Association of metformin with mortality or ARDS in patients with COVID-19 and type 2 diabetes: A retrospective cohort study
et al., Diabetes Research and Clinical Practice, doi:10.1016/j.diabres.2020.108619, Mar 2021
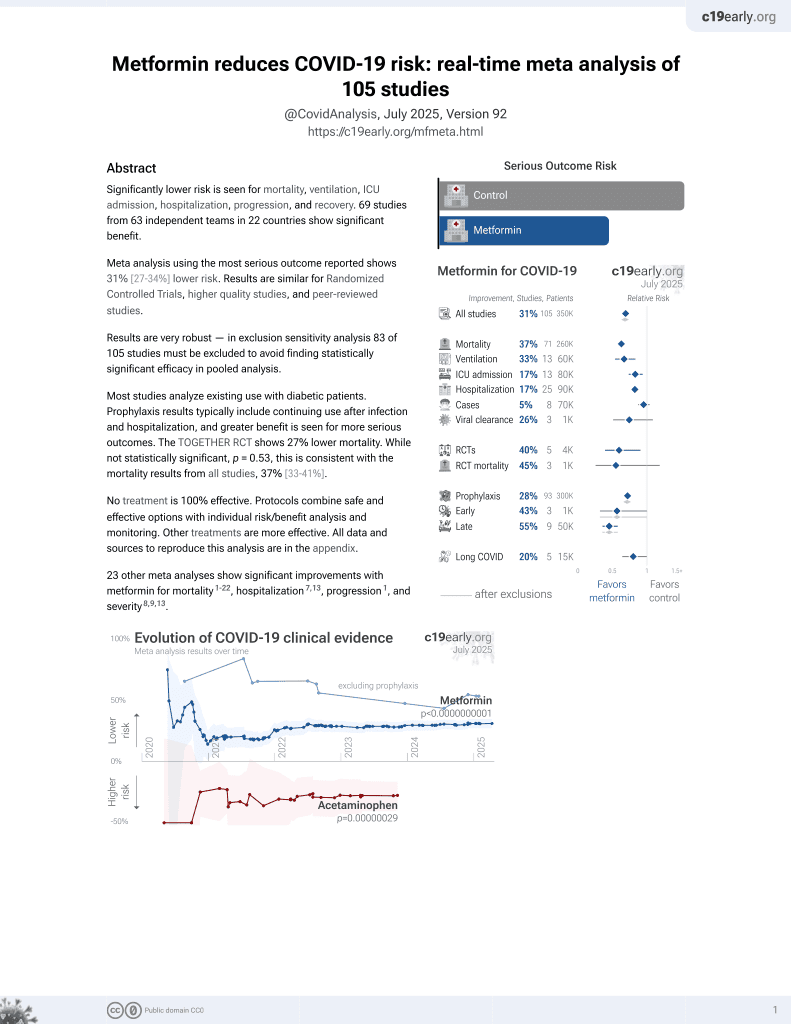
Metformin for COVID-19
3rd treatment shown to reduce risk in
July 2020, now with p < 0.00000000001 from 111 studies.
Lower risk for mortality, ventilation, ICU, hospitalization, progression, recovery, and viral clearance.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
Retrospective 328 COVID-19 patients with type 2 diabetes in China, showing significantly lower risk of ARDS with existing metformin use.
Standard of Care (SOC) for COVID-19 in the study country,
China, is poor with low average efficacy for approved treatments1.
|
risk of death, 46.0% lower, HR 0.54, p = 0.40, treatment 3 of 74 (4.1%), control 10 of 74 (13.5%), adjusted per study, mixed effect Cox, propensity score matching.
|
|
risk of ARDS, 80.2% lower, RR 0.20, p = 0.02, treatment 8 of 74 (10.8%), control 17 of 74 (23.0%), NNT 8.2, adjusted per study, odds ratio converted to relative risk, mixed effect Cox, propensity score matching.
|
| Effect extraction follows pre-specified rules prioritizing more serious outcomes. Submit updates |
Jiang et al., 31 Mar 2021, retrospective, China, peer-reviewed, 12 authors.
Association of metformin with mortality or ARDS in patients with COVID-19 and type 2 diabetes: A retrospective cohort study
Diabetes Research and Clinical Practice, doi:10.1016/j.diabres.2020.108619
To determine the association between metformin use and mortality and ARDS incidence in patients with COVID-19 and type 2 diabetes. Methods: This study was a multi-center retrospective analysis of COVID-19 patients with type 2 diabetes and admitted to four hospitals in Hubei province, China from December 31st, 2019 to March 31st, 2020. Patients were divided into two groups according to their exposure to metformin during hospitalization. The outcomes of interest were 30-day allcause mortality and incidence of ARDS. We used mixed-effect Cox model and random effect logistic regression to evaluate the associations of metformin use with outcomes, adjusted for baseline characteristics. Results: Of 328 patients with COVID-19 and type 2 diabetes included in the study cohort, 30.5% (100/328) were in the metformin group. In the mixed-effected model, metformin use was associated with the lower incidence of ARDS. There was no significant association between metformin use and 30-day all-cause mortality. Propensity score-matched analysis
Authors' Contributions ZC, NJ, XY, LL, NX, and YG, were responsible for the conception, design, and writing of the manuscript. HY, XT, JW, HL, and MT were responsible for the acquisition of data and literature research. ZC, NJ, XY, LL, ZL were responsible for the analysis and interpretation of data. All authors reviewed and revised the manuscript and approved the final version.
Appendix A. Supplementary material Supplementary data to this article can be found online at https://doi.org/10.1016/j.diabres.2020.108619.
R E F E R E N C E S
References
Bode, Garrett, Messler, Mcfarland, Crowe et al., Glycemic characteristics and clinical outcomes of COVID-19 patients hospitalized in the United States, J Diabetes Sci Technol, doi:10.1177/1932296820924469
Bornstein, Rubino, Khunti, Mingrone, Hopkins et al., Practical recommendations for the management of diabetes in patients with COVID-19, Lancet Diabetes Endocrinol, doi:10.1016/S2213-8587(20)30152-2
Bramante, Ingraham, Murray, Marmor, Hoversten et al., Observational Study of Metformin and Risk of Mortality in Patients Hospitalized with Covid-19, doi:10.1101/2020.06.19.20135095
Cheng, Liu, Li, Zhang, Lei et al., Metformin is associated with higher incidence of acidosis, but not mortality, in individuals with COVID-19 and pre-existing Type 2 diabetes, Cell Metab, doi:10.1016/j.cmet.2020.08.013
Do, Kim, Park, Cho, Kang, Is there an association between metformin use and clinical outcomes in diabetes patients with COVID-19?, Diabetes Metab
Hariyanto, Kurniawan, Metformin use is associated with reduced mortality rate from coronavirus disease 2019 (COVID-19) infection, Obes Med, doi:10.1016/j.obmed.2020.100290
Hyun, Shin, Lee, Metformin down-regulates TNF-a secretion via suppression of scavenger receptors in macrophages, Immune Netw, doi:10.4110/in.2013.13.4.123
Katulanda, Dissanayake, Ranathunga, Ratnasamy, Wijewickrama et al., Prevention and management of COVID-19 among patients with diabetes: an appraisal of the literature, Diabetologia, doi:10.1007/s00125-020-05164-x
Kumar, Arora, Sharma, Anikhindi, Bansal et al., Is diabetes mellitus associated with mortality and severity of COVID-19? A meta-analysis, Diabetes Metab Syndr, doi:10.1016/j.dsx.2020.04.044
Li, Ma, Acute respiratory failure in COVID-19: is it ''typical" ARDS?, Crit Care, doi:10.1186/s13054-020-02911-9
Luo, Qiu, Liu, Liu, Zheng et al., Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis, Am J Trop Med Hyg, doi:10.4269/ajtmh.20-0375
Oh, Song, Prior metformin therapy and 30-day mortality in patients with acute respiratory distress syndrome: a nationwide cohort study, Ann Palliat Med, doi:10.21037/apm.2020.04.25
Pe ´rez-Belmonte, Torres-Pen ˜a, Lo ´pez-Carmona, Ayala-Gutie ´rrez, Fuentes-Jime ´nez et al., Mortality and other adverse outcomes in patients with type 2 diabetes mellitus admitted for COVID-19 in association with glucose-lowering drugs: a nationwide cohort study, BMC Med, doi:10.1186/s12916-020-01832-2
Piwkowska, Rogacka, Jankowski, Dominiczak, Stepin ´ski et al., Metformin induces suppression of NAD(P)H oxidase activity in podocytes, Biochem Biophys Res Commun, doi:10.1016/j.bbrc.2010.01.119
Ranieri, Rubenfeld, Thompson, Ferguson, Caldwell, Acute respiratory distress syndrome: the Berlin Definition, JAMA, doi:10.1001/jama.2012.5669
Roncon, Zuin, Rigatelli, Zuliani, Diabetic patients with COVID-19 infection are at higher risk of ICU admission and poor short-term outcome, J Clin Virol, doi:10.1016/j.jcv.2020.104354
Saisho, Metformin and inflammation: its potential beyond glucose-lowering effect, Endocr Metab Immune Disord Drug Targets, doi:10.2174/1871530315666150316124019
Scheen, Metformin and COVID-19: from cellular mechanisms to reduced mortality, Diabetes Metab, doi:10.1016/j.diabet.2020.07.006
Schuiveling, Vazirpanah, Radstake, Zimmermann, Broen, Metformin, a new era for an old drug in the treatment of immune mediated disease?, Curr Drug Targets, doi:10.2174/1389450118666170613081730
Sharma, Ray, Sadasivam, Metformin in COVID-19: a possible role beyond diabetes, Diabetes Res Clin Pract, doi:10.1016/j.diabres.2020.108183
Simonnet, Chetboun, Poissy, Raverdy, Noulette et al., High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation, Silver Spring, doi:10.1002/oby.22831
Who, Coronavirus disease (COVID-9) outbreak situation
Who, Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases Interim guidance
Who, WHOTimeline-COVID-19
Wu, Mcgoogan, Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention, JAMA, doi:10.1001/jama.2020.2648
Yan, Yang, Wang, Ren, Zhang et al., Clinical characteristics and outcomes of patients with severe covid-19 with diabetes, BMJ Open Diabetes Res Care, doi:10.1136/bmjdrc-2020-001343
Yang, Zheng, Gou, Pu, Chen et al., Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis, Int J Infect Dis, doi:10.1016/j.ijid.2020.03.017
Ye, Wang, Mao, The pathogenesis and treatment of the 'Cytokine Storm' in COVID-19, J Infect, doi:10.1016/j.jinf.2020.03.037
Zangrillo, Beretta, Scandroglio, Monti, Fominskiy et al., Characteristics, treatment, outcomes and cause of death of invasively ventilated patients with COVID-19 ARDS in Milan, Italy, Crit Care Resusc
Zhang, Li, Niu, ACE2 and COVID-19 and the resulting ARDS, Postgrad Med J, doi:10.1136/postgradmedj-2020-137935
Zhang, Liu, Li, Luo, Liu et al., Antiinflammatory effects of matrine in LPS-induced acute lung injury in mice, Eur J Pharm Sci, doi:10.1016/j.ejps.2011.09.020
Zhu, She, Cheng, Qin, Zhang et al., Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes, Cell Metab, doi:10.1016/j.cmet.2020.04.021
Zumla, Hui, Azhar, Memish, Maeurer, Reducing mortality from 2019-nCoV: host-directed therapies should be an option, Lancet, doi:10.1016/S0140-6736(20)30305-6
DOI record:
{
"DOI": "10.1016/j.diabres.2020.108619",
"ISSN": [
"0168-8227"
],
"URL": "http://dx.doi.org/10.1016/j.diabres.2020.108619",
"alternative-id": [
"S0168822720308767"
],
"article-number": "108619",
"assertion": [
{
"label": "This article is maintained by",
"name": "publisher",
"value": "Elsevier"
},
{
"label": "Article Title",
"name": "articletitle",
"value": "Association of metformin with mortality or ARDS in patients with COVID-19 and type 2 diabetes: A retrospective cohort study"
},
{
"label": "Journal Title",
"name": "journaltitle",
"value": "Diabetes Research and Clinical Practice"
},
{
"label": "CrossRef DOI link to publisher maintained version",
"name": "articlelink",
"value": "https://doi.org/10.1016/j.diabres.2020.108619"
},
{
"label": "Content Type",
"name": "content_type",
"value": "article"
},
{
"label": "Copyright",
"name": "copyright",
"value": "© 2020 Elsevier B.V. All rights reserved."
}
],
"author": [
{
"affiliation": [],
"family": "Jiang",
"given": "Nan",
"sequence": "first"
},
{
"affiliation": [],
"family": "Chen",
"given": "Zhenyuan",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Liu",
"given": "Li",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yin",
"given": "Xiaoxv",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Yang",
"given": "Heping",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tan",
"given": "Xiangping",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Wang",
"given": "Jing",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Li",
"given": "Hui",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Tian",
"given": "Mengge",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Lu",
"given": "Zuxun",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Xiong",
"given": "Nian",
"sequence": "additional"
},
{
"affiliation": [],
"family": "Gong",
"given": "Yanhong",
"sequence": "additional"
}
],
"container-title": [
"Diabetes Research and Clinical Practice"
],
"content-domain": {
"crossmark-restriction": true,
"domain": [
"diabetesresearchclinicalpractice.com",
"clinicalkey.jp",
"clinicalkey.com",
"clinicalkey.es",
"clinicalkey.fr",
"clinicalkey.com.au",
"elsevier.com",
"sciencedirect.com"
]
},
"created": {
"date-parts": [
[
2020,
12,
10
]
],
"date-time": "2020-12-10T22:27:21Z",
"timestamp": 1607639241000
},
"deposited": {
"date-parts": [
[
2021,
3,
31
]
],
"date-time": "2021-03-31T21:36:12Z",
"timestamp": 1617226572000
},
"indexed": {
"date-parts": [
[
2021,
12,
15
]
],
"date-time": "2021-12-15T05:42:08Z",
"timestamp": 1639546928093
},
"is-referenced-by-count": 9,
"issn-type": [
{
"type": "print",
"value": "0168-8227"
}
],
"issued": {
"date-parts": [
[
2021,
3
]
]
},
"language": "en",
"license": [
{
"URL": "https://www.elsevier.com/tdm/userlicense/1.0/",
"content-version": "tdm",
"delay-in-days": 0,
"start": {
"date-parts": [
[
2021,
3,
1
]
],
"date-time": "2021-03-01T00:00:00Z",
"timestamp": 1614556800000
}
}
],
"link": [
{
"URL": "https://api.elsevier.com/content/article/PII:S0168822720308767?httpAccept=text/xml",
"content-type": "text/xml",
"content-version": "vor",
"intended-application": "text-mining"
},
{
"URL": "https://api.elsevier.com/content/article/PII:S0168822720308767?httpAccept=text/plain",
"content-type": "text/plain",
"content-version": "vor",
"intended-application": "text-mining"
}
],
"member": "78",
"original-title": [],
"page": "108619",
"prefix": "10.1016",
"published": {
"date-parts": [
[
2021,
3
]
]
},
"published-print": {
"date-parts": [
[
2021,
3
]
]
},
"publisher": "Elsevier BV",
"reference": [
{
"key": "10.1016/j.diabres.2020.108619_b0005",
"unstructured": "WHO. Coronavirus disease (COVID-9) outbreak situation, https://www.who.int/emergencies/diseases/novel-coronavirus-2019; 2020 [accessed 4 May 2020]."
},
{
"key": "10.1016/j.diabres.2020.108619_b0010",
"unstructured": "WHO. WHOTimeline-COVID-19, https://www.who.int/news-room/detail/27-04-2020-who-timeline---covid-19; 2020 [accessed May 4, 00]."
},
{
"DOI": "10.1016/j.ijid.2020.03.017",
"article-title": "Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis",
"author": "Yang",
"doi-asserted-by": "crossref",
"first-page": "91",
"journal-title": "Int J Infect Dis",
"key": "10.1016/j.diabres.2020.108619_b0015",
"volume": "94",
"year": "2020"
},
{
"DOI": "10.1016/S2213-8587(20)30152-2",
"article-title": "Practical recommendations for the management of diabetes in patients with COVID-19",
"author": "Bornstein",
"doi-asserted-by": "crossref",
"first-page": "546",
"issue": "6",
"journal-title": "Lancet Diabetes Endocrinol",
"key": "10.1016/j.diabres.2020.108619_b0020",
"volume": "8",
"year": "2020"
},
{
"DOI": "10.1007/s00125-020-05164-x",
"article-title": "Prevention and management of COVID-19 among patients with diabetes: an appraisal of the literature",
"author": "Katulanda",
"doi-asserted-by": "crossref",
"first-page": "1440",
"issue": "8",
"journal-title": "Diabetologia",
"key": "10.1016/j.diabres.2020.108619_b0025",
"volume": "63",
"year": "2020"
},
{
"DOI": "10.1016/j.dsx.2020.04.044",
"article-title": "Is diabetes mellitus associated with mortality and severity of COVID-19? A meta-analysis",
"author": "Kumar",
"doi-asserted-by": "crossref",
"first-page": "535",
"issue": "4",
"journal-title": "Diabetes Metab Syndr",
"key": "10.1016/j.diabres.2020.108619_b0030",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.1001/jama.2020.2648",
"article-title": "Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention",
"author": "Wu",
"doi-asserted-by": "crossref",
"first-page": "1239",
"issue": "13",
"journal-title": "JAMA",
"key": "10.1016/j.diabres.2020.108619_b0035",
"volume": "323",
"year": "2020"
},
{
"DOI": "10.1016/j.cmet.2020.04.021",
"article-title": "Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes",
"author": "Zhu",
"doi-asserted-by": "crossref",
"first-page": "1068",
"issue": "6",
"journal-title": "Cell Metab",
"key": "10.1016/j.diabres.2020.108619_b0040",
"volume": "31",
"year": "2020"
},
{
"DOI": "10.1177/1932296820924469",
"article-title": "Glycemic characteristics and clinical outcomes of COVID-19 patients hospitalized in the United States",
"author": "Bode",
"doi-asserted-by": "crossref",
"first-page": "813",
"issue": "4",
"journal-title": "J Diabetes Sci Technol",
"key": "10.1016/j.diabres.2020.108619_b0045",
"volume": "14",
"year": "2020"
},
{
"DOI": "10.2174/1871530315666150316124019",
"article-title": "Metformin and inflammation: its potential beyond glucose-lowering effect",
"author": "Saisho",
"doi-asserted-by": "crossref",
"first-page": "196",
"issue": "3",
"journal-title": "Endocr Metab Immune Disord Drug Targets",
"key": "10.1016/j.diabres.2020.108619_b0050",
"volume": "15",
"year": "2015"
},
{
"DOI": "10.1016/j.jinf.2020.03.037",
"article-title": "The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19",
"author": "Ye",
"doi-asserted-by": "crossref",
"first-page": "607",
"issue": "6",
"journal-title": "J Infect",
"key": "10.1016/j.diabres.2020.108619_b0055",
"volume": "80",
"year": "2020"
},
{
"DOI": "10.1136/postgradmedj-2020-137935",
"article-title": "ACE2 and COVID-19 and the resulting ARDS",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "403",
"issue": "1137",
"journal-title": "Postgrad Med J",
"key": "10.1016/j.diabres.2020.108619_b0060",
"volume": "96",
"year": "2020"
},
{
"DOI": "10.1016/S0140-6736(20)30305-6",
"article-title": "Reducing mortality from 2019-nCoV: host-directed therapies should be an option",
"author": "Zumla",
"doi-asserted-by": "crossref",
"first-page": "e35",
"issue": "10224",
"journal-title": "Lancet",
"key": "10.1016/j.diabres.2020.108619_b0065",
"volume": "395",
"year": "2020"
},
{
"DOI": "10.4269/ajtmh.20-0375",
"article-title": "Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis",
"author": "Luo",
"doi-asserted-by": "crossref",
"first-page": "69",
"issue": "1",
"journal-title": "Am J Trop Med Hyg",
"key": "10.1016/j.diabres.2020.108619_b0070",
"volume": "103",
"year": "2020"
},
{
"key": "10.1016/j.diabres.2020.108619_b0075",
"unstructured": "WHO. Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases Interim guidance, https://www.who.int/publications-detail/laboratory-testing-for-2019-novel-coronavirus-in-suspected-human-cases-20200117; 2020 [accessed 1 May 2020]."
},
{
"key": "10.1016/j.diabres.2020.108619_b0080",
"unstructured": "National Health Commission of the People’s Republic of China. New Coronavirus Pneumonia Prevention and Control Program (version 7.0), http://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989/files/ce3e6945832a438eaae415350a8ce964.pdf; 2020 [accessed 29 April 2020]."
},
{
"author": "American Diabetes",
"first-page": "S13",
"issue": "Suppl 1",
"journal-title": "Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2018. Diabetes care",
"key": "10.1016/j.diabres.2020.108619_b0085",
"volume": "41",
"year": "2018"
},
{
"article-title": "Acute respiratory distress syndrome: the Berlin Definition",
"author": "ARDS Definition Task Force",
"first-page": "2526",
"issue": "23",
"journal-title": "JAMA",
"key": "10.1016/j.diabres.2020.108619_b0090",
"volume": "307",
"year": "2012"
},
{
"article-title": "Characteristics, treatment, outcomes and cause of death of invasively ventilated patients with COVID-19 ARDS in Milan, Italy",
"author": "Zangrillo",
"first-page": "200",
"issue": "3",
"journal-title": "Crit Care Resusc",
"key": "10.1016/j.diabres.2020.108619_b0095",
"volume": "22",
"year": "2020"
},
{
"DOI": "10.1186/s13054-020-02911-9",
"article-title": "Acute respiratory failure in COVID-19: is it “typical” ARDS?",
"author": "Li",
"doi-asserted-by": "crossref",
"first-page": "198",
"issue": "1",
"journal-title": "Crit Care",
"key": "10.1016/j.diabres.2020.108619_b0100",
"volume": "24",
"year": "2020"
},
{
"DOI": "10.2174/1389450118666170613081730",
"article-title": "Metformin, a new era for an old drug in the treatment of immune mediated disease?",
"author": "Schuiveling",
"doi-asserted-by": "crossref",
"first-page": "945",
"issue": "8",
"journal-title": "Curr Drug Targets",
"key": "10.1016/j.diabres.2020.108619_b0105",
"volume": "19",
"year": "2018"
},
{
"DOI": "10.1016/j.bbrc.2010.01.119",
"article-title": "Metformin induces suppression of NAD(P)H oxidase activity in podocytes",
"author": "Piwkowska",
"doi-asserted-by": "crossref",
"first-page": "268",
"issue": "2",
"journal-title": "Biochem Biophys Res Commun",
"key": "10.1016/j.diabres.2020.108619_b0110",
"volume": "393",
"year": "2010"
},
{
"DOI": "10.1016/j.ejps.2011.09.020",
"article-title": "Antiinflammatory effects of matrine in LPS-induced acute lung injury in mice",
"author": "Zhang",
"doi-asserted-by": "crossref",
"first-page": "573",
"issue": "5",
"journal-title": "Eur J Pharm Sci",
"key": "10.1016/j.diabres.2020.108619_b0115",
"volume": "44",
"year": "2011"
},
{
"DOI": "10.1016/j.diabres.2020.108183",
"article-title": "Metformin in COVID-19: a possible role beyond diabetes",
"author": "Sharma",
"doi-asserted-by": "crossref",
"first-page": "108183",
"journal-title": "Diabetes Res Clin Pract",
"key": "10.1016/j.diabres.2020.108619_b0120",
"volume": "164",
"year": "2020"
},
{
"DOI": "10.1016/j.diabet.2020.07.006",
"article-title": "Metformin and COVID-19: from cellular mechanisms to reduced mortality",
"author": "Scheen",
"doi-asserted-by": "crossref",
"first-page": "423",
"issue": "6",
"journal-title": "Diabetes Metab",
"key": "10.1016/j.diabres.2020.108619_b0125",
"volume": "46",
"year": "2020"
},
{
"DOI": "10.1016/j.cmet.2020.08.013",
"article-title": "Metformin is associated with higher incidence of acidosis, but not mortality, in individuals with COVID-19 and pre-existing Type 2 diabetes",
"author": "Cheng",
"doi-asserted-by": "crossref",
"first-page": "534",
"issue": "4",
"journal-title": "Cell Metab",
"key": "10.1016/j.diabres.2020.108619_b0130",
"volume": "32",
"year": "2020"
},
{
"DOI": "10.21037/apm.2020.04.25",
"article-title": "Prior metformin therapy and 30-day mortality in patients with acute respiratory distress syndrome: a nationwide cohort study",
"author": "Oh",
"doi-asserted-by": "crossref",
"first-page": "903",
"issue": "3",
"journal-title": "Ann Palliat Med",
"key": "10.1016/j.diabres.2020.108619_b0135",
"volume": "9",
"year": "2020"
},
{
"DOI": "10.1016/j.obmed.2020.100290",
"article-title": "Metformin use is associated with reduced mortality rate from coronavirus disease 2019 (COVID-19) infection",
"author": "Hariyanto",
"doi-asserted-by": "crossref",
"first-page": "100290",
"journal-title": "Obes Med",
"key": "10.1016/j.diabres.2020.108619_b0140",
"volume": "19",
"year": "2020"
},
{
"DOI": "10.1101/2020.06.19.20135095",
"doi-asserted-by": "crossref",
"key": "10.1016/j.diabres.2020.108619_b0145",
"unstructured": "Bramante C, Ingraham N, Murray T, Marmor S, Hoversten S, Gronski J, et al. Observational Study of Metformin and Risk of Mortality in Patients Hospitalized with Covid-19. medRxiv [Preprint]. 2020 Jun 28:2020.06.19.20135095. http://doi.org/10.1101/2020.06.19.20135095."
},
{
"DOI": "10.4110/in.2013.13.4.123",
"article-title": "Metformin down-regulates TNF-α secretion via suppression of scavenger receptors in macrophages",
"author": "Hyun",
"doi-asserted-by": "crossref",
"first-page": "123",
"issue": "4",
"journal-title": "Immune Netw",
"key": "10.1016/j.diabres.2020.108619_b0150",
"volume": "13",
"year": "2013"
},
{
"DOI": "10.1136/bmjdrc-2020-001343",
"article-title": "Clinical characteristics and outcomes of patients with severe covid-19 with diabetes",
"author": "Yan",
"doi-asserted-by": "crossref",
"issue": "1",
"journal-title": "BMJ Open Diabetes Res Care",
"key": "10.1016/j.diabres.2020.108619_b0155",
"volume": "8",
"year": "2020"
},
{
"article-title": "Is there an association between metformin use and clinical outcomes in diabetes patients with COVID-19?",
"author": "Do",
"first-page": "30159",
"issue": "20",
"journal-title": "Diabetes Metab",
"key": "10.1016/j.diabres.2020.108619_b0160",
"volume": "S1262–3636",
"year": "2020"
},
{
"DOI": "10.1002/oby.22831",
"article-title": "High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation",
"author": "Simonnet",
"doi-asserted-by": "crossref",
"first-page": "1195",
"issue": "7",
"journal-title": "Obesity (Silver Spring)",
"key": "10.1016/j.diabres.2020.108619_b0165",
"volume": "28",
"year": "2020"
},
{
"DOI": "10.1186/s12916-020-01832-2",
"article-title": "Mortality and other adverse outcomes in patients with type 2 diabetes mellitus admitted for COVID-19 in association with glucose-lowering drugs: a nationwide cohort study",
"author": "Pérez-Belmonte",
"doi-asserted-by": "crossref",
"first-page": "359",
"issue": "1",
"journal-title": "BMC Med",
"key": "10.1016/j.diabres.2020.108619_b0170",
"volume": "18",
"year": "2020"
},
{
"DOI": "10.1016/j.jcv.2020.104354",
"article-title": "Diabetic patients with COVID-19 infection are at higher risk of ICU admission and poor short-term outcome",
"author": "Roncon",
"doi-asserted-by": "crossref",
"first-page": "104354",
"journal-title": "J Clin Virol",
"key": "10.1016/j.diabres.2020.108619_b0175",
"volume": "127",
"year": "2020"
}
],
"reference-count": 35,
"references-count": 35,
"relation": {},
"score": 1,
"short-container-title": [
"Diabetes Research and Clinical Practice"
],
"short-title": [],
"source": "Crossref",
"subject": [
"Endocrinology",
"General Medicine",
"Endocrinology, Diabetes and Metabolism",
"Internal Medicine"
],
"subtitle": [],
"title": [
"Association of metformin with mortality or ARDS in patients with COVID-19 and type 2 diabetes: A retrospective cohort study"
],
"type": "journal-article",
"update-policy": "http://dx.doi.org/10.1016/elsevier_cm_policy",
"volume": "173"
}