Regdanvimab reduced COVID-19 risk: real-time meta-analysis of 12 studies (Version 11)
, May 2026
39th treatment shown to reduce risk in
March 2022, now with p = 0.00049 from 12 studies, recognized in 27 countries.
Efficacy is variant dependent.
No treatment is 100% effective. Protocols
combine treatments.
6,600+ studies for
220+ treatments. c19early.org
|
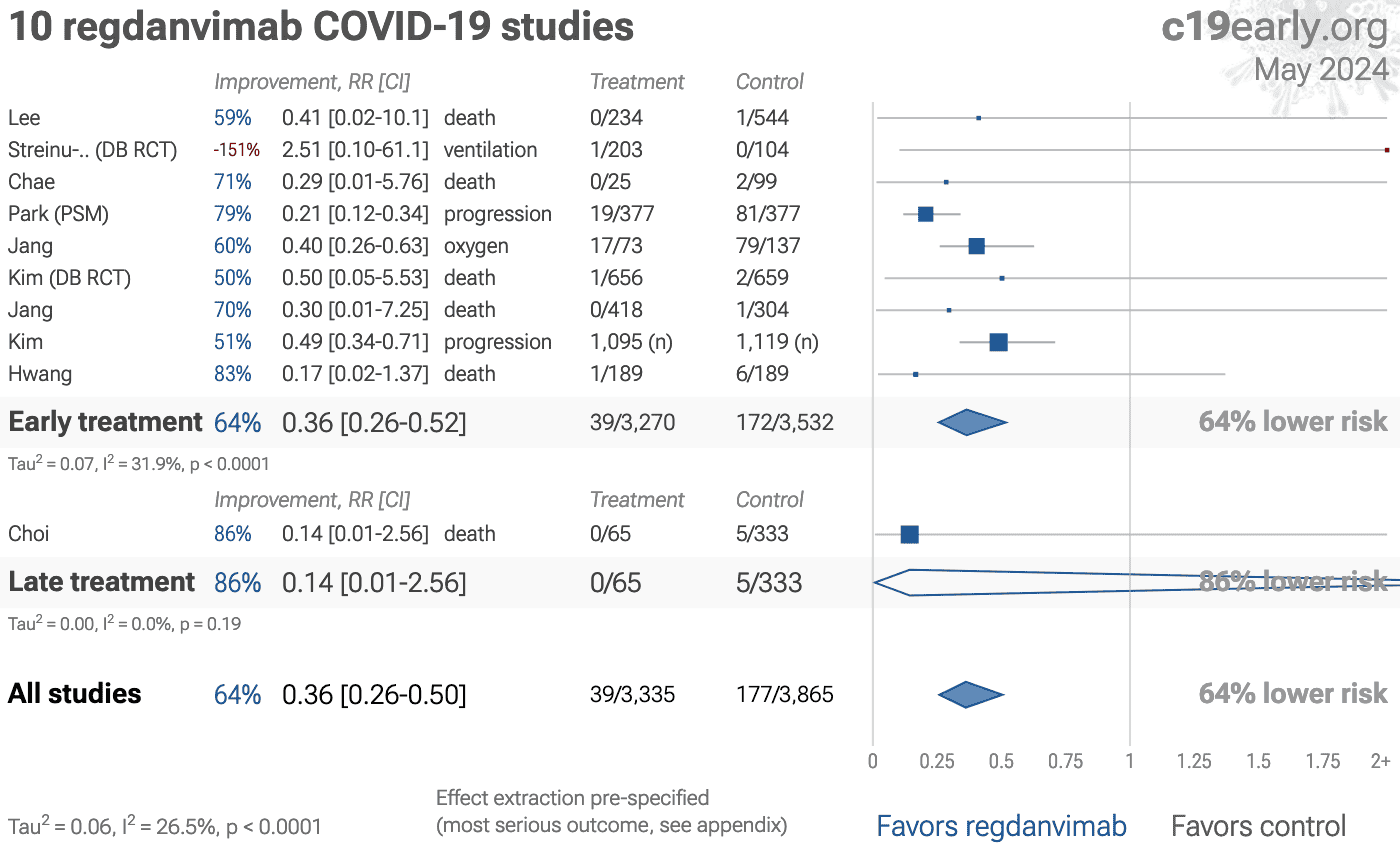
Significantly lower risk is seen for mortality, hospitalization, progression, and recovery. 10 studies from 7 independent teams (all from the same country) show significant benefit.
Meta-analysis using the most serious outcome reported shows 55% [30‑72%] lower risk. Results are similar for higher quality studies and worse for Randomized Controlled Trials.
Results are robust—in worst case exclusion sensitivity analysis 6 of 12 studies must be excluded before statistical significance is lost.
Control Regdanvimab
Efficacy is variant dependent. In Vitro research suggests a lack of efficacy for omicron BA.2, BA.4, BA.51, XBB.1.9.1, XBB.1.9.3, XBB.1.5.24, XBB.1.16, XBB.2.9, BQ.1.1.45, CL.1, and CH.1.12. mAb use may create new variants that spread globally3-5, and may be associated with increased risk of autoimmune disease6, prolonged viral loads, clinical deterioration, and immune escape4,7-11.
No treatment is 100% effective. Protocols combine safe and effective options with individual risk/benefit analysis and monitoring. All data and sources to reproduce this analysis are in the appendix.
Covid Analysis et al., May 2026, preprint, 1 author.
