Povidone-Iodine reduces COVID-19 risk: real-time meta-analysis of 21 studies (Version 40)
, May 2026
PVP-I for COVID-19
14th treatment shown to reduce risk in
February 2021, now with p = 0.0000000037 from 21 studies.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
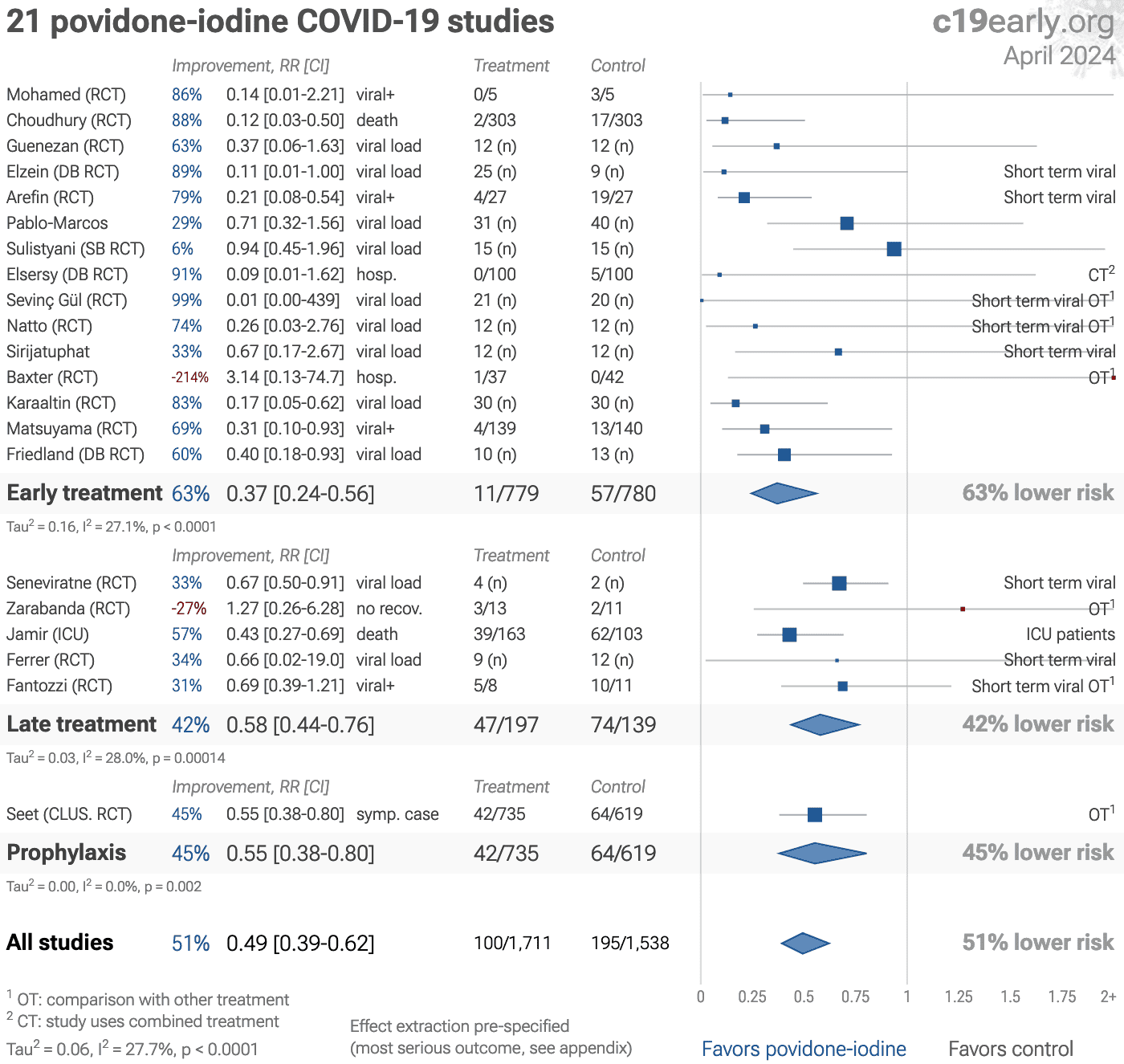
Significantly lower risk is seen for mortality, hospitalization, recovery, cases, and viral clearance. 11 studies from 11 independent teams in 9 countries show significant benefit.
Meta-analysis using the most serious outcome reported shows 48% [35‑58%] lower risk. Results are similar for Randomized Controlled Trials, higher quality studies, and peer-reviewed studies. Early treatment is more effective than late treatment.
Results are very robust—in worst case exclusion sensitivity analysis 17 of 21 studies must be excluded before statistical significance is lost. Emergent results matching biological mechanisms confirm efficacy: efficacy gradient across administration (p = 0.00022) and early vs. late treatment (p = 0.017).
Control Povidone-IodinePVP-I
2 RCTs with 295 patients have not reported results (up to 3 years late)1,2.
Excessive use of PVP-I could affect thyroid function.
No treatment is 100% effective. Protocols combine safe and effective options with individual risk/benefit analysis and monitoring. Povidone-Iodine may be detrimental to the natural microbiome, raising concern for side effects, especially with prolonged or excessive use. All data and sources to reproduce this analysis are in the appendix.
Other meta-analyses show significant improvements with povidone-iodine for viral load3-5 and viral clearance3.
3 meta-analyses show significant improvements with povidone-iodine for viral load1-3 and
viral clearance1.
1.
Hasan et al., Effects of Chlorhexidine and Povidone-Iodine on the SARS-CoV-2 Load: A Systematic Review and Meta-analysis, European Journal of Dentistry, doi:10.1055/s-0042-1753470.
Covid Analysis et al., May 2026, preprint, 1 author.
