Comparative risk of incidence and clinical outcomes of COVID-19 among proton pump inhibitor and histamine-2 receptor antagonist short-term users: a nationwide retrospective cohort study
Jimyung Park, Seng Chan You, Jaehyeong Cho, Chan Hyuk Park, Woon Geon Shin, Rae Woong Park, Seung In Seo
BMC Pharmacology and Toxicology, doi:10.1186/s40360-022-00549-7
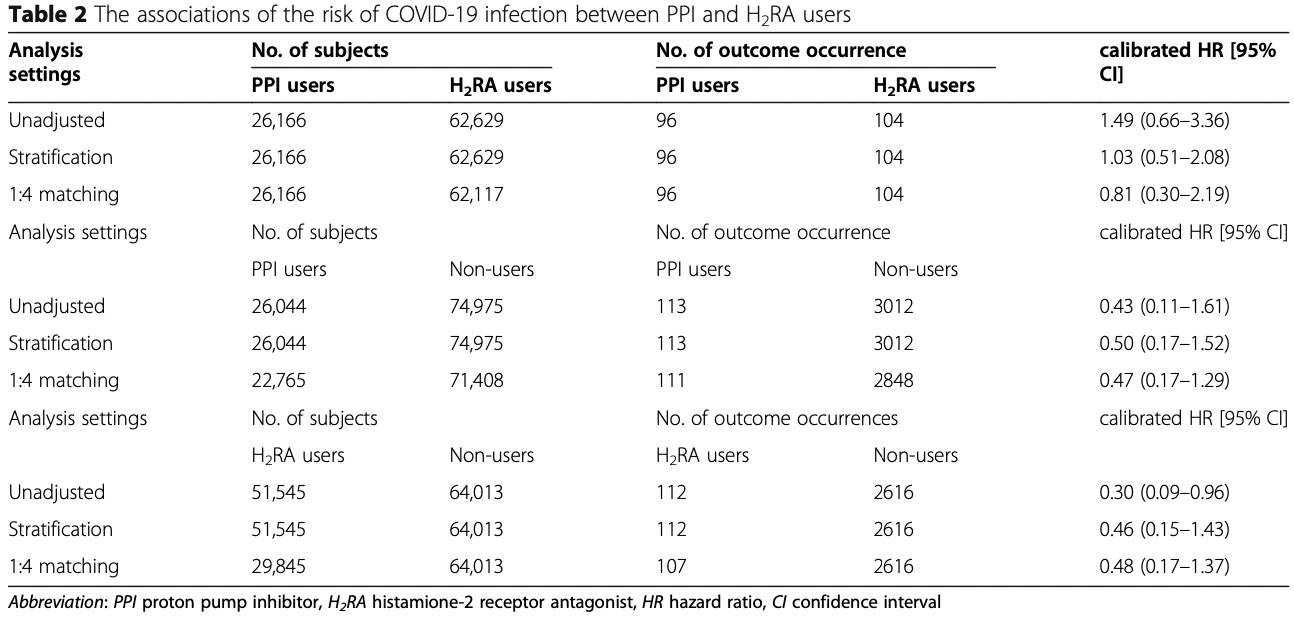
Background: This study aimed to evaluate incidence risk and adverse clinical outcomes in COVID-19 disease among short-term users of acid-suppressants in South Korea. Methods: This retrospective cohort study, conducted using a nationwide claims database for South Korea, used data from patients with COVID-19 tested between January 1 and May 15, 2020. Patients aged over 18 years and prescribed proton pump inhibitors (PPI) or histamine-2 receptor antagonist (H 2 RA) for more than 7 days were identified. Primary outcome was COVID-19 while secondary outcomes were all-cause mortality, hospitalization with respiratory disease, or intensive respiratory intervention. Large-scale propensity scores were used to match patients, while the Cox proportional hazard model was utilized to evaluate any association between exposure and outcome(s). The risk estimates were calibrated by using 123 negative control outcomes. Results: We identified 26,166 PPI users and 62,117 H 2 RA users. After propensity score matching, compared to H 2 RA use, PPI use was not significantly associated with lower risk of COVID-19 (calibrated hazard ratio [HR], 0.81 [95% confidence interval (CI), 0.30-2.19]); moreover, PPI use was not associated with adverse clinical outcomes in COVID-19, namely, hospitalization with respiratory disease (calibrated HR, 0.88 [95% CI, 0.72-1.08]), intensive respiratory interventions (calibrated HR, 0.92 [95% CI,), except for all-cause mortality (calibrated HR, 0.54 [95% CI, 0.31-0.95]).
Supplementary Information The online version contains supplementary material available at https://doi. org/10.1186/s40360-022-00549-7.
Additional file 1. Authors' contributions JP, SCY, JC, RWP, SIS, CHP, WGS contributed study concept and design. JP contributed statistical analysis of the study. JP, SCY, JC, SIS, and RWP contributed analysis and interpretation of data, drafting of the manuscript. RWP, SIS, WGS contributed obtaining funding. RWP and SIS contributed supervision of the study. JP and SCY contributed equally to this work. RWP and SIS contributed equally to this work. All author(s) read and approved the final manuscript.
Declarations Ethics and approval and consent to participate The Institutional review board of the Kangdong Sacred Heart Hospital has approved study and informed consent waiver. All methods were carried out in accordance with relevant guidelines and regulations.
Consent for publication Not applicable.
Competing interests The authors declare that they have no competing interests.
Publisher's Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Austin, An introduction to propensity score methods for reducing the effects of confounding in observational studies, Multivar Behav Res,
doi:10.1080/00273171.2011.568786Barkun, Bardou, Pham, Proton pump inhibitors vs. histamine 2 receptor antagonists for stress-related mucosal bleeding prophylaxis in critically ill patients: a meta-analysis, Am J Gastroenterol,
doi:10.1038/ajg.2011.474Charpiat, Bleyzac, Tod, Proton pump inhibitors are risk factors for viral infections: even for COVID-19?, Clin Drug Investig,
doi:10.1007/s40261-020-00963-xEom, Jeon, Lim, Use of acid-suppressive drugs and risk of pneumonia: a systematic review and meta-analysis, CMAJ,
doi:10.1503/cmaj.092129Gulmez, Holm, Frederiksen, Use of proton pump inhibitors and the risk of community-acquired pneumonia: a population-based casecontrol study, Arch Intern Med,
doi:10.1001/archinte.167.9.950Ho, Teng, Yang, Yeh, Wang et al., Association of Proton Pump Inhibitors Usage with risk of pneumonia in dementia patients, J Am Geriatr Soc,
doi:10.1111/jgs.14813Laheij, Sturkenboom, Hassing, Risk of community-acquired pneumonia and use of gastric acid-suppressive drugs, Jama,
doi:10.1001/jama.292.16.1955Lee, Ha, Yeniova, Severe clinical outcomes of COVID-19 associated with proton pump inhibitors: a nationwide cohort study with propensity score matching, Gut,
doi:10.1136/gutjnl-2020-322248Lipsitch, Tchetgen, Cohen, Negative controls: a tool for detecting confounding and bias in observational studies, Epidemiology,
doi:10.1097/EDE.0b013e3181d61eebLuxenburger, Sturm, Biever, Rieg, Duerschmied et al., Treatment with proton pump inhibitors increases the risk of secondary infections and ARDS in hospitalized patients with COVID-19: coincidence or underestimated risk factor?, J Intern Med,
doi:10.1111/joim.13121Maclaren, Reynolds, Allen, Histamine-2 receptor antagonists vs proton pump inhibitors on gastrointestinal tract hemorrhage and infectious complications in the intensive care unit, JAMA Intern Med,
doi:10.1001/jamainternmed.2013.14673Malfertheiner, Kandulski, Venerito, Proton-pump inhibitors: understanding the complications and risks, Nat Rev Gastroenterol Hepatol,
doi:10.1038/nrgastro.2017.117Othman, Crooks, Card, Community acquired pneumonia incidence before and after proton pump inhibitor prescription: population based study, BMJ,
doi:10.1136/bmj.i5813Rho, Cho, Son, COVID-19 international collaborative research by the health insurance review and assessment service using its nationwide real-world data: database, outcomes, and implications, J Prev Med Public Health,
doi:10.3961/jpmph.20.616Savarino, Tosetti, Benedetto, Compare, Nardone, Appropriateness in prescribing PPIs: a position paper of the Italian Society of Gastroenterology (SIGE) -study section "digestive diseases in primary care, Dig Liver Dis,
doi:10.1016/j.dld.2018.07.004Schuemie, Ryan, Dumouchel, Interpreting observational studies: why empirical calibration is needed to correct p-values, Stat Med,
doi:10.1002/sim.5925Suchard, Simpson, Zorych, Massive parallelization of serial inference algorithms for a complex generalized linear model, ACM Trans Model Comput Simul,
doi:10.1145/2414416.2414791Taştemur, Ataseven, Is it possible to use Proton Pump Inhibitors in COVID-19 treatment and prophylaxis?, Med Hypotheses,
doi:10.1016/j.mehy.2020.110018Tian, Schuemie, Suchard, Evaluating large-scale propensity score performance through real-world and synthetic data experiments, Int J Epidemiol,
doi:10.1093/ije/dyy120Voss, Boyce, Ryan, Accuracy of an automated knowledge base for identifying drug adverse reactions, J Biomed Inform,
doi:10.1016/j.jbi.2016.12.005Zhang, Li, Wu, Analysis of the Effect of Proton-Pump Inhibitors on the Course of COVID-19, J Inflamm Res,
doi:10.2147/jir.s292303{ 'indexed': {'date-parts': [[2022, 4, 5]], 'date-time': '2022-04-05T10:36:37Z', 'timestamp': 1649154997324},
'reference-count': 23,
'publisher': 'Springer Science and Business Media LLC',
'issue': '1',
'license': [ { 'start': { 'date-parts': [[2022, 1, 17]],
'date-time': '2022-01-17T00:00:00Z',
'timestamp': 1642377600000},
'content-version': 'tdm',
'delay-in-days': 0,
'URL': 'https://creativecommons.org/licenses/by/4.0'},
{ 'start': { 'date-parts': [[2022, 1, 17]],
'date-time': '2022-01-17T00:00:00Z',
'timestamp': 1642377600000},
'content-version': 'vor',
'delay-in-days': 0,
'URL': 'https://creativecommons.org/licenses/by/4.0'}],
'content-domain': {'domain': ['link.springer.com'], 'crossmark-restriction': False},
'short-container-title': ['BMC Pharmacol Toxicol'],
'published-print': {'date-parts': [[2022, 12]]},
'abstract': '<jats:title>Abstract</jats:title><jats:sec>\n'
' <jats:title>Background</jats:title>\n'
' <jats:p>This study aimed to evaluate incidence risk and adverse clinical '
'outcomes in COVID-19 disease among short-term users of acid-suppressants in South '
'Korea.</jats:p>\n'
' </jats:sec><jats:sec>\n'
' <jats:title>Methods</jats:title>\n'
' <jats:p>This retrospective cohort study, conducted using a nationwide claims '
'database for South Korea, used data from patients with COVID-19 tested between January 1 and '
'May 15, 2020. Patients aged over 18\u2009years and prescribed proton pump inhibitors (PPI) or '
'histamine-2 receptor antagonist (H<jats:sub>2</jats:sub>RA) for more than 7\u2009days were '
'identified. Primary outcome was COVID-19 while secondary outcomes were all-cause mortality, '
'hospitalization with respiratory disease, or intensive respiratory intervention. Large-scale '
'propensity scores were used to match patients, while the Cox proportional hazard model was '
'utilized to evaluate any association between exposure and outcome(s). The risk estimates were '
'calibrated by using 123 negative control outcomes.</jats:p>\n'
' </jats:sec><jats:sec>\n'
' <jats:title>Results</jats:title>\n'
' <jats:p>We identified 26,166 PPI users and 62,117 H<jats:sub>2</jats:sub>RA '
'users. After propensity score matching, compared to H<jats:sub>2</jats:sub>RA use, PPI use '
'was not significantly associated with lower risk of COVID-19 (calibrated hazard ratio [HR], '
'0.81 [95% confidence interval (CI), 0.30–2.19]); moreover, PPI use was not associated with '
'adverse clinical outcomes in COVID-19, namely, hospitalization with respiratory disease '
'(calibrated HR, 0.88 [95% CI, 0.72–1.08]), intensive respiratory interventions (calibrated '
'HR, 0.92 [95% CI, 0.46–1.82]), except for all-cause mortality (calibrated HR, 0.54 [95% CI, '
'0.31–0.95]).</jats:p>\n'
' </jats:sec><jats:sec>\n'
' <jats:title>Conclusions</jats:title>\n'
' <jats:p>In this study, we found that the PPI user was not associated with '
'risk of COVID-19 compared to H<jats:sub>2</jats:sub>RA users. There was no significant '
'relationship between severe clinical outcomes of COVID-19 and exposure to PPI compared with '
'H<jats:sub>2</jats:sub>RA, except for all-cause mortality.</jats:p>\n'
' </jats:sec>',
'DOI': '10.1186/s40360-022-00549-7',
'type': 'journal-article',
'created': {'date-parts': [[2022, 1, 17]], 'date-time': '2022-01-17T12:02:53Z', 'timestamp': 1642420973000},
'update-policy': 'http://dx.doi.org/10.1007/springer_crossmark_policy',
'source': 'Crossref',
'is-referenced-by-count': 0,
'title': [ 'Comparative risk of incidence and clinical outcomes of COVID-19 among proton pump inhibitor '
'and histamine-2 receptor antagonist short-term users: a nationwide retrospective cohort study'],
'prefix': '10.1186',
'volume': '23',
'author': [ {'given': 'Jimyung', 'family': 'Park', 'sequence': 'first', 'affiliation': []},
{'given': 'Seng Chan', 'family': 'You', 'sequence': 'additional', 'affiliation': []},
{'given': 'Jaehyeong', 'family': 'Cho', 'sequence': 'additional', 'affiliation': []},
{'given': 'Chan Hyuk', 'family': 'Park', 'sequence': 'additional', 'affiliation': []},
{'given': 'Woon Geon', 'family': 'Shin', 'sequence': 'additional', 'affiliation': []},
{'given': 'Rae Woong', 'family': 'Park', 'sequence': 'additional', 'affiliation': []},
{'given': 'Seung', 'family': 'In Seo', 'sequence': 'additional', 'affiliation': []}],
'member': '297',
'published-online': {'date-parts': [[2022, 1, 17]]},
'reference': [ { 'issue': '9',
'key': '549_CR1',
'doi-asserted-by': 'publisher',
'first-page': '894',
'DOI': '10.1016/j.dld.2018.07.004',
'volume': '50',
'author': 'V Savarino',
'year': '2018',
'unstructured': 'Savarino V, Tosetti C, Benedetto E, Compare D, Nardone G. '
'Appropriateness in prescribing PPIs: a position paper of the Italian '
'Society of Gastroenterology (SIGE) - study section “digestive diseases '
'in primary care”. Dig Liver Dis. 2018;50(9):894–902. '
'https://doi.org/10.1016/j.dld.2018.07.004.',
'journal-title': 'Dig Liver Dis'},
{ 'issue': '12',
'key': '549_CR2',
'doi-asserted-by': 'publisher',
'first-page': '697',
'DOI': '10.1038/nrgastro.2017.117',
'volume': '14',
'author': 'P Malfertheiner',
'year': '2017',
'unstructured': 'Malfertheiner P, Kandulski A, Venerito M. Proton-pump inhibitors: '
'understanding the complications and risks. Nat Rev Gastroenterol '
'Hepatol. 2017;14(12):697–710. https://doi.org/10.1038/nrgastro.2017.117.',
'journal-title': 'Nat Rev Gastroenterol Hepatol'},
{ 'issue': '1',
'key': '549_CR3',
'doi-asserted-by': 'publisher',
'first-page': '35',
'DOI': '10.1053/j.gastro.2017.04.047',
'volume': '153',
'author': 'MF Vaezi',
'year': '2017',
'unstructured': 'Vaezi MF, Yang YX, Howden CW. Complications of proton pump inhibitor '
'therapy. Gastroenterology. 2017;153(1):35–48. '
'https://doi.org/10.1053/j.gastro.2017.04.047.',
'journal-title': 'Gastroenterology'},
{ 'issue': '9',
'key': '549_CR4',
'doi-asserted-by': 'publisher',
'first-page': '950',
'DOI': '10.1001/archinte.167.9.950',
'volume': '167',
'author': 'SE Gulmez',
'year': '2007',
'unstructured': 'Gulmez SE, Holm A, Frederiksen H, et al. Use of proton pump inhibitors '
'and the risk of community-acquired pneumonia: a population-based '
'case-control study. Arch Intern Med. 2007;167(9):950–5. '
'https://doi.org/10.1001/archinte.167.9.950 [published Online First: '
'2007/05/16].',
'journal-title': 'Arch Intern Med'},
{ 'issue': '16',
'key': '549_CR5',
'doi-asserted-by': 'publisher',
'first-page': '1955',
'DOI': '10.1001/jama.292.16.1955',
'volume': '292',
'author': 'RJ Laheij',
'year': '2004',
'unstructured': 'Laheij RJ, Sturkenboom MC, Hassing RJ, et al. Risk of community-acquired '
'pneumonia and use of gastric acid-suppressive drugs. Jama. '
'2004;292(16):1955–60. https://doi.org/10.1001/jama.292.16.1955 '
'[published Online First: 2004/10/28].',
'journal-title': 'Jama'},
{ 'issue': '4',
'key': '549_CR6',
'doi-asserted-by': 'publisher',
'first-page': '507',
'DOI': '10.1038/ajg.2011.474',
'volume': '107',
'author': 'AN Barkun',
'year': '2012',
'unstructured': 'Barkun AN, Bardou M, Pham CQ, et al. Proton pump inhibitors vs. '
'histamine 2 receptor antagonists for stress-related mucosal bleeding '
'prophylaxis in critically ill patients: a meta-analysis. Am J '
'Gastroenterol. 2012;107(4):507–20. https://doi.org/10.1038/ajg.2011.474 '
'quiz 21. [published Online First: 2012/02/01].',
'journal-title': 'Am J Gastroenterol'},
{ 'issue': '3',
'key': '549_CR7',
'doi-asserted-by': 'publisher',
'first-page': '310',
'DOI': '10.1503/cmaj.092129',
'volume': '183',
'author': 'CS Eom',
'year': '2011',
'unstructured': 'Eom CS, Jeon CY, Lim JW, et al. Use of acid-suppressive drugs and risk '
'of pneumonia: a systematic review and meta-analysis. CMAJ. '
'2011;183(3):310–9. https://doi.org/10.1503/cmaj.092129 [published Online '
'First: 2010/12/22].',
'journal-title': 'CMAJ'},
{ 'issue': '7',
'key': '549_CR8',
'doi-asserted-by': 'publisher',
'first-page': '1441',
'DOI': '10.1111/jgs.14813',
'volume': '65',
'author': 'SW Ho',
'year': '2017',
'unstructured': 'Ho SW, Teng YH, Yang SF, Yeh HW, Wang YH, Chou MC, et al. Association of '
'Proton Pump Inhibitors Usage with risk of pneumonia in dementia '
'patients. J Am Geriatr Soc. 2017;65(7):1441–7. '
'https://doi.org/10.1111/jgs.14813.',
'journal-title': 'J Am Geriatr Soc'},
{ 'issue': '4',
'key': '549_CR9',
'doi-asserted-by': 'publisher',
'first-page': '564',
'DOI': '10.1001/jamainternmed.2013.14673',
'volume': '174',
'author': 'R MacLaren',
'year': '2014',
'unstructured': 'MacLaren R, Reynolds PM, Allen RR. Histamine-2 receptor antagonists vs '
'proton pump inhibitors on gastrointestinal tract hemorrhage and '
'infectious complications in the intensive care unit. JAMA Intern Med. '
'2014;174(4):564–74. https://doi.org/10.1001/jamainternmed.2013.14673 '
'[published Online First: 2014/02/19].',
'journal-title': 'JAMA Intern Med'},
{ 'key': '549_CR10',
'doi-asserted-by': 'publisher',
'first-page': 'i5813',
'DOI': '10.1136/bmj.i5813',
'volume': '355',
'author': 'F Othman',
'year': '2016',
'unstructured': 'Othman F, Crooks CJ, Card TR. Community acquired pneumonia incidence '
'before and after proton pump inhibitor prescription: population based '
'study. BMJ. 2016;355:i5813. https://doi.org/10.1136/bmj.i5813.',
'journal-title': 'BMJ'},
{ 'issue': '10',
'key': '549_CR11',
'doi-asserted-by': 'publisher',
'first-page': '1707',
'DOI': '10.14309/ajg.0000000000000798',
'volume': '115',
'author': 'CV Almario',
'year': '2020',
'unstructured': 'Almario CV, Chey WD, Spiegel BMR. Increased Risk of COVID-19 Among Users '
'of Proton Pump Inhibitors. Am J Gastroenterol. 2020;115(10):1707–15. '
'https://doi.org/10.14309/ajg.0000000000000798 [published Online First: '
'2020/08/28].',
'journal-title': 'Am J Gastroenterol'},
{ 'issue': '1',
'key': '549_CR12',
'doi-asserted-by': 'publisher',
'first-page': '76',
'DOI': '10.1136/gutjnl-2020-322248',
'volume': '70',
'author': 'SW Lee',
'year': '2021',
'unstructured': 'Lee SW, Ha EK, Yeniova A, et al. Severe clinical outcomes of COVID-19 '
'associated with proton pump inhibitors: a nationwide cohort study with '
'propensity score matching. Gut. 2021;70(1):76–84. '
'https://doi.org/10.1136/gutjnl-2020-322248 [published Online First: '
'2020/08/01].',
'journal-title': 'Gut'},
{ 'key': '549_CR13',
'doi-asserted-by': 'publisher',
'first-page': '287',
'DOI': '10.2147/jir.s292303',
'volume': '14',
'author': 'XY Zhang',
'year': '2021',
'unstructured': 'Zhang XY, Li T, Wu H, et al. Analysis of the Effect of Proton-Pump '
'Inhibitors on the Course of COVID-19. J Inflamm Res. 2021;14:287–98. '
'https://doi.org/10.2147/jir.s292303 [published Online First: '
'2021/02/13].',
'journal-title': 'J Inflamm Res'},
{ 'issue': '1',
'key': '549_CR14',
'doi-asserted-by': 'publisher',
'first-page': '121',
'DOI': '10.1111/joim.13121',
'volume': '289',
'author': 'H Luxenburger',
'year': '2020',
'unstructured': 'Luxenburger H, Sturm L, Biever P, Rieg S, Duerschmied D, Schultheiss M, '
'et al. Treatment with proton pump inhibitors increases the risk of '
'secondary infections and ARDS in hospitalized patients with COVID-19: '
'coincidence or underestimated risk factor? J Intern Med. '
'2020;289(1):121–4. https://doi.org/10.1111/joim.13121.',
'journal-title': 'J Intern Med'},
{ 'key': '549_CR15',
'doi-asserted-by': 'publisher',
'first-page': '110018',
'DOI': '10.1016/j.mehy.2020.110018',
'volume': '143',
'author': 'Ş Taştemur',
'year': '2020',
'unstructured': 'Taştemur Ş, Ataseven H. Is it possible to use Proton Pump Inhibitors in '
'COVID-19 treatment and prophylaxis? Med Hypotheses. 2020;143:110018. '
'https://doi.org/10.1016/j.mehy.2020.110018 [published Online First: '
'2020/07/18].',
'journal-title': 'Med Hypotheses'},
{ 'issue': '1',
'key': '549_CR16',
'doi-asserted-by': 'publisher',
'first-page': '8',
'DOI': '10.3961/jpmph.20.616',
'volume': '54',
'author': 'Y Rho',
'year': '2021',
'unstructured': 'Rho Y, Cho DY, Son Y, et al. COVID-19 international collaborative '
'research by the health insurance review and assessment service using its '
'nationwide real-world data: database, outcomes, and implications. J Prev '
'Med Public Health. 2021;54(1):8–16. https://doi.org/10.3961/jpmph.20.616 '
'[published Online First: 2021/02/24].',
'journal-title': 'J Prev Med Public Health'},
{ 'key': '549_CR17',
'doi-asserted-by': 'publisher',
'unstructured': 'Suchard MA, Simpson SE, Zorych I, et al. Massive parallelization of '
'serial inference algorithms for a complex generalized linear model. ACM '
'Trans Model Comput Simul. 2013;23(1). '
'https://doi.org/10.1145/2414416.2414791 [published Online First: '
'2013/01/01].',
'DOI': '10.1145/2414416.2414791'},
{ 'issue': '6',
'key': '549_CR18',
'doi-asserted-by': 'publisher',
'first-page': '2005',
'DOI': '10.1093/ije/dyy120',
'volume': '47',
'author': 'Y Tian',
'year': '2018',
'unstructured': 'Tian Y, Schuemie MJ, Suchard MA. Evaluating large-scale propensity score '
'performance through real-world and synthetic data experiments. Int J '
'Epidemiol. 2018;47(6):2005–14. https://doi.org/10.1093/ije/dyy120 '
'[published Online First: 2018/06/26].',
'journal-title': 'Int J Epidemiol'},
{ 'issue': '3',
'key': '549_CR19',
'doi-asserted-by': 'publisher',
'first-page': '399',
'DOI': '10.1080/00273171.2011.568786',
'volume': '46',
'author': 'PC Austin',
'year': '2011',
'unstructured': 'Austin PC. An introduction to propensity score methods for reducing the '
'effects of confounding in observational studies. Multivar Behav Res. '
'2011;46(3):399–424. https://doi.org/10.1080/00273171.2011.568786 '
'[published Online First: 2011/08/06].',
'journal-title': 'Multivar Behav Res'},
{ 'issue': '3',
'key': '549_CR20',
'doi-asserted-by': 'publisher',
'first-page': '383',
'DOI': '10.1097/EDE.0b013e3181d61eeb',
'volume': '21',
'author': 'M Lipsitch',
'year': '2010',
'unstructured': 'Lipsitch M, Tchetgen Tchetgen E, Cohen T. Negative controls: a tool for '
'detecting confounding and bias in observational studies. Epidemiology '
'(Cambridge, Mass). 2010;21(3):383–8. '
'https://doi.org/10.1097/EDE.0b013e3181d61eeb [published Online First: '
'2010/03/26].',
'journal-title': 'Epidemiology (Cambridge, Mass)'},
{ 'key': '549_CR21',
'doi-asserted-by': 'publisher',
'first-page': '72',
'DOI': '10.1016/j.jbi.2016.12.005',
'volume': '66',
'author': 'EA Voss',
'year': '2017',
'unstructured': 'Voss EA, Boyce RD, Ryan PB, et al. Accuracy of an automated knowledge '
'base for identifying drug adverse reactions. J Biomed Inform. '
'2017;66:72–81. https://doi.org/10.1016/j.jbi.2016.12.005 [published '
'Online First: 2016/12/21].',
'journal-title': 'J Biomed Inform'},
{ 'issue': '2',
'key': '549_CR22',
'doi-asserted-by': 'publisher',
'first-page': '209',
'DOI': '10.1002/sim.5925',
'volume': '33',
'author': 'MJ Schuemie',
'year': '2014',
'unstructured': 'Schuemie MJ, Ryan PB, DuMouchel W, et al. Interpreting observational '
'studies: why empirical calibration is needed to correct p-values. Stat '
'Med. 2014;33(2):209–18. https://doi.org/10.1002/sim.5925 [published '
'Online First: 2013/08/01].',
'journal-title': 'Stat Med'},
{ 'issue': '10',
'key': '549_CR23',
'doi-asserted-by': 'publisher',
'first-page': '897',
'DOI': '10.1007/s40261-020-00963-x',
'volume': '40',
'author': 'B Charpiat',
'year': '2020',
'unstructured': 'Charpiat B, Bleyzac N, Tod M. Proton pump inhibitors are risk factors '
'for viral infections: even for COVID-19? Clin Drug Investig. '
'2020;40(10):897–9. https://doi.org/10.1007/s40261-020-00963-x [published '
'Online First: 2020/08/12].',
'journal-title': 'Clin Drug Investig'}],
'container-title': ['BMC Pharmacology and Toxicology'],
'original-title': [],
'language': 'en',
'link': [ { 'URL': 'https://link.springer.com/content/pdf/10.1186/s40360-022-00549-7.pdf',
'content-type': 'application/pdf',
'content-version': 'vor',
'intended-application': 'text-mining'},
{ 'URL': 'https://link.springer.com/article/10.1186/s40360-022-00549-7/fulltext.html',
'content-type': 'text/html',
'content-version': 'vor',
'intended-application': 'text-mining'},
{ 'URL': 'https://link.springer.com/content/pdf/10.1186/s40360-022-00549-7.pdf',
'content-type': 'application/pdf',
'content-version': 'vor',
'intended-application': 'similarity-checking'}],
'deposited': { 'date-parts': [[2022, 1, 17]],
'date-time': '2022-01-17T12:03:08Z',
'timestamp': 1642420988000},
'score': 1,
'resource': { 'primary': { 'URL': 'https://bmcpharmacoltoxicol.biomedcentral.com/articles/10.1186/s40360-022-00549-7'}},
'subtitle': [],
'short-title': [],
'issued': {'date-parts': [[2022, 1, 17]]},
'references-count': 23,
'journal-issue': {'issue': '1', 'published-print': {'date-parts': [[2022, 12]]}},
'alternative-id': ['549'],
'URL': 'http://dx.doi.org/10.1186/s40360-022-00549-7',
'relation': {},
'ISSN': ['2050-6511'],
'issn-type': [{'value': '2050-6511', 'type': 'electronic'}],
'subject': ['Pharmacology (medical)', 'Pharmacology'],
'published': {'date-parts': [[2022, 1, 17]]},
'assertion': [ { 'value': '7 September 2021',
'order': 1,
'name': 'received',
'label': 'Received',
'group': {'name': 'ArticleHistory', 'label': 'Article History'}},
{ 'value': '11 January 2022',
'order': 2,
'name': 'accepted',
'label': 'Accepted',
'group': {'name': 'ArticleHistory', 'label': 'Article History'}},
{ 'value': '17 January 2022',
'order': 3,
'name': 'first_online',
'label': 'First Online',
'group': {'name': 'ArticleHistory', 'label': 'Article History'}},
{'order': 1, 'name': 'Ethics', 'group': {'name': 'EthicsHeading', 'label': 'Declarations'}},
{ 'value': 'The Institutional review board of the Kangdong Sacred Heart Hospital has '
'approved study and informed consent waiver. All methods were carried out in '
'accordance with relevant guidelines and regulations.',
'order': 2,
'name': 'Ethics',
'group': { 'name': 'EthicsHeading',
'label': 'Ethics and approval and consent to participate'}},
{ 'value': 'Not applicable.',
'order': 3,
'name': 'Ethics',
'group': {'name': 'EthicsHeading', 'label': 'Consent for publication'}},
{ 'value': 'The authors declare that they have no competing interests.',
'order': 4,
'name': 'Ethics',
'group': {'name': 'EthicsHeading', 'label': 'Competing interests'}}],
'article-number': '9'}