatient treatment of COVID‐19: A systematic review and meta‐analysis, Reviews in Medical Virology, doi:10.1002/rmv.2501.
Covid Analysis et al., May 2026, preprint, 1 author.